25 June 2021: Articles 
Rivaroxaban for Treatment of Left Ventricular Thrombus: A Case Report
Unusual clinical course, Unusual or unexpected effect of treatment, Educational Purpose (only if useful for a systematic review or synthesis)
Juhaina Salim Al-Maqbali1ABDEF, Maitha Al-Sibani2BE, Nasiba Al-Maqrashi3ABD, Abdullah M. Al Alawi24AEF*, Hatim Al LawatiDOI: 10.12659/AJCR.932140
Am J Case Rep 2021; 22:e932140
Abstract
BACKGROUND: Left ventricular thrombus (LVT) is a complication of left ventricular dysfunction and myocardial infarction (MI) and is associated with systemic thromboembolism. Two-dimensional transthoracic echocardiography (TTE) is considered the first-line diagnostic tool for detection of LVT. Vitamin K antagonists (VKA) targeting an international normalized ratio (INR) from 2 to 3 are the only approved treatments by the Food and Drug Administration (FDA). New emerging observational data support the use of direct oral anticoagulants (DOACs) as an alternative therapeutic option; however, their safety and efficacy have not been assessed in a good-quality randomized controlled trial.
CASE REPORT: Here, we present a case of a 43-year-old man diagnosed with human immunodeficiency virus (HIV)-associated dilated cardiomyopathy complicated with an LVT. He was treated with rivaroxaban for 9 consecutive months with no interruption of therapy at any point in time; however, he presented to the emergency department with symptoms of decompensated heart failure. A follow-up TTE demonstrated a significant increase in the size of his LVT. This case questions the efficacy of using factor Xa inhibitor (rivaroxaban) as an alternative option for LVT treatment.
CONCLUSIONS: This case demonstrates a failure of rivaroxaban in treating LVT in a patient with HIV-associated dilated cardiomyopathy. Good-quality randomized clinical trials or prospective studies are required to establish the efficacy and safety of DOACs for LVT treatment as an alternative to VKA.
Keywords: Atrial Flutter, Cardiomyopathy, Dilated, Embolism and Thrombosis, Factor Xa Inhibitors, Warfarin, Thrombosis, Anticoagulants, Prospective Studies, Rivaroxaban
Background
Left ventricular thrombus (LVT) is a known complication of myocardial infarction (MI) due to blood stasis, subendocardial injury, and hypercoagulable state. In addition, it often complicates large anterior left ventricular wall infarcts [1–3]. LVT often occurs between 1 to 14 days after MI and, if untreated, is associated with an increased risk of systemic embolization [4,5]. LVT can also occur in patients with severe left ventricular systolic dysfunction due to non-ischemic cardiomyopathy [6]. The estimated prevalence of LVT in patients with severe systolic dysfunction ranges from 2.1% to 7.0%, compared to 7% to 22% following MI [7].
LVT can be detected by two-dimensional transthoracic echocardiography (TTE), computed tomography (CT), or cardiac magnetic resonance imaging (CMRI) [1]. TTE is the first-line diagnostic modality due to its widespread availability. However, TTE has low sensitivity (21% to 35%) to detect mural LVT compared to CMRI, which has a reported sensitivity of up to 88% [1,8].
Vitamin K antagonists (VKA) are approved by the Food and Drug Administration (FDA) for treatment of LVT, and the American Heart Association (AHA) and the American Stroke Association (ASA) recommend the use of VKA targeting international normalized ratio (INR) from 2 to 3 [8,9]. Direct oral anticoagulants (DOACs) have been prescribed for LVT treatment, but their safety and efficacy have not been assessed in a good-quality randomized controlled trial (RCT) [2,10]. Generally, anticoagulation is maintained for 3 to 6 months, but the optimal treatment duration is unknown [2]. The decision to stop anticoagulation is usually made upon documenting complete thrombus resolution on follow-up imaging tests [6]. The present case reports documents failure of a trial of rivaroxaban that resulted in the progression of LVT. We present a few potential hypotheses for the failure of the desired therapeutic effect and review the evidence-based treatment of LVT.
Case Report
A 43-year-old man presented to the emergency department (ED) at Sultan Qaboos University Hospital (SQUH) in December 2020 with progressive dyspnea, ankle swelling, and abdominal fullness. His medical background included human immunodeficiency virus (HIV) infection on antiretroviral therapy with a good virologic response, HIV-associated dilated cardiomyopathy (left ventricular ejection fraction (LVEF) <20%), atrial flutter, and chronic limb ischemia. In November 2019 (13 months earlier), TTE demonstrated an apical thrombus measuring 24×35 mm (Figures 1A, 2A). The patient was anticoagulated with VKA (warfarin), targeting a therapeutic INR of 2 to 3 with a documented reduction in the size to 10×35 mm on the repeat TTE done 4 months later in March 2020 (Figures 1B, 2B). As a long-term anticoagulant option for both atrial flutter and LVT, and to obviate the need for regular INR monitoring, the patient was shifted to rivaroxaban 20 mg once daily. Other regular medications were carvedilol 12.5 mg 2 times daily, frusemide 40 mg 2 times daily, spironolactone 25 mg once daily, and antiretroviral combination therapy of efavirenz 600 mg/emtricitabine 200 mg/tenofovir 300 mg (atripla) once daily. He was an active smoker and consumed alcohol regularly.
On presentation to ED, his heart rate was 140 beats/min, blood pressure of 118/82 mmHg, and O2 saturation of 96% on room air. A physical examination revealed elevated jugular venous pressure; diminished breath sounds in the right lung base with fine crackles heard bilaterally. In addition, he had ascites and bilateral pitting leg edema. Other system examinations were unremarkable.
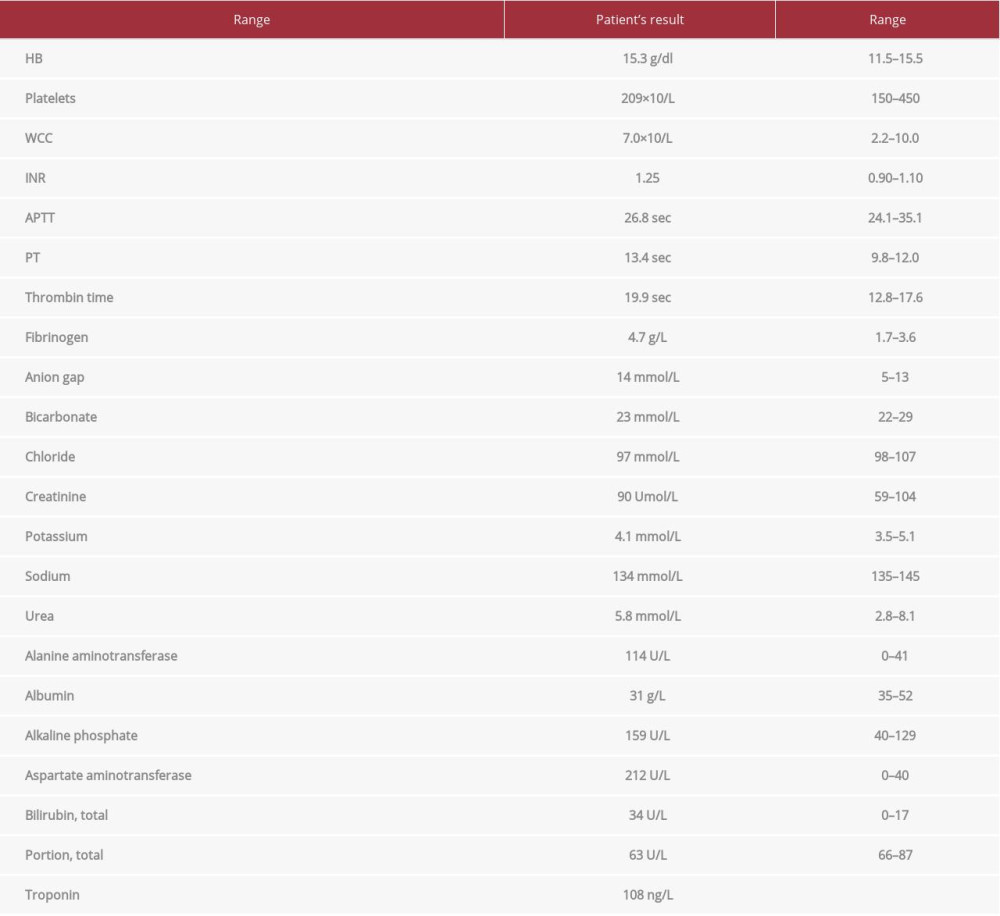
His laboratory tests (Table 1) were notable for troponin-T of 108 ng/L (normal reference range <14 ng/L) and INR of 1.25, with subsequent serial cardiac troponin levels unchanged. An electrocardiogram (ECG) showed sinus tachycardia of 130 beats per min; no significant ST-segment or T-wave changes. Chest radiography showed cardiomegaly, right-sided pleural effusion, and prominent pulmonary vascular markings consistent with pulmonary edema. The patient was admitted to the hospital to manage acute decompensated heart failure and to further optimize anti-failure therapy.
Follow-up TTE was arranged to assess LV systolic function, and it showed a significant increase in the size of LVT from 10×35 mm to 27×60 mm in diameter in the apical three 26×33 mm in the apical four-chamber view. The thrombus occupied the entire LV apex, and it was quite mobile, especially the part extending to the LVOT (Figures 1C, 2C). The patient reported that he was taking the rivaroxaban as prescribed, with no interruption at any point in time.
Considering the progression of LVT, rivaroxaban was switched back to warfarin. The patient was bridged with unfractionated heparin infusion. He was discharged home after he became euvolemic, with a therapeutic INR of 2.63.
Discussion
In this patient, despite 9 months of strict compliance, rivaroxaban at a dose of 20 mg daily was not effective in resolving the patient’s LVT; on the contrary, there was a paradoxical and significant increment in the clot burden as seen on his repeat TTE study. By contrast, there was an adequate clinical response with warfarin, evidenced by reducing LVT size on the first follow-up TTE 4 months after initiating warfarin.
The RED VELVET multicenter cohort showed that over a median follow-up of 351 days, patients anticoagulated with a DOAC, mainly patients on rivaroxaban and apixaban, has a significantly higher risk of stroke or systemic embolism (HR, 2.07; 95% CI, 1.17–3.66;
The No-LVT trial was the first randomized controlled study to compare rivaroxaban’s efficacy and safety for this indication compared to warfarin. Abdelnabi et al presented the results of this prospective, open-label multicenter study at the 2020 AHA annual scientific session. Rivaroxaban therapy was associated with a significantly greater thrombus resolution by 1 month (79% vs 47.5%,
It was unclear why the LVT progressed while the patient was on rivaroxaban despite strict compliance by our patient.
Drug-drug interactions were considered. All of his regular medications (carvedilol, frusemide, spironolactone, and Atripla) were cross-checked against rivaroxaban using validated drug-interaction tools (Lexicomp and Micromedex). There were no major interactions detected among all medications except with Atripla, which can reduce the efficacy of rivaroxaban via inhibition of CYP3A4 enzyme (class B). In addition, rivaroxaban has no interaction with ethanol as per the manufacturing label [18–20].
Patients with reduced ejection fraction are vulnerable to developing LVT [6]. Therefore, heart failure therapy, including beta-blockers, diuretics, and mineralocorticoid antagonists, was started in our patient but maintained at low doses due to symptomatic hypotension.
There is a paucity of systematic studies such as RCTs describing the efficacy of DOACs in treating LVT. The dose regimen used currently has been extrapolated from the non-valvular atrial fibrillation literature. It is conceivable that higher doses may be more effective for LV thrombus resolution [6]. However, unconventionally higher doses of rivaroxaban may be associated with unacceptably high bleeding risk. Therefore, until further evidence is available, no conclusions can be drawn from the currently available literature to guide clinical practice.
Conclusions
This case demonstrates the failure of rivaroxaban in treating LVT in a patient with HIV-associated dilated cardiomyopathy. At present, VKA is the only FDA-approved anticoagulant for this indication. DOACs are an attractive alternative given the ease of use and lack of need for regular monitoring of anticoagulant effect. However, their use for this specific indication remains questionable until more evidence is available to prove their efficacy and safety.
Figures
References:
1.. Delewi R, Zijlstra F, Piek JJ, Left ventricular thrombus formation after acute myocardial infarction: Heart, 2012; 98(23); 1743-49
2.. Iqbal H, Straw S, Craven TP, Direct oral anticoagulants compared to vitamin K antagonist for the management of left ventricular thrombus: ESC Heart Fail, 2020; 7(5); 2032-41
3.. Moey MYY, Tomdio AN, Achike O, Kabirdas D, Large left ventricular thrombus as a cause of recurrent cardioembolic stroke while on dabigatran: CASE (Phila), 2018; 2(5); 222-24
4.. Vaitkus PT, Barnathan ES, Embolic potential, prevention and management of mural thrombus complicating anterior myocardial infarction: A meta-analysis: J Am Coll Cardiol, 1993; 22(4); 1004-9
5.. Keren A, Goldberg S, Gottlieb S, Natural history of left ventricular thrombi: their appearance and resolution in the posthospitalization period of acute myocardial infarction: J Am Coll Cardiol, 1990; 15(4); 790-800
6.. Habash F, Vallurupalli S, Challenges in management of left ventricular thrombus: Ther Adv Cardiovasc Dis, 2017; 11(8); 203-13
7.. Zhou X, Shi R, Wu G, The prevalence, predictors, and outcomes of spontaneous echocardiographic contrast or left ventricular thrombus in patients with HFrEF: ESC Heart Fail, 2021; 8(2); 1284-94
8.. McCarthy CP, Vaduganathan M, McCarthy KJ, Left ventricular thrombus after acute myocardial infarction: Screening, prevention, and treatment: JAMA Cardiol, 2018; 3(7); 642-49
9.. Kernan WN, Ovbiagele B, Black HR, Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline for healthcare professionals from the American Heart Association/American Stroke Association: Stroke, 2014; 45(7); 2160-236
10.. Willeford A, Zhu W, Stevens C, Thomas IC, Direct oral anticoagulants versus warfarin in the treatment of left ventricular thrombus: Ann Pharmacother, 2020; 55(7); 839-45
11.. Robinson AA, Trankle CR, Eubanks G, Off-label use of direct oral anticoagulants compared with warfarin for left ventricular thrombi: JAMA Cardiol, 2020; 5(6); 685-92
12.. Daher J, Da Costa A, Hilaire C, Management of left ventricular thrombi with direct oral anticoagulants: Retrospective comparative study with vitamin K antagonists: Clin Drug Investig, 2020; 40(4); 343-53
13.. Ali Z, Isom N, Dalia T, Direct oral anticoagulant use in left ventricular thrombus: Thromb J, 2020; 18(1); 29
14.. Jones DA, Wright P, Alizadeh MA, The use of novel oral anti-coagulant’s (NOAC) compared to vitamin K antagonists (Warfarin) in patients with left ventricular thrombus after acute myocardial infarction (AMI): Eur Heart J Cardiovasc Pharmacother; 2020 [Online ahead of print]
15.. Fleddermann AM, Hayes CH, Magalski A, Main ML, Efficacy of direct acting oral anticoagulants in treatment of left ventricular thrombus: Am J Cardiol, 2019; 124(3); 367-72
16.. Kajy M, Shokr M, Ramappa P, Use of direct oral anticoagulants in the treatment of left ventricular thrombus: systematic review of current literature: Am J Ther, 2020; 27(6); e584-90
17.. Abdelnabi MH: Comparative study of oral. Anticoagulation in left ventricular thrombi (No-LVT Trial), 2020, America, AHA Annual Scientific Sessions
18.. : Xarelto (rivaroxaban) {prescribing information} March, 2017, Titusville, NJ, Janssen Pharmaceuticals Inc.
19.. : Xarelto (rivaroxaban) {product monograph} July, 2015, Toronto, Ontario, Canada, Bayer Inc.
20.. Altena R, van Roon E, Folkeringa R, Clinical challenges related to novel oral anticoagulants: Drug–drug interactions and monitoring: Haematologica, 2014; 99(2); e26-27
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133