02 September 2021: Articles 
Megadose Methylprednisolone for Immune Thrombocytopenia in an Infant Positive for SARS-CoV-2: A Case Report
Management of emergency care, Rare coexistence of disease or pathology
Harapan Parlindungan RingoringoDOI: 10.12659/AJCR.931517
Am J Case Rep 2021; 22:e931517
Abstract
BACKGROUND: Immune thrombocytopenia (ITP) is rare in infants under 1 year old. Bleeding often occurs when the platelet count is <20 000/uL. The disease can progress because of accompanying COVID-19 disease.
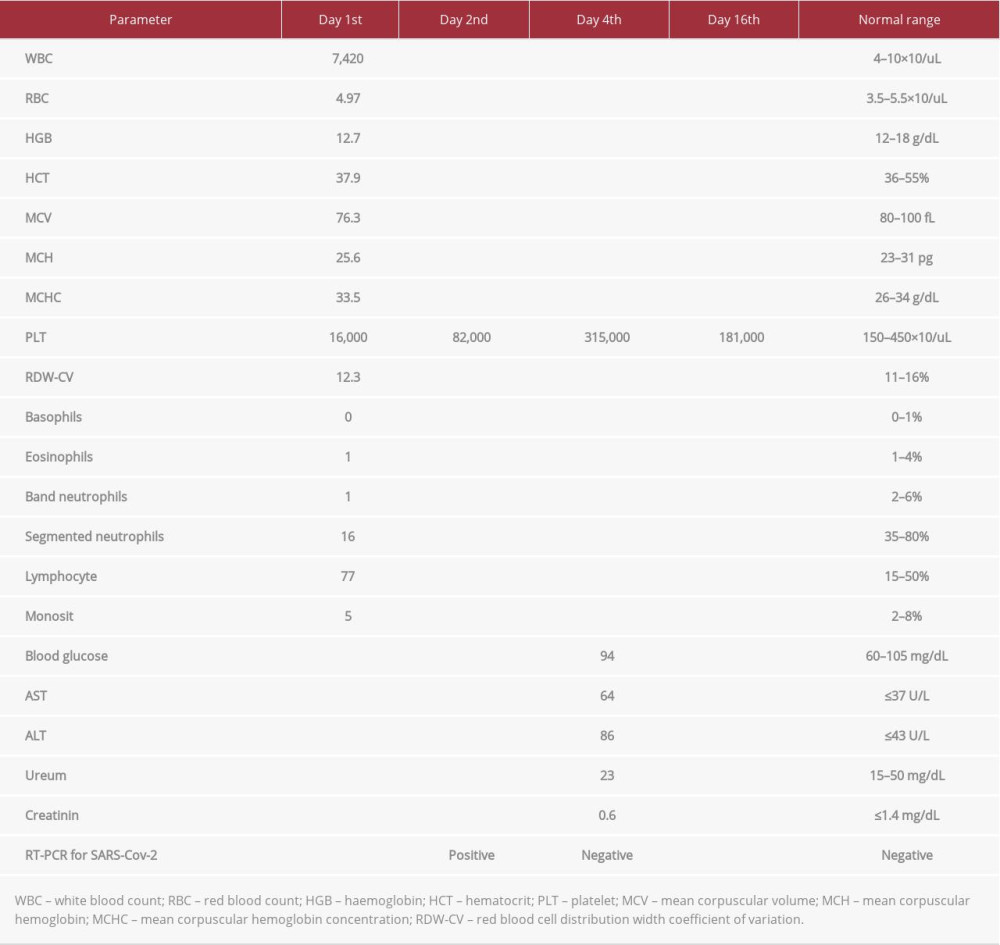
CASE REPORT: A 9-month-old boy, weighing 8.5 kg, came to the hospital with petechiae on the forehead, cheeks, mouth, and extremities. The patient had rhinorrhea for 3 days previously and was febrile, pale, weak, and could not drink. He had the measles-rubella vaccination 19 days prior. Physical examination showed no abnormalities of the eyes, ears, nose, throat, and mouth. Heart and lungs were within normal limits, with no organomegaly, lymphadenopathy, or congenital anomaly of the abdomen. Laboratory examination showed hemoglobin, 12.7 g/dL; leukocytes, 7420/uL; platelet count, 16 000/uL; and hematocrit, 37.9%. Erythrocyte sedimentation rate was 14 mm at 1 h and 21 mm at 2 h. Peripheral blood smear showed normal RBC morphology, normal leukocytes, and few platelets. IgG was reactive and IgM was nonreactive on rapid antibody test. RT-PCR was positive for SARS-COV-2. Chest-X-ray showed pneumonia. The diagnosis was newly diagnosed ITP with COVID-19. Patient was treated with 30 mg/kg body weight/day of IV methylprednisolone for 3 days (250 mg); then 20 mg/kg body weight/day (175 mg) orally for 4 days in 3 divided doses. Azithromycin 100 mg/day, zinc 20 mg/day, and vitamin C 50 mg/day orally were also given.
CONCLUSIONS: COVID-19 screening is highly recommended during this pandemic to identify it as a potential cause of childhood ITP. Megadose methylprednisolone had an excellent response in alleviating ITP with confirmed COVID-19 in an infant.
Keywords: COVID-19, infant health, Methylprednisolone Hemisuccinate, Purpura, Thrombocytopenic, Idiopathic, COVID-19, Humans, Infant, Methylprednisolone, Pandemics, SARS-CoV-2
Background
Immune thrombocytopenia (ITP) is an acquired acute bleeding disease that presents as petechiae and purpura on the skin and/or mucous membranes caused by thrombocytopenia (platelet count <100 000/uL). Approximately two-thirds of children with primary ITP have a history of viral infection during the previous month [1]. Viruses commonly identified as triggers of ITP include cytomegalovirus, hepatitis C, herpes, varicella-zoster, Epstein-Barr, influenza, and HIV [2–5]. ITP is a potential presentation of COVID-19. Tsao et al reported the case of a 10-year-old girl who had ITP and a positive reverse transcriptase-polymerase chain reaction (RT-PCR) test result for severe acute respiratory syndrome coronavirus (SARS-CoV-2) [6]. SARS-CoV-2 viral testing should be considered in these patients to allow for appropriate hospital triaging and isolation to limit community spread and healthcare worker infection during pandemics. We report the case of an infant with ITP that was likely caused by SARS-CoV-2 infection.
Case Report
A 9-month-old boy, weighing 8.5 kg, came to the hospital with petechiae on the forehead, cheeks, mouth, and extremities. He had rhinorrhea for 3 days previously and was febrile, pale, weak, and could not drink. The patient had the measles-rubella vaccination 19 days prior. Physical examination showed a respiration rate of 32 breaths per min and heart rate of 100 beats per min. The examination of the eye, ear, nose, throat, and mouth showed no abnormalities. Examination of the heart, lungs, and upper and lower extremities showed no abnormalities. There was no organomegaly, lymphadenopathy, or congenital anomaly of the abdomen. The laboratory findings are shown in Table 1. The erythrocyte sedimentation rate (ESR) was 14 mm at 1 h and 21 mm at 2 h. A peripheral blood smear showed normochromic and normocytic red blood cells, normal leukocytes, and few platelets. The patient’s blood type was B. Urinalysis results were within the standard range. IgG was reactive and IgM was nonreactive on the rapid antibody test. A chest X-ray showed pneumonia. The diagnosis made was newly diagnosed ITP with suspected COVID-19. The patient was treated with 30 mg/kg body weight/day of intravenous (IV) methylprednisolone for 3 days (250 mg), and then 20 mg/kg body weight/day (175 mg) oral methylprednisolone for 4 days in 3 divided doses. The patient was also given azithromycin 100 mg/day, zinc 20 mg/day, vitamin C 50 mg/day. The patient’s skin petechiae appearance can be seen in Figure 1. The chest X-ray showed mild right upper-lobe opacity, as shown in Figure 2.
On day 2 of treatment, the initial RT-PCR test for SARSCoV-2 was performed, and the result was positive, confirming COVID-19. The results of routine blood tests, including complete blood count and blood chemistry on days 2, 4, and 16, are shown in Table 1. After 2 IV administrations of 250 mg methylprednisolone, the platelet count was 82 000/uL. The patient’s condition improved, and the petechiae were significantly reduced. On day 4 of treatment, the second RT-PCR test for SARS-CoV-2 was negative. Because the patient’s condition was good and the petechiae were gone, the patient was discharged. The patient went home with oral methylprednisolone and did not undergo tapering of methylprednisolone. Nine days after stopping methylprednisolone (day 16), the patient’s condition was stable. There were no signs of corticosteroid side effects such as “moon face”, and the platelet count was 181 000/uL. The neutrophil to lymphocyte ratio was 0.42.
Discussion
According to the course of the disease, ITP in children is divided into 3 types: 1) newly diagnosed, with diagnosis within 3 months, accounting for 50% of cases; 2) persistent ITP, within 3 to 12 months from diagnosis, accounting for 25% of cases; and 3) chronic ITP, more than 12 months from diagnosis [7].
ITP most commonly occurs in children 2 to 7 years of age [8]. The incidence of pediatric ITP is 4.3 per 100 000 children per year, and in children aged 2 to 5 years, the incidence is higher in boys than in girls [9]. This disease is rarely found in infants less than 1 year old, probably because of the immature baby’s immune system. In general, there is always a history of viral infection of the upper respiratory tract or gastrointestinal system or immunizations 2 to 4 weeks before diagnosis [8]. In a study including 257 pediatric patients with ITP, Yong et al reported a history of infection in 20.2% of patients and immunization in 8.6% [9]. In comparison, Hashemi et al reported a history of the common cold in 47% of cases and a history of immunization in 25.8% [10]. In the present case, the patient was febrile and had rhinorrhea for 3 days previously. Also, the rapid antibody test was reactive for IgG and nonre-active for IgM, and the positive RT-PCR test for SARS-COV-2 meant the patient was in the late or recurrent stage of infection. Therefore, ITP was triggered by SARS-COV-2 infection and was not related to the patient’s immunization history. However, ITP could have been associated with the patient’s immunization history and, at the same time, the patient had COVID-19.
Bleeding often occurs with a platelet count <20 000/uL. Purpuric, mucosal, and intracranial hemorrhage was found in 83.3%, 40.95%, and 3% of cases, respectively [10]. A total of 80% of children with ITP who have acute bleeding will recover within days or weeks with or without treatment and will recover within 6 months. The platelet count will return to normal (>150 000/uL) in 50% of cases within 1 to 3 months and in 60% to 75% of cases within 6 months of the onset of thrombocytopenia [2,11]. In the present case, the patient presented with petechial bleeding, was febrile for 3 days previously, was weak, and could not drink. The complete blood count results showed a platelet count of 16 000/uL and the peripheral blood smear showed few platelets. The rapid antibody test result was reactive for IgG and was nonreactive for IgM. A chest X-ray showed pneumonia. This situation was an emergency because platelet counts of only 16 000/uL indicate a high risk of intracranial bleeding. Intracranial bleeding is rare, occurring in only 0.5% to 1.0% of cases when the platelet count is <10 000/uL [12]; however, this risk should be considered. In emergency cases, intravenous immunoglobulin (IVIG) can be given; however, it is expensive (the family in this case could not afford it), and therefore a megadose of methylprednisolone, which is inexpensive, was given instead of the usual dosage. A 30 mg/kg body weight/day dosage of methylprednisolone was given for 3 days and then 20 mg/kg body weight/day was given for 4 days. Albayrak et al reported no significant differences in ITP treatment to achieve normal platelet values with either IVIG 0.5 g/kg body weight/day for 5 days or methylprednisolone 50 mg/kg body weight/day for 7 days or methylprednisolone 30 mg/kg body weight/day for 7 days [13]. Our patient’s condition was improving with the megadose of methylprednisolone. On day 2 of treatment, the platelet count rose to 82 000/uL. On day 4, it was 315 000/uL, and the petechiae disappeared. To discharge the patient on day 4 of treatment, the second RT-PCR test for SARSCoV-2 was performed, and the result was negative. The patient went home in good condition. In the present case, there was no tapering of methylprednisolone, and it was shown that 9 days after the methylprednisolone was stopped, the platelet count was 181 000/uL and there were no signs of bleeding. In 2 weeks, remission was achieved.
The course of COVID-19 in children is generally mild and has better outcomes overall compared with that of adults [14–17]. Fever, cough, and sore throat are the most frequent symptoms [15,17,18]. Research in China of 2143 children with suspected COVID-19 showed that the proportions of severe and critical cases by age group were 10.6% for <1 year, 7.3% for 1 to 5 years, 4.2% for 6 to 10 years, 4.1% for 11 to 15 years, and 3.0% for ≥16 years [19]. The hematological profile of an infant or child with COVID-19 is not specific. Several studies have shown the presence of leukopenia [20], leukocytosis [18], or normal leukocyte counts [18,21,22]. Henry et al reported that leukocytes were not an indicator of the disease’s severity [21]. Other reports have shown that neutropenia is most common [18,20]. Generally, lymphocytosis has been found in infants [22]. Kosmeri et al stated that platelets are generally normal in children with COVID-19 [22]. Ambike and Bijarniya stated that thrombocytopenia is rare in children with COVID-19 [23], but Kharoud et al and Yarali et al reported that there is also thrombocytosis in children with COVID-19 [18,20]. Inflammatory biomarkers are usually increased, such as the ESR [18]. Pediatric patients with COVID-19 also showed an increase in AST and ALT [18]. In some cases, there was an increase in ESR, AST, ALT, and lymphocytosis [21,22]. Even though our patient had not shown signs of difficulty breathing, the patient was febrile, the ESR was increased, the chest X-ray showed pneumonia, and the PCR test was positive; therefore, we gave the patient azithromycin. According to the Indonesian COVID-19 management protocol, children with pneumonia and a positive PCR test for SARS-CoV-2 should be given azithromycin, corticosteroid, vitamin C, and zinc [24]. Vitamin C and zinc had been given to increase the immune system of the child [25].
The main goal of ITP treatment is not merely to increase the platelet count to an expected value, but is to achieve an effective platelet count to stop bleeding. Generally, if the platelet count is above 30 000/uL, the clinical experience will be better. In the present case, the platelet increment was significant even after the methylprednisolone pulse was given only twice. After 4 administrations of methylprednisolone, the platelet count had reached an expected value of 315 000/uL.
At the start of the disease, we cannot differentiate between acute and chronic ITP. In infants with ITP, 9% will have chronic ITP. In children with ITP aged 25 to 119 months, 18% will develop chronic ITP, and in children ≥120 months with ITP, 47% will develop chronic ITP [26]. Jung et al reported that 85.9% of children with ITP would have remission and 14.1% would have chronic ITP; the prognostic factors for chronic ITP are children of an older age and the slow appearance of clinical symptoms [27]. Makis et al reported that the prognostic factors for developing chronic ITP are age >10 years, no previous infection history, longer duration of clinical manifestations, and initial platelet count >10 000/uL [28]. Other investigators reported that the prognostic factors that provided complete remission when ITP was established were a low mean platelet volume level <8 fL and previous infection history [29]. The prognosis of patients with COVID-19 is inadequate if the neutrophil to lymphocyte ratio is ≥3.13 [30]. If the treatment of acute ITP is required, megadose methylprednisolone is the most effective and affordable approach. The chronicity of acute ITP would probably be less with megadose methylprednisolone treatment [31]. In the present case, the patient was 9 months old at the time of initial diagnosis of ITP, with a history of illness 3 days before presentation, bleeding that appeared suddenly, initial platelet count <20 000/uL, neutrophil to lymphocyte ratio of 0.42, and complete remission within 2 weeks; therefore, the prognosis of this patient was good, and he will not develop chronic ITP.
Conclusions
COVID-19 screening is highly recommended during this pandemic to identify it as a potential cause of childhood ITP. In cases when IVIG is not available or the patient is financially disadvantaged, megadose methylprednisolone can be an effective and inexpensive primary choice. Megadose methylprednisolone should be given in the acute phase of ITP to increase the platelet count as soon as possible.
References:
1.. Kühne T, Buchanan GR, Zimmerman S, A prospective comparative study of 2540 infants and children with newly diagnosed idiopathic thrombocytopenic purpura (ITP) from the intercontinental childhood ITP study group: J Pediatr, 2003; 143(5); 605-8
2.. DiMaggio D, Anderson A, Bussel JB, Cytomegalovirus can make immune thrombocytopenic purpura refractory: Br J Haematol, 2009; 146(1); 104-12
3.. Rand PhD ML, Fraser Wright Ph.D J, Virus-associated idiopathic thrombocytopenic purpura: Transfus Sci, 1998; 19(3); 253-29
4.. Kitamura K, Ohta H, Ihara T, Idiopathic thrombocytopenic purpura after human herpesvirus 6 infection: Lancet, 1994; 344(8925); 830
5.. Hamada M, Yasumoto S, Furue M, A case of varicella-associated idiopathic thrombocytopenic purpura in adulthood: J Dermatol, 2004; 31(6); 477-79
6.. Tsao HS, Chason HM, Fearon DM, Immune thrombocytopenia (ITP) in a pediatric patient positive for SARS-CoV-2: Pediatrics, 2020; 146(2); e20201419
7.. Rodeghiero F, Stasi R, Gernsheimer T, Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: Report from an international working group: Blood, 2009; 113(11); 2386-93
8.. Fogarty PF, Segal JB, The epidemiology of immune thrombocytopenic purpura: Curr Opin Hematol, 2007; 14(5); 515-19
9.. Yong M, Schoonen WM, Li L, Epidemiology of paediatric immune thrombocytopenia in the General Practice Research Database: Br J Haematol, 2010; 149(6); 855-64
10.. Hashemi A, Kargar F, Souzani A, Hazar N, Acute immune thrombocytopenic purpura in infants key words: Iran J Pediatr Hematol Oncol, 2011; 1(3); 104-9
11.. Rosthøj S, Hedlund-Treutiger I, Rajantie J, Duration and morbidity of newly diagnosed idiopathic thrombocytopenic purpura in children: A prospective Nordic study of an unselected cohort: J Pediatr, 2003; 143(3); 302-7
12.. Arnold DM, Bleeding complications in immune thrombocytopenia: Hematology, 2015; 2015(1); 237-42
13.. Albayrak D, Islek I, Kalayci AG, Nuran Gurses N, Acute immune thrombocytopenic purpura’ A comparative study of very high oral doses of methylprednisolone and intravenously administered immune globulin: J Pediatr, 1994; 125(6); 1004-7
14.. Henry BM, Lippi G, Plebani M, Laboratory abnormalities in children with novel coronavirus disease 2019: Clin Chem Lab Med, 2020; 58(7); 1135-38
15.. Patel NA, Pediatric COVID-19: Systematic review of the literature: Am J Otolaryngol-Head Neck Med Surg, 2020; 41(5); 102573
16.. Dhochak N, Singhal T, Kabra SK, Lodha R, Pathophysiology of COVID-19: Why children fare better than adults?: Indian J Pediatr, 2020; 87(7); 537-46
17.. Hoang A, Chorath K, Moreira A, COVID-19 in 7780 pediatric patients: A systematic review: EClinicalMedicine, 2020; 24; 100433
18.. Kharoud HK, Asim R, Siegel L, Review of clinical characteristics and laboratory findings of COVID-19 in children-Systematic review and Meta-analysis: medRxiv, 2020; 612; 2020
19.. Dong Y, Dong Y, Mo X, Epidemiology of COVID-19 among children in China: Pediatrics, 2020; 145(6); e20200702
20.. Yarali N, Akcabelen YM, Unal Y, Parlakay AN, Hematological parameters and peripheral blood morphologic abnormalities in children with COVID-19: Pediatr Blood Cancer, 2021; 68(2); e28596
21.. Henry BM, Benoit SW, de Oliveira MHS, Laboratory abnormalities in children with mild and severe coronavirus disease 2019 (COVID-19): A pooled analysis and review: Clin Biochem, 2020; 81; 1-8
22.. Kosmeri C, Koumpis E, Tsabouri S, Hematological manifestations of SARS-CoV-2 in children: Pediatr Blood Cancer, 2020; 67(12); e28745
23.. Ambike D, Bijarniya K, Asymptomatic pediatric COVID-19 presenting with thrombocytopenia – a rare finding: Indian J Pediatr, 2020; 88(1); 103-4
24.. Kementrian Kesehatan RI, Protokol tata laksana COVID-19: Jakarta, 2021; 1-99, Hal [in Indonesian]
25.. Wintergerst E, Maggini S, Hornig D, Immune-enhancing role of vitamin C and zinc and effect on clinical conditions: Ann Nutr Metab, 2006; 50; 85-94
26.. Sandoval C, Visintainer P, Ozkaynak MF, Clinical features and treatment outcomes of 79 infants with immune thrombocytopenic purpura: Pediatr Blood Cancer, 2004; 42(1); 109-12
27.. Jung JY, Rum O A, Kim JK, Park M, Clinical course and prognostic factors of childhood immune thrombocytopenia: Single center experience of 10 years: Korean J Pediatr, 2016; 59(8); 335-40
28.. Makis A, Gkoutsias A, Palianopoulos T, Prognostic factors for immune thrombocytopenia outcome in Greek children: A retrospective single-centered analysis: Adv Hematol, 2017; 2017; 1-7
29.. Ahmed S, Siddiqui AK, Shahid RK, Prognostic variables in newly diagnosed childhood immune thrombocytopenia: Am J Hematol, 2004; 77(4); 358-62
30.. Liu J, Liu Y, Xiang P, Neutrophil-to-lymphocyte ratio predicts critical illness patients with 2019 coronavirus disease in the early stage: J Transl Med, 2020; 18(1); 206
31.. Özsoylu Ş, Megadose methylprednisolone for childhood idiopathic trombocytopenic purpura (ITP): Turkish J Med Sci, 2005; 35(6); 347-56
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133