04 May 2021: Articles 
Rare Mixed Adenoneuroendocrine Carcinoma of the Gallbladder: Case Report and Review of Literature
Rare disease
Adriano C. Costa12ADE, Camilla L.C. Cavalcanti3BCF, Henrique G.B. Coelho4BDEF*, Luís Henrique A. Leão4CEF, Daniel Tenório Camelo Soares4BEF, Fernando Santa-Cruz4CDE, Alexandre R. Paz5BCD, Álvaro A.B. Ferraz36AEFDOI: 10.12659/AJCR.929511
Am J Case Rep 2021; 22:e929511
Abstract
BACKGROUND: Mixed adenoneuroendocrine carcinoma of the gallbladder (gMANEC) is an extremely rare cancer. Most of the cases are reported in Asia, North America, and Europe, with no cases reported in Latin America; this is the first report for this region, and the 24th case reported worldwide.
CASE REPORT: A 68-year-old woman was referred to our department due to asthenia and moderate abdominal pain in the right upper quadrant for 6 months, with imaging examinations showing a solid heterogeneous expansive lesion in gallbladder topography and segment IV of the liver. The MRI displayed an expansive and heterogeneous lesion with inaccurate limits in the gallbladder affecting segment IVb of the liver, in addition to lymphadenopathy in the hepatic hilum. A cholecystectomy with resection of segments IV-B and V of the liver (radical cholecystectomy) and hepatic hilar lymphadenectomy were performed. Anatomopathological examination and immunohistochemistry confirmed a primary mixed adenoneuroendocrine carcinoma of the gallbladder. The patient received adjuvant chemotherapy and radiotherapy; however, after the patient reported experiencing low back pain, a CT was performed, revealing retroperitoneal metastasis, and the radiotherapy was interrupted. Currently, the patient has a stable disease, following a protocol of 5-Fluorouracil and somatostatin, and she reports having low back pain of low intensity.
CONCLUSIONS: This is the 24th gMANEC case reported in the literature. The tumor was successfully resected; however, the patient presented retroperitoneal metastasis 6 months after surgery, despite combined adjuvant therapy.
Keywords: Carcinoma, Mixed Tumor, Malignant, neuroendocrine tumors, Cholecystectomy, Lymph Node Excision
Background
Papillary adenocarcinoma is the most common neoplasm of the gallbladder, accounting for about 98% of all cases, while critical neuroendocrine tumors account for only 0.5% of all neoplasms of this organ [1]. In 2019, the WHO classification of digestive system tumors classified the neoplasms composed of neuroendocrine and non-neuro endocrine components as mixed neuroendocrine non-neuroendocrine neoplasms (MINENs). Mixed adenoneuroendocrine carcinomas (MANEC) are an aggressive subtype of MINEN [2].
MANECs are even more difficult to diagnose as a primary tumor of the gallbladder. MANECs belong to a specific group of tumors that exhibit great heterogeneity, given that the percentages of adenocarcinomatous and neuroendocrine differentiation can vary according to their classification. To define the presence of MANEC, at least 30% of each tumor component must be determined [3]. In most cases, this tumor appears in the colon, appendix, rectum, or stomach, and is rarely found in the biliary tract, pancreas, or gallbladder [4,5].
Clinical symptoms of mixed adenoneuroendocrine carcinoma of the gallbladder (gMANEC) are nonspecific, such as epigastric or right hypochondrium pain, nausea, and vomiting [4]. However, asymptomatic cases are also reported [6–11]. These factors contribute to a late diagnosis, which worsens the prognosis and survival rates. The low number of reported cases limits the acknowledgement of primary gMANEC as a possible diagnosis.
Herein, we report a case of primary gMANEC, the first reported in Latin America and the 24th worldwide [5–22]. This case has been reported in line with the SCARE criteria [23].
Case Report
SURGICAL FINDINGS AND ANATOMOPATHOLOGICAL EXAMINATION:
During the surgery, an extensive tumor in the gallbladder was observed, extending to segments IV and V of the liver, as described in the preoperative imaging exams. Explorations of the cystic and common bile ducts were performed and no signs of neoplastic invasion were found. Lymph node enlargement was observed in the hepatic hilum; however, there were no signs of peritoneal spread or other organ metastasis. A cholecystectomy associated with resection of segments IV-B and V of the liver and lymphadenectomy of the hepatic hilum were performed. The postoperative course was uneventful and the patient was discharged on the 6th postoperative day.
The excised gallbladder and liver segments together measured 13.0×10.0×9.0 cm. The gallbladder presented a smooth and brownish external surface and greenish rough mucosa. A vegetating lesion measuring 2.0×1.0 cm was detected. Furthermore, there was a bloody area on the gallbladder surface, measuring 19.0×7.0 cm, firmly adhered to the liver.
The cuts revealed a brownish lesion, sometimes firm-elastic, sometimes friable, involving the gallbladder wall and liver parenchyma, measuring 9.0×4.0 cm. Microscopical examination showed an intestinal morphology pattern along with extensive solid areas with poorly differentiated histological grade (G3). Two of the 3 lymph nodes removed presented neoplastic invasion. There was also a perforation in the visceral peritoneal, with neoplastic infiltration in the adjacent liver segment and intratumoral coagulative necrosis. The surgical margin had no evidence of neoplastic involvement. Therefore, the pathological staging was TNM: pT3 pN1M0/IIIB.
Histopathology evidenced 2 morphological components, one with neuroendocrine differentiation and the other with poorly differentiated adenocarcinomatous component (Figure 3A). The immunohistochemical (IHC) study of the tumor cells was positive for the following markers: CK7 (SP52), CK20 (SP33) in foci, Synaptophysin (SynA), and chromogranin A (CgA), CDX2 (AMT 28), and the proliferation index Ki-67 was 90% (Figure 3B). Therefore, a mixed adenoneuroendocrine carcinoma of the gallbladder (gMANEC) was confirmed.
POSTOPERATIVE COURSE:
The patient has been followed up by clinical oncology with the initial proposal of adjuvant treatment with 10 chemo-therapy sessions with 5-Fluorouracil (5-FU) and radiotherapy. Initially, the oncology team opted not to prescribe somatostatin analogs due to the absence of symptoms related to neuroendocrine neoplasm. However, 6 months after surgery, the patient started reporting persistent low back pain of moderate intensity. A CT was then performed, revealing retroperitoneal metastasis, evidenced by multiple lymph nodes enlargement in paraaortic, retrocaval, and precaval locations at the level of the pancreas head (Figure 4A, 4B). Therefore, radio-therapy was interrupted and somatostatin analogs were started. Currently, 3 months later, the patient has stable disease, with mild back pain.
Discussion
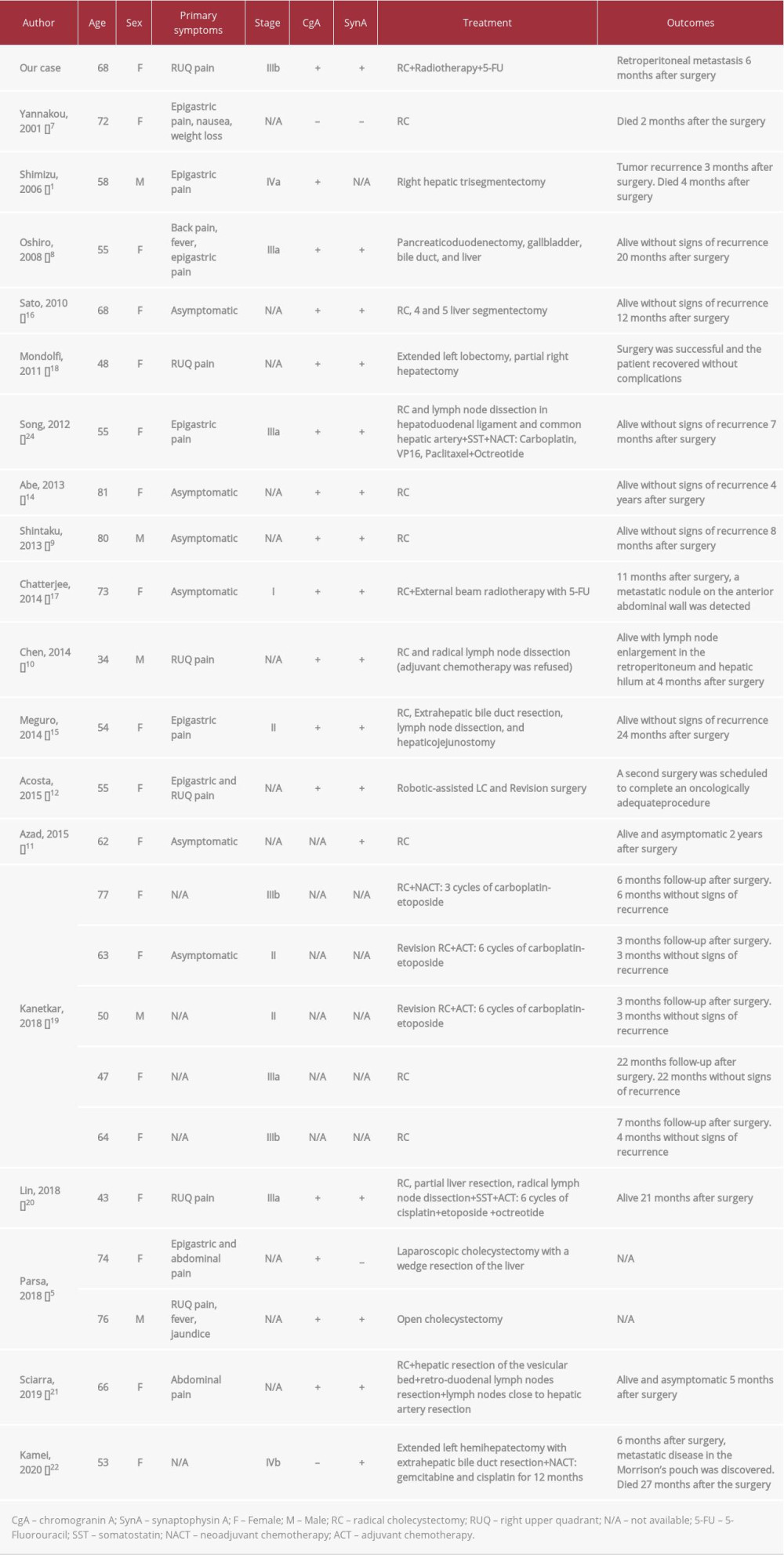
The majority of the literature on gMANEC consists of case reports and small case series, all from Asia (67%), North America, and Europe. Moreover, to date, only 23 cases had been described, and none were from Latin America (Table 1). To date, including data from the present study, the mean age of patients with gMANEC is 61 years and the sex ratio is 5: 19 (male: female).
As mentioned above, the clinical features of gMANEC are non-specific [4]. A recent systematic review, published in 2019, showed that the majority of them (60%) presented epigastric or right upper quadrant (RUQ) pain, whereas 40% were asymptomatic at diagnosis [4]. Furthermore, MANECs can be classified as functional or non-functional depending on the hormone synthesis and endocrine syndrome inductions, although only 3.3% presented syndromes involving hormone production [1] and most of the patients do not present carcinoid syndrome [24].
Besides these nonspecific clinical features, imaging examinations also show low accuracy in this diagnostic process, which highlights the importance of the postoperative histopathological examination in confirming the diagnosis [19]. Moreover, frequently, only a single histological component of the tumor is identified, which impairs the diagnosis and treatment processes [6]. Therefore, chromogranin A (CgA) and synaptophysin (SynA) markers are of paramount importance, given their high sensitivity for MANECs diagnosis, being present in up to 92.3% of patients [4].
The World Health Organization (WHO) Grading for Neuroendocrine Tumor establishes 3 degrees of tumor differentiation: G1 (mitotic count <2 per 10 HPF and/or a Ki-67 index ≤2%); G2 (mitotic count 2–20 per 10 HPF and/or Ki-67 index of 3–20%); and G3 (mitotic count >20 per 10 HPF and/or Ki-67 index >20%) [3]. NETs classified as G1 or G2 are associated with a better prognosis than the NETs of other histological subtypes [25]. In our case, the neuroendocrine portion presented a Ki-67 index of 90%, which would make it a neuroendocrine neoplasm of third grade or low differentiation according to the WHO classification [6,25,26]. Moreover, the adenocarcinoma portion was also classified as Grade 3; therefore, a poor prognosis is expected.
The ideal therapy is still undetermined, although the combination of complete surgical resection, adjuvant chemotherapy, and the use of somatostatin seems to be the mainstay of treatment [4]. In the majority of cases reported in the literature, the decision was made to perform a radical cholecystectomy, with segmentectomy (segments IV and V) of the liver along with lymphadenectomy. Generally, it is expected that 74% of patients undergoing cholecystectomy due to a gallbladder neuroendocrine tumor will present recurrence or metastasis; however, a cholecystectomy combined with hepatic segmentectomy and lymphadenectomy increases the 5-year survival rate from 21.3% to 60.4% in patients with this type of tumor [24].
Evidence suggests that neoadjuvant chemotherapy has good results, but it remains restricted to the few cases of MANEC diagnosed preoperatively [4,14]. However, due to the tumor’s rarity and histological heterogeneity, there are no established adjuvant chemotherapy protocols for gMANEC, which makes it difficult to determine its effectiveness [4,27,28]. Therefore, the adjuvant therapy usually targets the most aggressive component of the tumor [5,6,29].
The prognosis depends on the size of the tumor, infiltration, differentiation, and metastasis [24,30]. The unpredictable biological behavior of MANECs contributes to an uncertain prognosis that seems to be poor [5]. There is evidence that the neuroendocrine component of the tumor may present a higher proliferative rate than the adenocarcinomatous, suggesting that this component may influence the long-term results in these patients [1]. However, there are too few cases of MANEC to form definitive conclusions regarding how it acts.
The main limitation of this report is the short follow-up of the patient, which prevented determination of the long-term prognosis. However, even with this short-term follow-up, the patient had already presented recurrence, with metastatic disease to the retroperitoneum, revealing a poor prognosis.
Conclusions
Our case is the first Western study to report metastasis in the first 6 months after surgical resection. In addition, the unpredictable biological behavior of gMANEC was also observed, since even after successful surgery and adjuvant therapy, early recurrence of disease was detected. Thus, it demonstrates the importance of careful follow-up by the medical team and the need to standardize more effective adjuvant therapy protocols and follow-up guidelines. Current recommendations do not assist in medical team decision-making, since early disease recurrence is also reported in the post-surgical period of low-stage gMANEC. More studies are needed regarding this neoplasm, especially ones focused on improving the patient’s prognosis.
Primary gMANEC is still a challenging pathology with a low number of cases reported in the literature. Our case presents an early recurrence of disease, demonstrating the importance of careful follow-up by the medical team. More studies are needed regarding this neoplasm, especially research focused on improving the patient’s prognosis.
Figures
References:
1.. Shimizu T, Tajiri T, Akimaru K, Combined neuroendocrine cell carcinoma and adenocarcinoma of the gallbladder: Report of a case: J Nippon Med Sch, 2006; 73(2); 101-5
2.. Klimstra DS, Klöppel G, La Rosa S, Rindi G, Classification of neuroendocrine neoplasms of the digestive system: WHO Classification of Tumors Editorial Board Digestive System Tumors, WHO Classification of Tumors, 2019; 16-19, Lyon, International Agency for Research on Cancer Press
3.. Zheng Z, Chen C, Li B, Biliary neuroendocrine neoplasms: Clinical profiles, management, and analysis of prognostic factors: Front Oncol, 2019; 9; 38
4.. Machairas N, Paspala A, Frountzas M, Mixed adenoneuroendocrine carcinoma (MANEC) of the gallbladder: A systematic review of outcomes following surgical management: In Vivo, 2019; 33(6); 1721-26
5.. Parsa C, Orlando R, Narayanan K, Primary mixed adenoneuroendocrine carcinoma (MANEC) of gallbladder, Report of two cases with different histologic and immunohistochemical features: J Tumour Res Reports, 2018; 3(2); 1-5
6.. La Rosa S, Marando A, Sessa F, Capella C, Mixed adenoneuroendocrine carcinomas (MANECs) of the gastrointestinal tract: An update: Cancers (Basel), 2012; 4(1); 11-30
7.. Yannakou N, Rizos S, Parissi-Mathiou P, Mixed (composite) glandular-endocrine cell carcinoma of the gallbladder: HPB (Oxford), 2001; 3(1); 7-9
8.. Oshiro H, Matsuo K, Mawatari H, Mucin-producing gallbladder adenocarcinoma with focal small cell and large cell neuroendocrine differentiation associated with pancreaticobiliary maljunction: Pathol Int, 2008; 58(12); 780-86
9.. Shintaku M, Kataoka K, Kawabata K, Mixed adenoneuroendocrine carcinoma of the gallbladder with squamous cell carcinomatous and osteosarcomatous differentiation: Report of a case: Pathol Int, 2013; 63(2); 113-19
10.. Chen H, Shen YY, Ni XZ, Two cases of neuroendocrine carcinoma of the gall-bladder: World J Gastroenterol, 2014; 20(33); 11916-20
11.. Azad S, Shukla D, Garg A, Mixed adenoneuroendocrine carcinoma of the gallbladder, histopathological features: Indian J Pathol Microbiol, 2015; 58(4); 543-45
12.. Acosta AM, Hamedani FS, Kajdacsy-Balla A, Wiley EL, Primary mixed adenoneuroendocrine carcinoma of the gallbladder in a 55-year-old female patient: A case report and review of the literature: Int J Surg Pathol, 2015; 23(5); 414-18
13.. Harada K, Sato Y, Ikeda H, Clinicopathologic study of mixed adenoneuroendocrine carcinomas of hepatobiliary organs: Virchows Arch, 2012; 460(3); 281-89
14.. Abe T, Kajiyama K, Harimoto N, Composite adeno-endocrine carcinoma of the gallbladder with long-term survival: Int J Surg Case Rep, 2013; 4(5); 504-7
15.. Meguro Y, Fukushima N, Koizumi M, A case of mixed adenoneuroendocrine carcinoma of the gallbladder arising from an intracystic papillary neoplasm associated with pancreaticobiliary maljunction: Pathol Int, 2014; 64(9); 465-71
16.. Sato K, Imai T, Shirota Y, Combined large cell neuroendocrine carcinoma and adenocarcinoma of the gallbladder: Pathol Res Pract, 2010; 206(6); 397-400
17.. Chatterjee D, Wang H, Mixed adenoneuroendocrine carcinoma arising in a papillary adenoma of gallbladder: American Journal of Cancer Case Reports, 2014; 2(1); 37-42
18.. Paniz Mondolfi AE, Slova D, Fan W, Mixed adenoneuroendocrine carcinoma (MANEC) of the gallbladder: a possible stem cell tumor?: Pathol Int, 2011; 61(10); 608-14
19.. Kanetkar AV, Patkar S, Khobragade KH, Neuroendocrine carcinoma of gallbladder: A step beyond palliative therapy, experience of 25 cases: J Gastrointest Cancer, 2019; 50(2); 298-303
20.. Lin YX, Jia QB, Fu YY, Cheng NS, Mixed adenoneuroendocrine carcinoma of the gallbladder: J Gastrointest Surg, 2018; 22(8); 1452-54
21.. Sciarra A, Missiaglia E, Trimech M, Gallbladder mixed neuroendocrinenon-neuroendocrine neoplasm (MiNEN) arising in intracholecystic papillary neoplasm: Clinicopathologic and molecular analysis of a case and review of the literature: Endocr Pathol, 2020; 31(1); 84-93
22.. Kamei K, Shindoh J, Kiya Y, Conversion surgery after extensive chemotherapy for stage IV mixed adenoneuroendocrine carcinoma (MANEC) of the gallbladder: Clinical implications from the patterns of response and recurrence: Clin J Gastroenterol, 2020; 13(2); 240-46
23.. Agha RA, Borrelli MR, Farwana R, The SCARE 2018 statement: Updating consensus Surgical CAse REport (SCARE) guidelines: Int J Surg, 2018; 60; 132-36
24.. Song W, Chen W, Zhang S, Successful treatment of gallbladder mixed adenoneuroendocrine carcinoma with neo-adjuvant chemotherapy: Diagn Pathol, 2012; 7; 163
25.. Kim J, Lee WJ, Lee SH, Clinical features of 20 patients with curatively resected biliary neuroendocrine tumours: Dig Liver Dis, 2011; 43(12); 965-70
26.. Owen DH, Alexander AJ, Konda B, Combination therapy with capecitabine and temozolomide in patients with low and high grade neuroendocrine tumors, with an exploratory analysis of O6-methylguanine DNA methyltransferase as a biomarker for response: Oncotarget, 2017; 8(61); 104046-56
27.. Watanabe J, Suwa Y, Ota M, Clinicopathological and prognostic evaluations of mixed adenoneuroendocrine carcinoma of the colon and rectum: A case-matched study: Dis Colon Rectum, 2016; 59(12); 1160-67
28.. Apostolidis L, Bergmann F, Jaeger D, Winkler EC, Treatment outcomes of patients with mixed adenoneuroendocrine carcinoma (MANEC): J Clin Oncol, 2015; 33(15); e15187
29.. Hervieu V, Scoazec JY, [Mixed endocrine tumors]: Ann Pathol, 2005; 25(6); 511-28 [in French]
30.. Soga J, Primary endocrinomas (carcinoids and variant neoplasms) of the gallbladder. A statistical evaluation of 138 reported cases: J Exp Clin Cancer Res, 2003; 22(1); 5-15
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133