18 October 2021: Articles 
Hypercalcemia and CYP24A1 Gene Mutation Diagnosed in the 2 Trimester of a Twin Pregnancy: A Case Report
Challenging differential diagnosis, Unusual setting of medical care, Rare disease, Clinical situation which can not be reproduced for ethical reasons
Andris Romašovs12ABCDEF*, Lauma Jaunozola1DF, Egija Berga-Švītiņa3CF, Zanda Daneberga3CF, Edvīns Miklaševičs3CD, Valdis Pīrāgs12AEFDOI: 10.12659/AJCR.931116
Am J Case Rep 2021; 22:e931116
Abstract
BACKGROUND: Loss-of-function mutations of the CYP24A1 gene cause a deficiency of the CYP24A1 enzyme, which is involved in the catabolism of 1,25-dihydroxyvitamin D3. Patients who are CYP24A1 enzyme deficient are at increased risk of developing hypercalcemia during pregnancy and should avoid additional vitamin D supplementation. This case report provides additional information for managing and diagnosing patients with a CYP24A1 gene mutation.
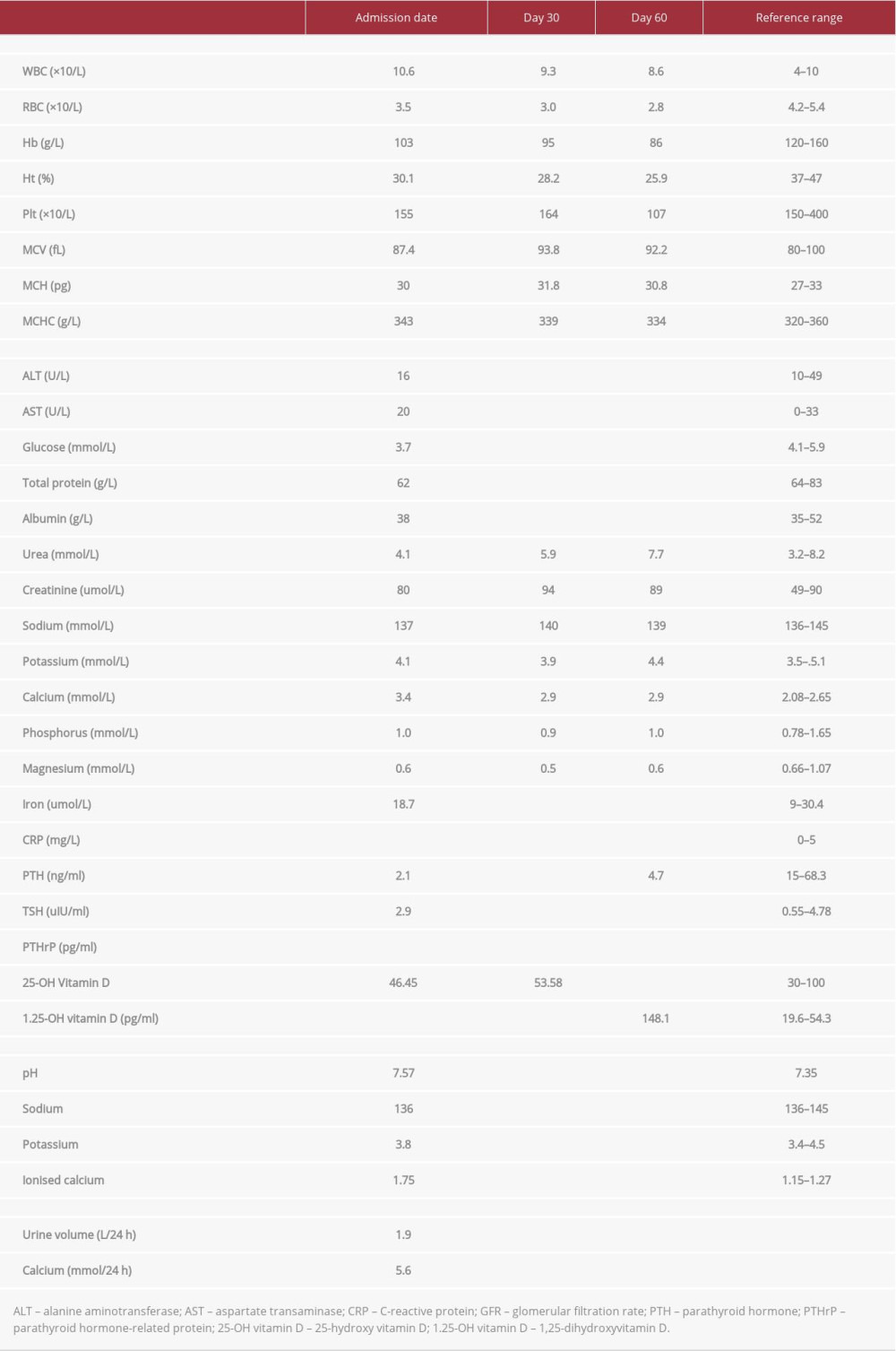
CASE REPORT: A primipara woman with a twin pregnancy was admitted to our hospital for frequent hypertensive crises. She had no history of hypercalcemia-associated signs and symptoms except nephrocalcinosis, and reported no other abnormalities or discomfort at presentation. Laboratory tests revealed that the parathyroid hormone level was suppressed and the serum calcium level was markedly increased. The 25-hydroxyvitamin D level was at the upper limit of the reference range while the 1,25-dihydroxyvitamin D3 level was elevated, suggesting a vitamin D catabolism disorder. A genetic test was performed and a homozygous likely pathogenic variant (based on the American College of Medical Genetics and Genomics guidelines) c.964G>A (p.Glu322Lys) was detected in the CYP24A1 gene (NM_000782.5). A cesarean section delivery was performed due to a single intrauterine demise at 33 weeks of gestation. The preterm newborn was diagnosed with transitional hypercalcemia and hyperphosphatemia; however, he was not treated, as he was asymptomatic.
CONCLUSIONS: Patients with a CYP24A1 gene mutation are at increased risk of hypercalcemia and fetal demise; therefore, 25-hydroxyvitamin D and calcium levels should be monitored in routine blood tests during pregnancy. Hypercalcemia in a newborn should be carefully evaluated and treated, as hypercalciuria can lead to nephrocalcinosis.
Keywords: CYP24A1 Protein, Human, Hypercalcemia, Idiopathic, of Infancy, Vitamin D3 24-Hydroxylase, Cesarean Section, Female, Humans, Hypercalcemia, Infant, Newborn, Male, Mutation, Pregnancy, Pregnancy, Twin
Background
Hypercalcemia is a rare disorder during pregnancy and its early detection is crucial for lowering maternal and fetal morbidity and mortality rates [1]. The symptoms of hypercalcemia overlap with common discomforts during pregnancy, including nausea, constipation, anxiety, hypertension, and preeclampsia, presenting a serious challenge to the clinician and often resulting in a delayed diagnosis. This clinical case gives additional information for clinicians managing and diagnosing patients with a CYP24A1 gene mutation.
In the 1950s, relatively high doses of vitamin D supplementation (up to 4000 IU/day) were prescribed for infants. Within 2 years of receiving infant formula and fortified milk supplemented with vitamin D, nearly 200 infants in the UK had severe hypercalcemia and related symptoms (including anorexia, polyuria, dehydration, constipation, abdominal pain, and lethargy) [2,3]. Their blood test results showed suppressed parathyroid hormone (PTH) levels and hypercalcemia. At that time, it was mainly attributed to vitamin D supplementation and defined as idiopathic infantile hypercalcemia (IIH). In 2011, Schlingmann et al identified a mutation of the CYP24A1 gene that is inherited in an autosomal recessive pattern and causes hypercalcemia in patients with IIH [4]. Since 2011, >20 different mutations in the CYP24A1 gene have been identified [5]. Hypercalcemia due to the CYP24A1 gene mutation not only affects infants and children but can also be asymptomatic and manifest during pregnancy and late adulthood.
We believe IIH is an obsolete term and genetic testing should be used to determine the cause of hypercalcemia. We present a case of gestational hypercalcemia caused by mutation of the CYP24A1 gene.
Case Report
INVESTIGATIONS:
We obtained measurements of blood gases and determined the calciumion levels in a 24-h urine sample. Calcifediol was at the upper end of the reference range, which raised suspicion of hypercalcemia caused by vitamin D, as vitamin D hypovitaminosis is observed at our geographical latitude in Eastern Europe [6]. The calciumion level was above the reference range and the 24-h urine sample revealed no increase in calcium secretion through the kidneys. At this point, it was obvious that this case of hypercalcemia was PTH-independent and it was important to exclude humoral hypercalcemia of malignancy and possible parathyroid hormone-related protein (PTHrP) overproduction by the placenta or breast tissue. We performed magnetic resonance imaging of the thorax, and ultrasonography of the thyroid and parathyroid glands, breasts, and abdomen. None of these radiological examinations revealed any pathology. She remained under close observation by the Gynecologist and Obstetrics Specialist and the ultrasonography was repeated every 2 weeks to 3 weeks due to the twin-growth discordance. The levels of 1,25-dihydroxyvitamin D3 (calcitriol) and PTHrP were measured. PTHrP was within the normal range, while calcitriol was significantly elevated. The CYP24A1 single-gene test was performed and a homozygous, likely pathogenic variant (based on the American College of Medical Genetics and Genomics guidelines) c.964G>A (p.Glu322Lys) was detected in the CYP24A1 gene (NM_000782.5) [7]. Due to the number of homozygous benign variants detected in the coding and noncoding parts of the gene, the possibility of partial or whole gene deletion in 1 allele was proposed; however, no further investigations have been performed to date.
DIFFERENTIAL DIAGNOSIS:
In the modern world, excessive consumption of over-the-counter food supplements and vitamin D poses a risk of hypervitaminosis D, which should be a diagnosis of exclusion in patients with hypercalcemia. Although rarely observed, hyper-vitaminosis A can also cause hypercalcemia. During the laboratory work-up for hypercalcemia, we used the clinical pathway proposed by Goltzman (Figure 1) and found it to be very useful, as it provided a logical sequence for the follow-up and summarized all the information provided in a journal article by the same author [8]. By performing radiological examinations, we excluded tuberculosis, granulomatous diseases, and malignancies. During the pregnancy, she had anemia and by performing serum protein electrophoresis, we excluded multiple myeloma from the differential diagnosis. After these tests, genetic pathologies (including familial hypocalciuria, hypercalcemia, and CYP24A1 gene mutation) were considered to be the most probable causes of the hypercalcemia.
TREATMENT:
Antihypertensive therapy with 250 mg methyldopa 3 times daily was initiated and it brought her blood pressure to within the normal range. Hypercalcemia was partly managed using furosemide (10 mg twice daily) to control diuresis and hydration. In alternate weeks, blood was analyzed to determine her electrolyte levels. There was a significant decrease in the calcium levels; however, they were never within the reference range. Throughout the observation period (3rd trimester), hypomagnesemia was recorded, and supplementation with magnesium was ineffective. She refused treatment with glucocorticoids.
OUTCOME:
During a regular prenatal visit, an ultrasound investigation revealed no heartbeat in 1 of the 2 fetuses and she was admitted to the Emergency Department. A cesarean section was performed due to a single intrauterine demise at 33 weeks of gestation. The second fetus (male) was delivered with an Apgar score of 3/5/6 and admitted for further care to the Children’s Clinical University Hospital. He had significant developmental disorders, including anorectal malformation. He was hyper-calcemic and diagnosed with transitional hypercalcemia and hyperphosphatemia. At 8 months, he remained hypercalcemic; however, he was not treated, as he was asymptomatic.
After giving birth, the mother refused to be treated for hypercalcemia and was followed up by the family physician. Nevertheless, we maintained contact with the patient to provide medical care and treatment for hypercalcemia in case she decided to accept treatment. We are aware that she is still mildly hypercalcemic, and 3 months after the delivery, she was diagnosed (by ultrasonography) with sand-like particles in both kidneys.
This clinical case report was conducted in accordance with the Declaration of Helsinki, and its protocol was approved by the Ethics Committee of Pauls Stradiņš Clinical University Hospital. The patient gave informed consent for the publication of this case report.
Discussion
Hypercalcemia during pregnancy, due to the loss-of-function mutation of the CYP24A1 gene, is rarely described in the literature. In our patient, hypercalcemia and hypervitaminosis D was due to the biallelic, autosomal recessive gene mutation of CYP24A1 [4]. The CYP24A1 gene encodes vitamin D 24-hydroxylase, a mitochondrial enzyme that breaks down calcitriol and calcifediol via the C24- and C23-oxidation pathways, thus controlling the availability of active vitamin D in the body. The loss-of-function variants of this gene lead to the overproduction of 1,25-dihydroxyvitamin D3 (calcitriol), which results in hypercalcemia, hypercalciuria, hypertension, anxiety, headache, and nephrosclerosis [9].
Griffin et al reported that CYP24A1 gene mutations are inherited and can be benign or likely benign [10]. The CYP24A1 gene mutation is inherited in an autosomal recessive pattern, as reported by Schlingmann et al [4]. The proband in the case report by Griffin et al developed hypercalcemia during the 2nd trimester of her pregnancy and was treated by administration of intravenous fluids and glucocorticoids to maintain normocalcemia. Similar to our case, their patient developed hyper-tension with subsequent preeclampsia and the pregnancy was resolved by an emergency cesarean section. Previously, the proband had been asymptomatic and normotensive. The proband’s family members were also asymptomatic and only 1 member reported nephrolithiasis.
The placenta controls the transfer of minerals between the mother and the fetus. Receptors for calcium transportation through the placenta are expressed on the trophoblast surface and provide transportation of calcium against the concentration gradient. Fetal calcium concentration is significantly higher compared to the maternal calcium levels. During significant maternal hypovitaminosis D, normocalcemia is maintained for the fetus [11]. High concentrations of PTHrP, calcitonin, and PTH are found in developing fetuses. For example, the PTHrP concentration in cord blood is 15 times to 20 times higher than the maternal PTH levels. The concentration of calcium rises as early as 15 weeks’ gestation to 20 weeks’ gestation. This rise in the calcium level is explained by the calcification of skeletal structures [12,13].
Although calcitonin does not play a role in calcium transportation, the deactivation of calcitonin receptors was found to cause fetal death [12]. Many fetal tissues were identified as producers of PTHrP; however, the placenta and the fetal parathyroid glands are thought to play a major role in the synthesis of PTHrP. There are few studies that describe a PTHrP-dependent hypercalcemic crisis during pregnancy, which can be due to the physiological overproduction of PTHrP by the placenta, breast tissues, or malignant cells [11,14,15]. In animal studies, a parathyroidectomy and low levels of PTH and PTHrP caused hypocalcemia in the fetus, consequently leading to a wide range of complications, including neonatal tetany, stillbirth, and skeletal disorders [12,15].
Our knowledge about the treatment, clinical presentation, and diagnostics of hypercalcemia caused by the CYP24A1 gene mutation is mainly based on case reports. Clinical presentations can vary; for example, Woods et al reported a case of gestational hypercalcemia, which presented with acute pancreatitis and hypertension [16]. Acute pancreatitis can occur due to hypercalcemia and not directly due to the CYP24A1 gene mutation. Although hypercalcemia is a rare cause of acute pancreatitis, clinicians should always take steps to rule it out from the differential diagnosis [17].
In our opinion, nonpharmacological and pharmacological treatment of hypercalcemia should be initiated simultaneously. Nonpharmacological treatment should be initiated by the cessation of over-the-counter food supplements, including vitamin D and multivitamins, and lowering the intake of calcium-rich foods. There are restrictions in the pharmacological treatment options for hypercalcemia during pregnancy, as many treatment options pose risks to the fetus and mother. Biphosphonate use during pregnancy poses a risk of fetal hypocalcemia. For nonpregnant individuals, treatment options include bisphosphonates, calcitonin, loop diuretics, and glucocorticoids. Only some of these treatment options are safe during pregnancy. Treatment with bisphosphonates is not advised during pregnancy, as there are uncertainties about the possibility of teratogenic effects, since some studies have demonstrated that congenital malformations can occur. Bisphosphonates are small molecules that cross the placenta and can cause fetal hypocalcemia and related complications [14,18,19]. In our experience, treatment with loop diuretics should be initiated as soon as possible. As torasemide is potassium- and calcium-sparing, it is less effective than furosemide in the treatment of hypercalcemia [20]. Glucocorticoids are an option for the treatment of hypercalcemia, as they directly act on the osteocytes and osteoblasts, promote apoptosis [21], reduce osteo-blast proliferation, inhibit the production of cytokines, and reduce the activity of cytokines in osteoblasts [22,23]. A few studies have investigated the action of corticosteroids on the CYP24A1 gene and the expression of vitamin D 24-hydroxylase. They report that glucocorticoids suppress the degradation of calcitriol by 24-hydroxylase in the bone cells, which could increase the concentration of active vitamin D. This in turn suppresses the action of the corticosteroids [23].
In patients with therapy-resistant hypercalcemia, concomitant hyperparathyroidism should be ruled out by obtaining serum PTH levels. Due to hypercalcemia, nephrosclerosis and chronic kidney disease can develop and cause secondary hyperparathyroidism and further increase calcium levels. Before performing any surgical interventions, additional laboratory investigations are needed, including the determination of phosphate and 1,25-dihydroxy vitamin D levels [24].
Conclusions
Patients with CYP24A1 gene mutation can be oligosymptomatic, although they are at an increased risk of hypercalcemia and fetal demise during pregnancy. Our patient only reported frequent headaches, nausea, and weakness during the pregnancy. To avoid fetal demise as seen in the present case (1 fetal demise in the twin pregnancy), the measurement of Ca2+ and vitamin D metabolites should be included in routine blood tests early in the gestational period.
References:
1.. Koren R, Neeman O, Koren S, Benbassat CA, Humoral hypercalcemia of pregnancy treated with bisphosphonates: Arch Endocrinol Metab, 2018; 62(1); 125-28
2.. Lightwood R, Stapleton T, Idiopathic hypercalcaemia in infants: Lancet, 1953; 265(6779); 255-56
3.. Fanconi G, [Chronic disorders of calcium and phosphate metabolism in children.]: Schweiz Med Wochenschr, 1951; 81(38); 908-13 [in German]
4.. Schlingmann KP, Kaufmann M, Weber S, Mutations in CYP24A1 and idiopathic infantile hypercalcemia: N Engl J Med, 2011; 365(5); 410-21
5.. Jones G, Kottler ML, Schlingmann KP, Genetic diseases of vitamin D metabolizing enzymes: Endocrinol Metab Clin North Am, 2017; 46(4); 1095-117
6.. Lips P, Cashman KD, Lamberg-Allardt C, Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society: Eur J Endocrinol, 2019; 180(4); P23-54
7.. Richards S, Aziz N, Bale S, Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology: Genet Med, 2015; 17(5); 405-23
8.. Goltzman D, Feingold KR, Anawalt B, Approach to hypercalcemia: Endotext, 2019, Copyright by South Dartmouth (MA), MDText.com, Inc https://www.ncbi.nlm.nih.gov/books/NBK279129/
9.. Sadiq NM, Naganathan S, Badireddy M: Hypercalcemia, 2020, StatPearls Publishing [cited 2020/05/29]. www.ncbi.nlm.nih.gov/books/NBK430714/
10.. Griffin TP, Joyce CM, Alkanderi S, Biallelic CYP24A1 variants presenting during pregnancy: clinical and biochemical phenotypes: Endocr Connect, 2020; 9(6); 530-41
11.. Melmed S, Auchus RJ, Goldfine AB: Williams Textbook of Endocrinology, 2020; 854-55, Elsevier Health Sciences Division
12.. Kovacs CS: Genetics of bone biology and skeletal disease, 2018, Elsevier Academic Press
13.. Sato K, Hypercalcemia during pregnancy, puerperium, and lactation: Review and a case report of hypercalcemic crisis after delivery due to excessive production of PTH-related protein (PTHrP) without malignancy (humoral hypercalcemia of pregnancy): Endocr J, 2008; 55(6); 959-66
14.. Machairiotis N, Ntali G, Kouroutou P, Michala L, Clinical evidence of the ef-fect of bisphosphonates on pregnancy and the infant: Horm Mol Biol Clin Investig, 2019; 40(2); j/hmbci.2019.40
15.. Lieben L, Stockmans I, Moermans K, Carmeliet G, Maternal hypervitaminosis D reduces fetal bone mass and mineral acquisition and leads to neonatal lethality: Bone, 2013; 57(1); 123-31
16.. Woods GN, Saitman A, Gao H, A young woman with recurrent gestational hypercalcemia and acute pancreatitis caused by CYP24A1 deficiency: J Bone Miner Res, 2016; 31; 1841-44
17.. Kumar A, Kumar PG, Pujahari AK, Sampath S, Hypercalcemia related pancreatitis: Med J Armed Forces India, 2010; 66(4); 385-86
18.. Sokal A, Elefant E, Leturcq T, Pregnancy and newborn outcomes after exposure to bisphosphonates: A case-control study: Osteoporosis Int, 2019; 30(1); 221-29
19.. Ioannis SP, Chrysoula LG, Aikaterini K, The use of bisphosphonates in women prior to or during pregnancy and lactation: Hormones (Athens, Greece), 2011; 10(4); 280-91
20.. Friedel HA, Buckley MM, Torasemide: A review of its pharmacological properties and therapeutic potential: Drugs, 1991; 41(1); 81-103
21.. O’Brien CA, Jia D, Plotkin LI, Glucocorticoids act directly on osteoblasts and osteocytes to induce their apoptosis and reduce bone formation and strength: Endocrinology, 2004; 145(4); 1835-41
22.. Canalis E, Delany AM, Mechanisms of glucocorticoid action in bone: Ann NY Acad Sci, 2002; 966; 73-81
23.. Zayny A, Almokhtar M, Wikvall K, Effects of glucocorticoids on vitamin D 3-metabolizing 24-hydroxylase (CYP24A1) in Saos-2 cells and primary human osteoblasts: Mol Cell Endocrinol, 2019; 496; 110525
24.. David K, Khalil R, Hannon H, Therapy-resistant hypercalcemia in a patient with inactivating CYP24A1 mutation and recurrent nephrolithiasis: Beware of concomitant hyperparathyroidism: Calcif Tissue Int, 2020; 107(5); 524-28
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133