31 August 2021: Articles 
Vulvar Paget’s Disease Presenting with Fever and Left Inguinal and Peritoneal Lymphadenopathies
Mistake in diagnosis
Risa Hirata1ABEF, Masaki Tago1AEF*, Yoshio Hisata1AEF, Shu-ichi Yamashita1AEFDOI: 10.12659/AJCR.931600
Am J Case Rep 2021; 22:e931600
Abstract
BACKGROUND: Asymptomatic vulvar Paget’s disease is rare and commonly presents with vulvar eczema, erosions, or pruritus. The time from onset to diagnosis of vulvar Paget’s disease tends to be rather long because of difficulty making a correct diagnosis owing to similar skin findings with eczema or patients’ reluctance to undergo physical examination of their pubic area because of embarrassment.
CASE REPORT: A 55-year-old woman experienced recurrent episodes of fever for 10 months. Her primary care physician indicated inguinal lymphadenopathy 2 months prior to presentation at our hospital. Contrast-enhanced abdominal computed tomography revealed multiple intra-abdominal lymphadenopathies. With the failure of finding the primary lesion after biopsy, and with a diagnosis of metastatic carcinoma, she was referred to our hospital. On admission, she did not report having vulvar symptoms. As imaging studies revealed no primary lesions, we subsequently added immunostaining to the lymph node biopsy specimens, which suggested Paget’s disease. We finally performed a vulvar physical examination and identified eczema. We performed a skin biopsy and histopathological examinations, which provided the final diagnosis of vulvar Paget’s disease.
CONCLUSIONS: We experienced a case of vulvar Paget’s disease presenting with inguinal and intraperitoneal lymphadenopathies, without a patient report of vulvar symptoms. When identifying lymphadenopathies, it is crucial to obtain a careful history and perform appropriate physical examinations, suspecting diseases of the vulva or perineum. In addition, immunostaining of lymph node biopsy specimens could be useful in making a correct diagnosis.
Keywords: delayed diagnosis, lymphadenopathy, Paget’s Disease, Extramammary, Female, Humans, Paget Disease, Extramammary, Vulvar Neoplasms
Background
Vulvar Paget’s disease, a type of extramammary Paget’s disease, is a rare type of malignancy [1], especially in Japan, which originates from skin appendages such as apocrine glands [1]. Many cases of extramammary Paget’s disease were reported to have vulvar abnormalities, 87% to 98% of which were common skin lesions, such as eczema or erosions, followed by 78% to 98% which were pruritus [2,3]. Vulvar Paget’s disease is well known for the notoriously long time needed to achieve a correct diagnosis [2,3]. The time from onset to diagnosis of vulvar Paget’s disease is approximately 20 months [2–4] because it is extremely difficult to distinguish the skin lesions of Paget’s disease from benign conditions, such as contact dermatitis, fungal infections, or Bowen’s disease, using only gross physical findings without biopsy [1,5]. Therefore, biopsy is essential with recalcitrant vulvar skin lesions, despite usual treatments [6]. Furthermore, vulvar Paget’s disease could present only as inguinal lymphadenopathies as a chief complaint without vulvar symptoms [5], and patients’ embarrassment regarding examination of their pubic area could actually prevent physical examination [7,8].
We report a case of a patient with vulvar Paget’s disease showing fever and lymphadenopathies, without a report of vulvar skin symptoms, whose correct diagnosis was suspected only after histopathological examination of the left inguinal and intra-abdominal lymphadenopathies.
Case Report
A 55-year-old woman experienced recurrent episodes of fever >38.5°C, generalized arthralgia, and myalgia lasting a few days, with spontaneous improvement without respiratory, urinary, or gastrointestinal symptoms, once a month since onset. She visited her primary care physician, who indicated inguinal lymphadenopathy in the eighth month after onset. Her fever frequency increased to twice a month by the tenth month, without night sweats or weight loss. Contrast-enhanced computed tomography (CT) revealed left inguinal and multiple intra-abdominal lymphadenopathies. Histopathological findings for the inguinal lymph node biopsy specimens suggested metastatic carcinoma, but a primary lesion was not identified. She was referred to our hospital in the tenth month.
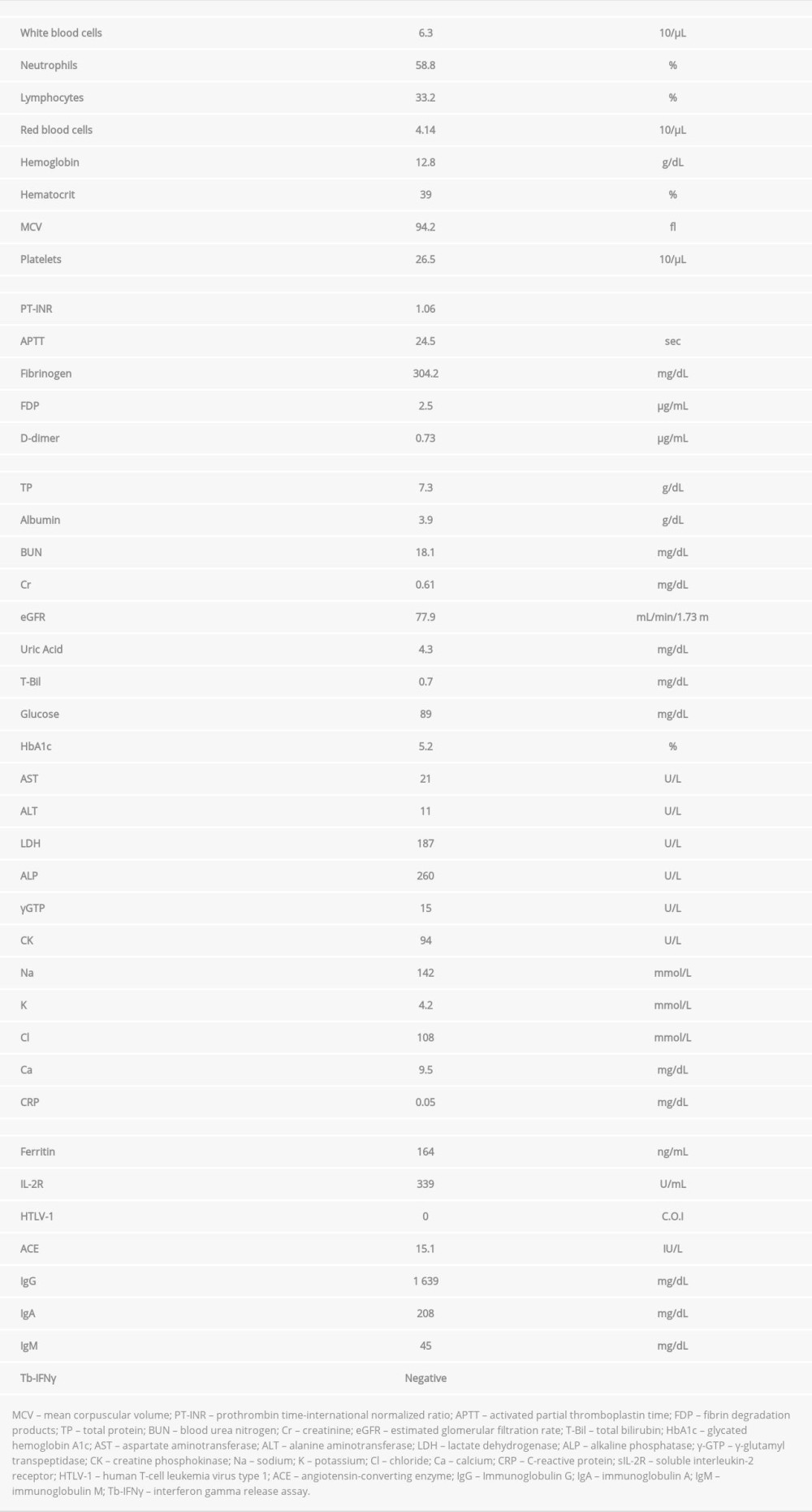
On her first visit to our hospital, the patient’s height was 161.8 cm, body weight was 49.2 kg, body mass index was 18.9 kg/m2, body temperature was 36.5°C, blood pressure was 113/70 mm Hg, pulse rate was 62 beats/min, percutaneous oxygen saturation was 96% on room air, and respiratory rate was 12 breaths/min. Physical examination revealed movable and painless left inguinal lymphadenopathies. The cervical, supraclavicular, and axillary lymph nodes were impalpable, and there were no other abnormalities in her trunk and extremities. The laboratory findings on admission are shown in Table 1. The white blood cell count was 6 300/µL; neutrophils, 58.8%; lymphocytes, 33.2%; hemoglobin, 12.8 g/dL; and platelets, 265 000/µL, with no abnormal cells in the peripheral blood. Other results were as follows: total protein, 7.3 g/dL; albumin, 3.9 g/dL; lactate dehydrogenase (LDH), 187 U/L; C-reactive protein, 0.05 mg/dL; ferritin, 164 ng/mL; and sIL-2R, 339 U/mL. Renal and liver functions were normal with normal immunoglobulin concentrations. No antibodies to
When we obtained another history from the patient, focusing on her vulvar symptoms and signs and suspecting vulvar Paget’s disease, she finally and reluctantly disclosed the long history of having pruritus and discharge related to eczematous lesions on her vulvar and perineal areas, which had been treated by her dermatologist with topical dermatological medications without improvement for 26 months before the first visit to our hospital. We subsequently examined her vulvar and perineal areas, which revealed erythematous lesions with erosions spreading from the area just above the clitoris, bilateral labia minora and labia majora, and left perineal area to the anus (Figure 4). The pelvic examination and vaginal speculum examination showed no abnormalities, and the cervical cytology was negative. As these skin findings strongly suggested vulvar Paget’s disease, we performed skin biopsy of a vulvar lesion and histopathological examinations, which provided a final diagnosis of vulvar Paget’s disease (Figure 5). Finally, other imaging tests showed no abnormalities in the uterus or ovaries. She was treated with anti-cancer chemotherapy, including docetaxel, after undergoing surgical resection of vulvar and perineal tumors. She has been receiving anti-cancer chemotherapy, including docetaxel, for more than 16 months.
Discussion
Patients with vulvar Paget’s disease presenting with fever and lymphadenopathies as chief symptoms are extremely rare, with only one report showing inguinal lymphadenopathy as a chief symptom [5]. In the present case, we initially tried making a diagnosis by finding the cause of fever and lymphadenopathy. Culprit lesions of inguinal lymphadenopathy are often found with other pathological conditions of the lower extremities, vulvae, anus, or genitalia [9], including malignant lymphoma, gastrointestinal, urinary tract, or gynecological malignancies, tuberculosis [10], sarcoidosis [11], or cat scratch disease [12]. We used PET-CT to find the primary lesion, suspecting malignant lymphoma or gastrointestinal malignancies as prime candidates, which failed to identify lesions in the pelvic cavity.
The key to making the diagnosis of vulvar Paget’s disease is to obtain a meticulous history and to perform appropriate physical examination of skin lesions, which we failed to perform until the histopathological examination findings suggested the diagnosis. Although our patient had actually experienced vulvar symptoms for a long time, we were unable to realize that because of our failure to obtain an appropriate history on her first visit to our department. In Japan, physical examinations of the pubic area, especially of women, are rarely performed without reports of symptoms by the patient. Practically, a patient’s embarrassment [7,8] or misunderstanding of the lesion could force a patient to leave lesions in the pubic area untreated, which could prevent an appropriate diagnostic process. From an anatomical point of view, because lymphatic vessels distributed in the dermal tissue of the vulva run horizontally to superficial inguinal lymph nodes, whereas lymphatic vessels distributed in the clitoris and anterior labia minora flow directly into the inguinal or internal iliac lymph nodes [13], physical examination of the vulvar area is mandatory when inguinal lymphadenopathies are seen, especially without detecting a primary lesion in the pelvis or lower extremities. Thus, repetitive history taking or physical examinations in accordance with a system review [14] and anatomical knowledge [13] are valuable to prevent inadequate medical practice. In addition, a respectful attitude by the physician to make a patient feel less embarrassed is required when proposing and explaining procedures to the patient as well as when performing the procedures, when examinations of embarrassing areas are mandatory. Needless to say, it is also essential to create good physician–patient relationships in daily medical practice [15].
In the present case, we added immunostaining to the inguinal lymph node biopsy specimens obtained at the previous hospital, which helped us make a correct diagnosis. More specifically, positive CK7 and negative CK20 led us to suspect carcinoma of the lung, breast, thyroid, endometrium, bile duct, or pancreas [16,17], resulting in adequate denial of those as a primary lesion by subsequent appropriate examinations. Moreover, positive results for CK7, CEA, and GCDFP-15, which were reported to be 100%, 84.2%, and 52.6% positive, respectively, by special immunostaining in a study of primary extramammary Paget’s disease, led us to suspect extramammary Paget’s disease as the primary lesion [18]. Therefore, it is essential to add immunostaining of the lymph node biopsy specimens, especially when a primary lesion for lymph node metastasis is not readily found with standard screening examinations.
Conclusions
Patients with vulvar Paget’s disease can present with inguinal or intraperitoneal lymphadenopathies without reports of vulvar symptoms. For such patients, it is crucial to obtain a meticulous history and perform appropriate physical examinations, suspecting the presence of vulvar or perineal diseases. In addition, immunostaining of lymph node biopsy specimens could help make a correct diagnosis.
Figures
References:
1.. Loiacono RMR, Traversi P, Deliso MA, Paget disease of the vulva an analysis of 24 cases: Medicine (Baltimore), 2019; 98; 41
2.. Tebes S, Cardosi R, Hoffman M, Paget’s disease of the vulva: Am J Obstet Gynecol, 2002; 187; 281-84
3.. Shaco-Levy R, Bean SM, Vollmer RT, Paget disease of the vulva: A study of 56 cases: Eur J Obstet Gynecol Reprod Biol X, 2010; 149; 86-91
4.. Jones IS, Crandon A, Sanday K, Paget’s disease of the vulva: Diagnosis and follow-up key to management; A retrospective study of 50 cases from Queensland: Gynecol Oncol, 2011; 122; 42-44
5.. Chan JYW, Li GKH, Chung JHP, Chow VLY, Extramammary Paget’s disease: 20 years of experience in Chinese population: Int J Surg Oncol, 2012; 2012; 416418
6.. Hillmann BR, Pereira AA, Sommacal LF, Extramammary Paget disease of the vulva-case report: Rev Bras Ginecol Obstet, 2016; 38; 524-28
7.. Gavriilidis P, Chrysanthopoulos K, Gerasimidou D, Extramammary Paget’s disease of the vulva: Case Rep, 2013; 2013; bcr2013200623
8.. Hinten F, Molijn A, Eckhardt L, Vulvar cancer: Two pathways with different localization and prognosis: Gynecol Oncol, 2018; 149; 310-17
9.. Zaren HA, Copeland EM, Inguinal node metastases: Cancer, 1978; 41; 919-23
10.. Yang ZG, Min PQ, Sone S, Tuberculosis versus lymphomas in the abdominal lymph nodes: Evaluation with contrast-enhanced CT: Am J Roentgenol, 1999; 172; 619-23
11.. Yanardag H, Caner M, Papila I, Diagnostic value of peripheral lymph node biopsy in sarcoidosis: A report of 67 cases: Can Respir J, 2007; 14; 209-11
12.. Mancino P, Ucciferri C, Falasca K: Infez Med, 2008; 16; 91-93
13.. Way S, Hunterian Lecture: Hunterian Lecture delivered at the Royal College of Surgeons of England on 13th February, 1948: Ann R Coll Surg Engl, 1948; 3; 187
14.. Bickley L: Bates’ guide to physical examination and history-taking 11th, 2012; 10-13, Lippincott Williams & Wilkins
15.. Evans BJ, Kiellerup FD, Stanley RO, A communication skills programme for increasing patients’ satisfaction with general practice consultations: Br J Med Psychol, 1987; 60(4); 373-78
16.. Fizazi K, Greco FA, Pavlidis N, Pentheroudakis G, Cancers of unknown primary site: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up: Ann Oncol, 2011; 22; vi64-68
17.. Chu P, Wu E, Weiss LM, Cytokeratin 7 and cytokeratin 20 expression in epithelial neoplasms: A survey of 435 cases: Mod Pathol, 2000; 13; 962-72
18.. Zhao Y, Gong X, Li N, Primary extramammary Paget’s disease: A clinicopathological study of 28 cases: Int J Clin Exp Pathol, 2019; 12; 3426-32
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133