27 May 2021: Articles 
Toxocariasis-Associated Acute Perimyocarditis with Cardiogenic Shock: A Case Report
Challenging differential diagnosis, Rare disease
Se-Jin Park1E, Chae-Won Jang2B, Yong-Kyun Kim2B, Young-Hoon Seo2BD, Ki-Hong Kim1AE, Taek-Geun Kwon2BD, Jang-Ho Bae2AE*DOI: 10.12659/AJCR.930573
Am J Case Rep 2021; 22:e930573
Abstract
BACKGROUND: Toxocariasis is an infection due to ingestion of the helminth parasite larvae found in dogs (Toxocara canis) or cats (Toxocara cati). Symptoms vary from being asymptomatic to shock, depending on the organ invaded by the parasite. However, cardiac involvement with shock in toxocariasis is very rare.
CASE REPORT: A 21-year-old woman without any history of underlying conditions visited the Emergency Department because of epigastric pain, vomiting, headache, and dizziness. Her blood pressure was 80/60 mmHg. Computed tomography (CT) of the brain showed no abnormal lesions. The abdominal-pelvic CT with contrast showed right pleural effusion, pericardial effusion, and focal ascites in the pelvic cavity. Laboratory tests revealed an elevation of eosinophils (40%) and cardiac enzymes (creatinine kinase-MB 27.6 ng/mL, high-sensitive cardiac troponin T 1.21 ng/mL). The transthoracic echocardiogram showed left ventricular systolic dysfunction (ejection fraction 44%) and moderate pericardial effusion. She was presumptively diagnosed with hypereosinophilic perimyocarditis and admitted to the Intensive Care Unit for shock. The pericardial effusion increased during treatment; therefore, pericardiocentesis was performed. Analysis of the pericardial effusion showed eosinophilia (eosinophils 90%) and the serologic test for parasites was positive for Toxocara and Sparganum. A combination therapy of albendazole, praziquantel, and corticosteroid resolved the pericardial effusion and the peripheral blood eosinophil count normalized. She was discharged without any other complications. At Outpatient Clinic follow-ups and observations over the next 2 years there were no abnormal findings, including pericardial effusion or eosinophilia.
CONCLUSIONS: Toxocariasis rarely causes perimyocarditis with cardiogenic shock. Patients who present with pericardial effusion and eosinophilia need to be evaluated for parasitic infection.
Keywords: myocarditis, Pericarditis, Shock, Cardiogenic, Toxocariasis, albendazole, Cats, Dogs, Eosinophilia, Eosinophils
Background
Toxocariasis is a human illness caused by the larvae of
Classic VLM is a systemic form of toxocariasis, which invades the liver and less frequently, the other organs (lungs, kidneys, skin, brain, and muscle) are involved [7,8]; cardiac involvement is rare [9]. Here, we report a case of toxocariasis-associated perimyocarditis with shock in a 21-year-old woman.
Case Report
A 21-year-old woman visited the Emergency Department with epigastric pain, nausea, vomiting, headache, and dizziness for the past 3 days. She had taken medications including nonsteroidal anti-inflammatory drugs for an upper respiratory infection at an Outpatient Clinic. There were no underlying medical conditions or remarkable family history, including any history of ingestion of raw cow meat and liver. She had no pets (no dog or cat). She lived in an urban area and had not made any recent overseas trips. She was single and did not have a baby. She denied any history of playing in the sand box as a child.
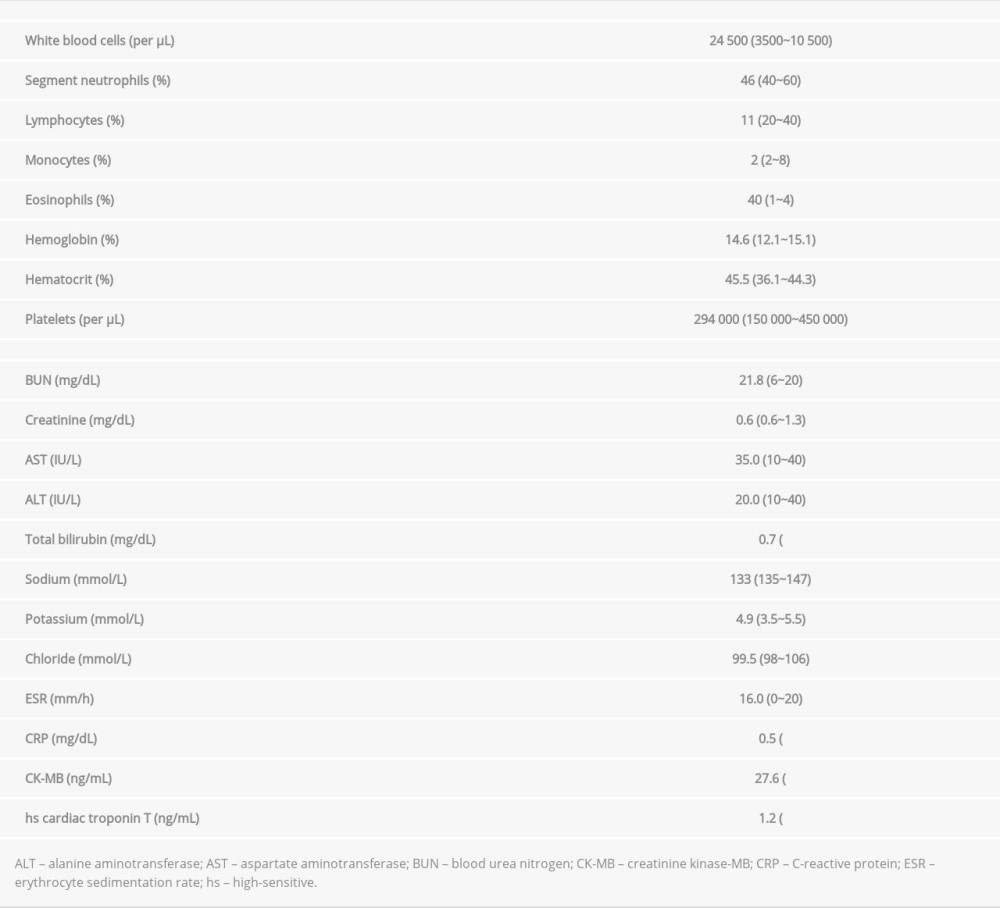
On examination, she appeared to be acutely ill. The examination showed blood pressure of 80/60 mmHg, pulse rate of 100/min, respiratory rate was 20/min, and body temperature was 36.6°C. Heart auscultation revealed normal S1 and S2 without murmurs, rubs, or gallops. An electrocardiogram on arrival showed normal sinus rhythm with ST-segment elevation in the anterior precordial leads. Laboratory tests revealed a white blood cell count of 24 500/uL with eosinophilia (neutrophils 46%, lymphocytes 11%, monocytes 2%, and eosinophils 40%). The erythrocyte sedimentation rate was 16 mm/h (normal, <20 mm/h) and C-reactive protein was 0.5 mg/dL (normal, <0.5 mg/dL). The creatine kinase-MB (CK-MB) was 27.6 ng/mL (normal, <6.3 ng/mL), and high-sensitive cardiac troponin T was 1.21 ng/mL (normal, <0.014 ng/mL) (Table 1). A computed tomography (CT) of the brain showed no abnormal lesions. The abdominal-pelvic CT with contrast showed right pleural effusion, pericardial effusion, and focal ascites in the pelvic cavity (Figure 1A, 1B); however, there was no hepatomegaly. The transthoracic echocardiogram revealed left ventricular (LV) systolic dysfunction (ejection fraction 44%), with moderate pericardial effusion (fluid thickness, 0.9~1.3 cm). She was presumptively diagnosed with hypereosinophilic perimyocarditis and admitted to the Intensive Care Unit (ICU).
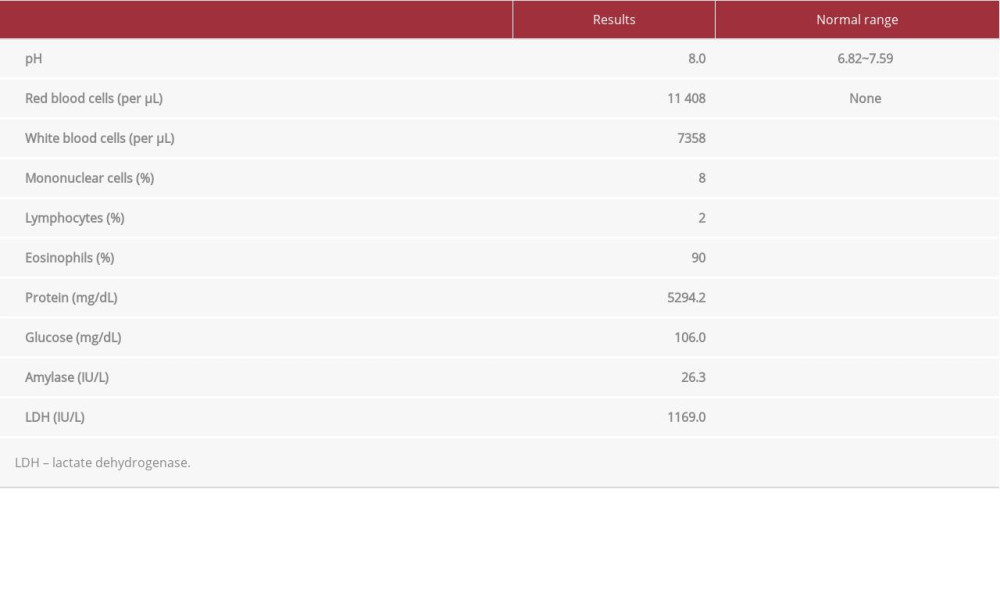
After admission to the ICU, corticosteroid (methylprednisolone 60 mg/day), ceftriaxone (2.0 g/day), and catecholamine (dopamine 20 mcg/kg/minute and dobutamine 10 mcg/kg/minute) were administered intravenously. Another transthoracic echocardiogram was performed 3 days later as a follow-up. Her LV systolic dysfunction had recovered (ejection fraction 73%); however, the pericardial effusion had increased (Figure 2A–2C). She underwent pericardiocentesis with pericardial fluid analysis. The pericardial effusion was exudative and differential counts of white blood cells revealed eosinophilia (eosinophils 90%, monocytes 8%, and lymphocytes 2%) (Table 2).
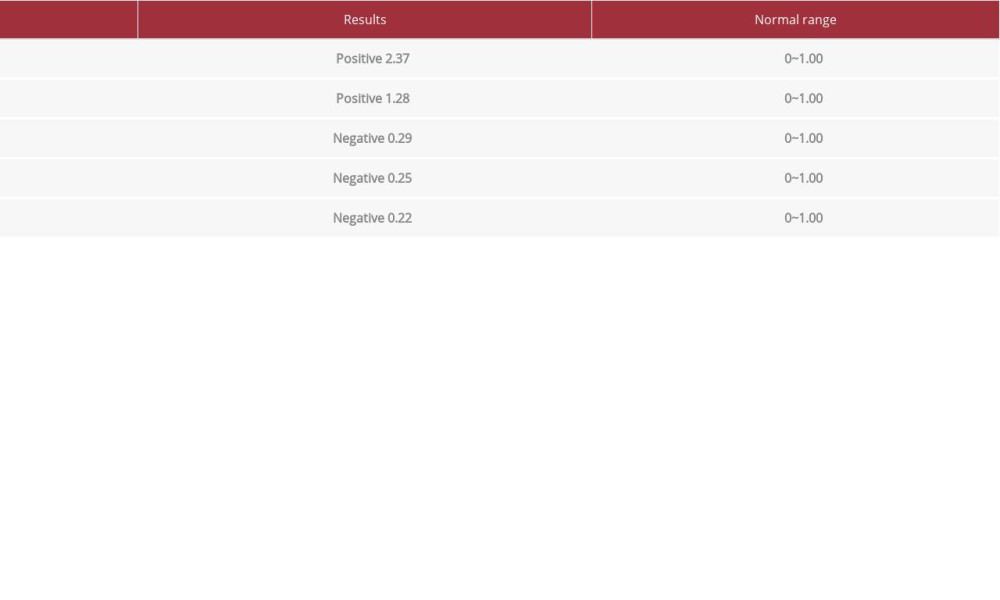
Serologic tests for parasites were performed (Table 3). The enzyme-linked immunosorbent assay to detect immuoglobulin G antibodies against the
This case occurred several years ago and the authors have not obtained informed consent from the patient. However, the patient has been sufficiently anonymized and could not be identified from the information in this case report.
Discussion
It is rare for toxocariasis to invade the heart, and it is especially unusual for it to manifest as shock. Extracorporeal membrane oxygenation treatment has been required to treat toxocariasis with cardiogenic shock [10]. In the present case, the parasitic infection was identified in a perimyocarditis patient with shock accompanied by esosinophilia, which improved with medication.
In most cases,
A confirmed diagnosis of toxocariasis is a tissue biopsy to prove the existence of the
The patient in the present case report was Korean, and the prevalence of toxocariasis accompanied by eosinophilia in Koreans is 20% to 40% [5,13]. Unlike other countries, there is a high prevalence of toxocariasis infection in Korean adults due to the consumption of raw meat and liver [4,5]. Therefore, it is possible that the patient in this case was infected by
In this case, the
Perimyocarditis is defined as myocarditis with some degree of concurrent pericardial inflammation (ie, pericarditis). Acute pericarditis and myocarditis often occur together, although they are not typically equivalent. Myopericarditis and perimyocarditis are similar in that they are accompanied by pericardial effusion and cardiac enzyme elevation (ie, troponin I or T, and CK-MB fraction). However, the regional wall abnormality and LV systolic dysfunction are only accompanied by perimyocarditis [15]. In the present case, the patient showed LV systolic dysfunction with a reduced ejection fraction, suggesting a myocarditis-dominant syndrome (ie, perimyocarditis).
Establishing the underlying cause of perimyocarditis/myopericarditis is of the utmost importance as it can alter the treatment and outcome. Perimyocarditis and myopericarditis share common infectious or noninfectious etiologic agents (including systemic inflammatory diseases, drug- or vaccine-related, rarely iatrogenic) [16]. Among infectious pathogens, viral infections, including the
The present case report has several limitations. First, we did not conduct a western blot procedure for the immunodiagnosis of toxocariasis. Therefore, only the possibility of a cross reaction between toxocariasis and sparganosis was known. The level of brain natriuretic peptide was not checked and virus tests had not been conducted before steroid therapy because we focused on the eosinophilia and did not consider that she could have a viral disease. We should have considered disseminated strongyloidiasis in the differential diagnosis because strongyloidiasis could be a cause of pericardial effusion with eosinophilia, which could be resolved with albendazole [18].
Conclusions
We report a case of perimyocarditis caused by
Figures
References:
1.. Chen J, Liu Q, Liu GH, Toxocariasis: A silent threat with a progressive public health impact: Infect Dis Poverty, 2018; 7(1); 59
2.. Ma G, Holland CV, Wang T, Human toxocariasis: Lancet Infect Dis, 2018; 18(1); e14-24
3.. Overgaauw PA, van Knapen F, Veterinary and public health aspects of toxocara spp: Vet Parasitol, 2013; 193(4); 398-403
4.. Choi D, Lim JH, Choi DC, Toxocariasis and ingestion of raw cow liver in patients with eosinophilia: Korean J Parasitol, 2008; 46(3); 139-43
5.. Seo M, Yoon SC, A seroepidemiological survey of toxocariasis among eosinophilia patients in Chungcheongnam-do: Korean J Parasitol, 2012; 50(3); 249-51
6.. Pawlowski Z, Toxocariasis in humans: Clinical expression and treatment dilemma: J Helminthol, 2001; 75(4); 299-305
7.. Bass JL, Mehta KA, Glickman LT, Eppes BM, Clinically inapparent toxocara infection in children: N Engl J Med, 1983; 308(12); 723-24
8.. Moreira GM, de Lima Telmo P, Mendonça M, Human toxocariasis: Current advances in diagnostics, treatment, and interventions: Trends Parasitol, 2014; 30(9); 456-64
9.. Altcheh J, Nallar M, Conca M, Biancardi M, Freilij H, Toxocariasis: Clinical and laboratory features in 54 patients: Ann Pediatr (Barc), 2003; 58(5); 425-31
10.. Enko K, Tada T, Ohgo KO, Fulminant eosinophilic myocarditis associated with visceral larva migrans caused by Toxocara canis infection: Circ J, 2009; 73(7); 1344-48
11.. Kuenzli E, Neumayr A, Chaney M, Blum J, Toxocariasis-associated cardiac diseases-A systematic review of the literature: Acta Trop, 2016; 154; 107-20
12.. Zibaei M, Helminth infections and cardiovascular diseases: Toxocara species is contributing to the disease: Curr Cardiol Rev, 2017; 13(1); 56-62
13.. Song HB, Lee D, Jin Y, Prevalence of toxocariasis and its risk factors in patients with eosinophilia in Korea: Korean J Parasitol, 2020; 58(4); 413-19
14.. Magnaval JF, Fabre R, Maurières P, Application of the western blotting procedure for the immunodiagnosis of human toxocariasis: Parasitol Res, 1991; 77(8); 697-702
15.. Imazio M, Gaita F, LeWinter M, Evaluation and treatment of pericarditis: A systematic review: JAMA, 2015; 314(14); 1498-506
16.. Chlabicz M, Jakim P, Zalewska-Adamiec M, A rare case of acute pleuropericarditis as a complication of permanent pacemaker insertion: Am J Case Rep, 2021; 22; e928188
17.. Imazio M, Trinchero R, Myopericarditis: Etiology, management, and prognosis: Int J Cardiol, 2008; 127(1); 17-26
18.. Lai CP, Hsu YH, Wang JH, Lin CM, Strongyloides stercoralis infection with bloody pericardial effusion in a non-immunosuppressed patient: Circ J, 2002; 66(6); 613-14
Figures
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942824
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943118
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942826
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250