08 September 2021: Articles 
Role of Cerebrolysin in Rehabilitation in Ischemic Stroke: A Case Report
Unusual or unexpected effect of treatment
Ratanapat Chanubol1ABCDEF*, Punjama Lertbutsayanukul2DEDOI: 10.12659/AJCR.932365
Am J Case Rep 2021; 22:e932365
Abstract
BACKGROUND: The criterion standard treatments for ischemic stroke patients, starting from systemic thrombolysis and/or undergoing endovascular recanalization therapy to intensive rehabilitation, are the best options available nowadays, but still cannot achieve total recovery. Neuroprotective and neurotrophic agents seem to be promising therapeutic targets in stroke, even in ischemic and/or hemorrhagic stroke, either in the acute stage or to support neuro-recovery in subacute to chronic stages. Therefore, new therapies are needed as adjuvants in the rehabilitation phase for promoting the recovery and monitoring adverse effects of treatment.
CASE REPORT: We describe a patient with an acute occlusion of the right middle cerebral artery who was treated with recombinant tissue-plasminogen activator (rtPA), underwent mechanical thrombectomy, and was then enrolled in a 1-month rehabilitation program. After the post-stroke recovery plateau, the patient received 10 days of 30 mL intravenous Cerebrolysin® to support further neuro-recovery, together with long-term rehabilitation. We utilized clinical standard assessment tools, including National Institutes of Health Stroke Scale (NIHSS), modified Rankin scale (mRS), modified Barthel Index (MBI), and function of ambulation, to evaluate the outcome of the patient, together with the adverse events monitoring. After Cerebrolysin® administration, the patient demonstrated improvement in all assessment scores at 1, 3, and 6 months.
CONCLUSIONS: Postoperative treatment with Cerebrolysin® in our patient with subacute ischemic stroke, after plateau recovery in the rehabilitation phase, together with the standard acute stroke regimen, improved the patient’s recovery outcomes. No serious adverse effects were observed.
Keywords: Rehabilitation, Stroke, Mechanical Thrombolysis, Amino Acids, Brain Ischemia, Fibrinolytic Agents, Humans, ischemic stroke, Thrombolytic Therapy, Tissue Plasminogen Activator
Background
Intravenous administration of recombinant tissue-plasminogen activator (rtPA) within 4.5 hours of stroke onset together with other endovascular reperfusion strategies has been approved as a standard of care for acute ischemic stroke [1,2]. Furthermore, intensive rehabilitation programs have shown to reduce neurological and physical impairment, and improve function [3]. However, despite all therapeutic efforts, patients with stroke often remain with impaired motor functions, limiting their activities of daily living. Therefore, they have an unmet need for recovery treatment. Cerebrolysin® has anti-apoptotic, anti-oxidative, anti-inflammatory properties and stimulates neurogenesis and synaptic plasticity, which accelerates recovery of neurological functions after stroke [4–6]. Recent evidence from some clinical trials suggests that the neurotrophic agent Cerebrolysin® supports neuro-recovery in the acute to the very early subacute phases of stroke [7,8], but there is still a lack of evidence in the late subacute to chronic phases.
Case Report
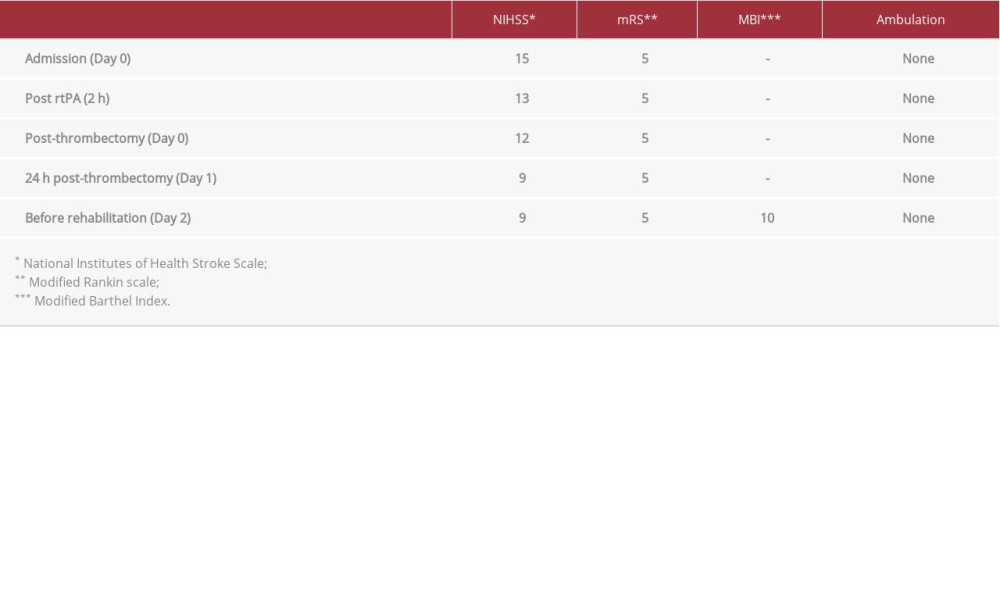
A 71-year-old Thai man, with a history of hypertension and atrial fibrillation, treated with Warfarin (3 mg) once at bedtime and Bisoprolol (2.5mg) once daily, was admitted to the hospital 1.5 hours after the onset of symptoms and presented with left hemiplegia, strabismus, severe dysarthria, left facial weakness, and left hemi-inattention with a baseline National Institutes of Health Stroke Scale (NIHSS) score of 15. His premorbid status was normal ambulation and independence in activities of daily living (modified Rankin scale=0). The non-contrast computed tomography (CT) scan revealed right hyperdense middle cerebral artery (MCA) sign and hyperacute right MCA infarction with an Alberta Stroke Program Early Computed Tomography Score (ASPECTS) of 7. The thrombolysis therapy was initiated according to the standard protocol by intravenous (i.v.) rtPA, total 66 mg (0.9 mg/kg). Computed tomography angiography (CTA) of the brain and neck after the rtPA treatment revealed right proximal MCA occlusion around the M2 segment, with an NIHSS score of 13. The patient was immediately referred to our institution for the mechanical thrombectomy. The thrombectomy was performed within 5 hours after stroke onset by using the stent retriever. An angiogram of the right internal carotid artery (ICA) revealed a complete occlusion of the whole posterior division of the right MCA (M2 segment), and distal occlusion of a few right ACA branches, including posterior internal frontal and paracentral sulcus branches. A post-thrombectomy angiogram showed recanalization of right proximal MCA and branches with a Thrombolysis in Cerebral Infarction (TICI) score of 3. No immediate complications were noted. The pre- and post-thrombectomy angiogram images of the right ICA are shown in Figures 1 and 2. An immediate post-thrombectomy CT brain showed a small hypodense hyperacute infarction at the right paracentral lobule, right preand post-central gyri, and right insular lobe. The patient still had hemiplegia, with a NIHSS score of 12. The 24-hour post-thrombectomy CT brain revealed intracerebral hemorrhage as a hyperdense lesion at the right pre- and postcentral gyri (Figure 3).
Serial assessments of the NIHSS scores and modified Rankin scale (mRS) were obtained by vascular neurologists in the acute phase (Table 1) from baseline (pre-rtPA-bolus) until day 2 of admission to the rehabilitation program. The manual muscle testing was still grade 0 on the left upper and lower extremities. The functional outcome, assessed by modified Barthel Index (MBI) and ambulatory function, was evaluated on the day of enrollment for rehabilitation.
According to the guidelines for adult stroke rehabilitation and recovery, 2016 [9], the patient was then admitted to the rehabilitation unit on day 2 after admission and had participated in the rehabilitation program for 7 months post stroke. As soon as he became neurologically stable, an intensive rehabilitation therapy program started for 3 hours per day on every working day (5 days per week) consisting of 1 hour of physical therapy, 1 hour of occupational therapy, half an hour of speech therapy, and resting time of 30 minutes. Two weeks after starting the rehabilitation program, none of the assessment scores or muscle power tests showed any improvement. By the completion of the 4-week program, he seemed to have plateaued, but a follow-up CT scan 2 weeks after completion of the rehabilitation program showed interval changes of a sub-acute right MCA infarction with decreased density of hemorrhagic transformation at the right frontal lobe and postcentral gyrus, and a small subacute cortical infarction at the right inferior parietal lobe (Figure 4). Cerebrolysin® was initiated once after 1 month of rehabilitation program by 30 mL Cerebrolysin® diluted in 70 mL normal saline intravenously for 10 consecutive days as an add-on to the conventional rehabilitation. Follow-up clinical assessments of impairment and disability were performed using the NIHSS, mRS, modified Barthel Index (MBI), and assessment of walking ability at baseline (before the initiation of Cerebrolysin® treatment) and at 1, 3, and 6 months after Cerebrolysin® administration. Oral medications were still taken: Dabigatran (150 mg) twice daily, Atorvastatin (40 mg) once daily, Bisoprolol (2.5 mg) once daily, Omeprazole (20 mg) once daily, and Lercanidipine (20 mg) once daily.
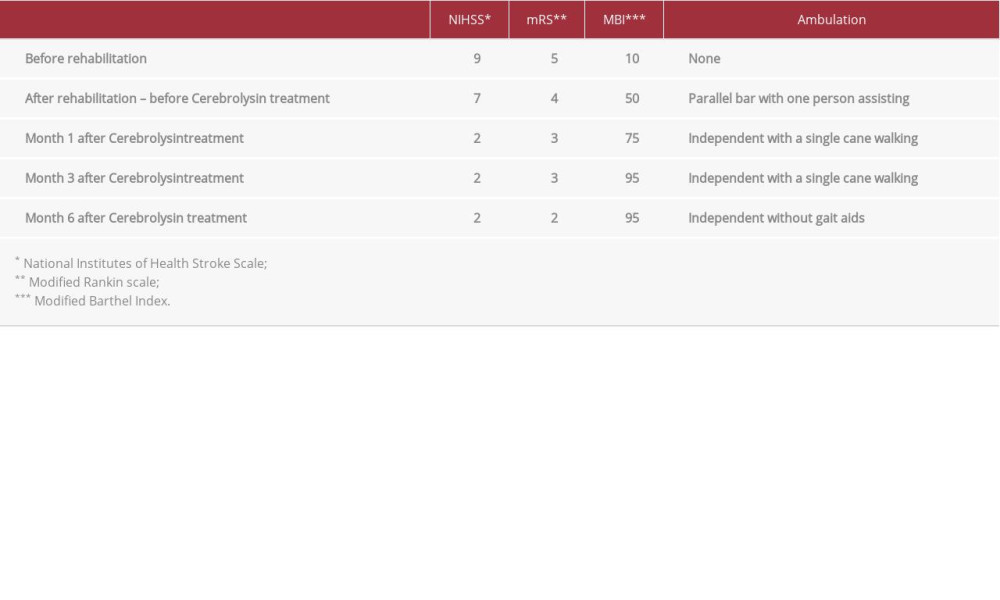
Initially, prior to the neurorehabilitation program, the patient had an NIHSS score of 9, scored 5 on the MRS, and 10 on the MBI. During the first 2 weeks of neurorehabilitation, the patient’s functional and neurological status had improved to an NIHSS score of 7, a mRS of 4, and a MBI score of 50. The power of the left arm was graded 3 and the left leg was graded 0 by manual muscle testing. The patient was able to ambulate in a parallel bar with a left knee brace, plastic ankle-foot orthosis, and 1 personal assistant. After the second to fourth weeks, neither muscle power nor any function had changed. The patient was re-evaluated and asked to continue the rehabilitation program as an inpatient for another 6 months.
Further significant clinical improvement was evident at the follow-up visits at 1, 3, and 6 months after the initiation of Cerebrolysin® administration. Again, impairment and disability were assessed by the NIHSS, mRS, MBI, and walking ability (Table 2). While the initial improvement achieved with neurorehabilitation reached a plateau with an NIHSS of 7, mRS of 4, and a MBI of 50, the patient experienced further significant and clinically relevant improvement after treatment with Cerebrolysin®. One month after Cerebrolysin® treatment, the patient’s neurological and functional status improved to a NIHSS of 2, mRS of 3, and MBI of 75 and he was already able to walk independently with a single cane. He continued to further improve his functional capabilities until month 3, with a MBI of 95, while the neurological status remained the same (NIHSS of 2). At the final examination 6 months after treatment with Cerebrolysin®, the patient was able to walk independently without gait aids. No serious adverse reactions to Cerebrolysin® occurred during or after the administration. He felt feverish and slightly uncomfortable during the i.v. administration. The patient was impressed with the standard of treatment, including the addition of the Cerebrolysin®, and has been able to return to work.
Discussion
There is still an ongoing controversy about the evidence of Cerebrolysin® in ischemic stroke patients. The findings in the Cochrane review in 2017 do not demonstrate clinical benefits of Cerebrolysin for treating acute ischemic stroke [10]. In contrast, a meta-analysis of 9 randomized clinical trials of Cerebrolysin® in 2018 demonstrated superiority of Cerebrolysin compared with placebo in different dosages from 30 to 50 mL for 10–21 days after stroke, with either an immediate onset of treatment or with a 7-day delay [7,11–15]. The clinical research on Cerebrolysin® in the late subacute to chronic phases is still ongoing, with only 1 published retrospective study on the reduction of spasticity, and 1 in patients with minimally conscious state after stroke [16,17]. Additionally, sufficient safety data in several neurological conditions, such as subarachnoid hemorrhage, traumatic brain injury, and mild cognitive impairment and dementia, have been published [18–20].
Generally, the published research clearly shows that patients with a baseline NIHSS of 15 only have a 23% chance of excellent recovery after 3 months [21]. We decided to treat such a patient with a 30-mL daily dosage for 10 days, which is the lowest dose, based on the established evidence and also based on the findings from the Copenhagen Stroke Study of 1995, which showed that functional recovery was completed within 12.5 weeks (95% confidence interval (CI) 11.6 to 13.4) from stroke onset in 95% of patients [22]. Finally, this patient became independent in activities of daily living and ambulation. He also returned to work (at home).
With an improved NIHSS score, modified Barthel index, modified Rankin scale, and walking ability, the short course of Cerebrolysin® has demonstrated effectiveness and safety in a subacute ischemic stroke patient who had reached a recovery plateau after 1 month of a neurorehabilitation program.
Cerebrolysin® together with rehabilitation dramatically improved the impairment and function in our patient, which had a great impact on his quality of life. The efficacy of Cerebrolysin® can be attributed to its neuroprotective and neurotrophic properties, similar to endogenous neurotrophic factors [23,24]. Further randomized controlled studies should be performed to compare the efficacy and safety of Cerebrolysin® at all stages of stroke recovery, especially the treatment window in late subacute to chronic phases of stroke. With increasing evidence from randomized clinical trials, the general public should benefit from Cerebrolysin treatment, as well as by reimbursed treatment costs.
Conclusions
The data suggest that Cerebrolysin® further enhances neurological recovery in late subacute stroke patients who previously underwent a standard acute stroke treatment regimen as well as a standard neurorehabilitation program. No serious adverse reaction was observed during and after the drug administration.
Figures
References:
1.. Catanese L, Tarsia J, Fisher M, Acute ischemic stroke therapy overview: Circ Res, 2017; 120(3); 541-58
2.. Powers WJ, Rabinstein AA, Ackerson T, Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association: Stroke, 2019; 50(12); e344-418
3.. Kwakkel G, van Peppen R, Wagenaar RC, Effects of augmented exercise therapy time after stroke: A meta-analysis: Stroke, 2004; 35(11); 2529-39
4.. Alcántara-González F, Mendoza-Perez CR, Zaragoza N, Combined administration of cerebrolysin and donepezil induces plastic changes in pre-frontal cortex in aged mice: Synapse, 2012; 66; 938-49
5.. Zhang C, Chopp M, Cui Y, Cerebrolysin enhances neurogenesis in the ischemic brain and improves functional outcome after stroke: J Neurosci Res, 2010; 88(15); 3275-81
6.. Zhang L, Chopp M, Meier DH, Sonic hedgehog signaling pathway mediates cerebrolysin-improved neurological function after stroke: Stroke, 2013; 44(7); 1965-72
7.. Bornstein NM, Guekht A, Vester J, Safety and efficacy of Cerebrolysin in early post-stroke recovery: A meta-analysis of nine randomized clinical trials: Neurol Sci, 2018; 39; 629-40
8.. Chang WH, Park CH, Kim DY, Cerebrolysin combined with rehabilitation promotes motor recovery in patients with severe motor impairment after stroke: BMC Neurol, 2016; 16; 31
9.. Winstein CJ, Stein J, Arena R, Guidelines for adult stroke rehabilitation and recovery: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association: Stroke, 2016; 47(6); e98-169
10.. Ziganshina LE, Abakumova T, Vernay L, Cerebrolysin for acute ischaemic stroke: Cochrane Database Syst Rev, 2017; 4(4); CD007026
11.. Heiss WD, Brainin M, Bornstein NM, Cerebrolysin in patients with acute ischemic stroke in Asia: Results of a double-blind, placebo-controlled randomized trial: Stroke, 2012; 43(3); 630-36
12.. Lang W, Stadler CH, Poljakovic Z, Fleet D, A prospective, randomized, placebo-controlled, double-blind trial about safety and efficacy of combined treatment with alteplase (rt-PA) and Cerebrolysin in acute ischaemic hemispheric stroke: Int J Stroke, 2013; 8(2); 95-104
13.. Gharagozli K, Harandi AA, Houshmand S, Efficacy and safety of Cerebrolysin treatment in early recovery after acute ischemic stroke: A randomized, placebo-controlled, double-blinded, multicenter clinical trial: J Med Life, 2017; 10(3); 153-60
14.. Muresanu DF, Heiss WD, Hoemberg V, Cerebrolysin and recovery after stroke (CARS): A randomized, placebo-controlled, double-blind, multi-center trial: Stroke, 2016; 47(1); 151-59
15.. Guekht A, Heiss D, Gusev E, Cerebrolysin and recovery after stroke (CARS 2): A randomized, placebo-controlled, double-blind, multicenter clinical study: J Neurol Sci, 2015; 357; e103
16.. Martinez RM, Efficacy of Cerebrolysin in the reduction of spasticity during stroke rehabilitation: J Med Life, 2017; 10(3); 161-66
17.. Kim JY, Kim HJ, Choi HS, Effects of Cerebrolysin® in patients with minimally conscious state after stroke: an observational retrospective clinical study: Front Neurol, 2019; 10; 803
18.. Woo PYM, Ho JWK, Ko NMW, Randomized, placebo-controlled, double-blind, pilot trial to investigate safety and efficacy of Cerebrolysin in patients with aneurysmal subarachnoid hemorrhage: BMC Neurol, 2020; 20(1); 401
19.. Chen CC, Weiv ST, Tsaia SC, Cerebrolysin enhances cognitive recovery of mild traumatic brain injury patients: Double-blind, placebo-controlled, randomized study: Br J Neurosurg, 2013; 27(6); 803-7
20.. Gavrilova SI, Alvarez A, Cerebrolysin in the therapy of mild cognitive impairment and dementia due to Alzheimer’s disease: 30 years of clinical use: Med Res Rev, 2020 [Online ahead of print]
21.. Adams HP, Davis PH, Leira EC, Baseline NIH Stroke Scale score strongly predicts outcome after stroke: A report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST): Neurology, 1999; 53(1); 126-31
22.. Jørgensen HS, Nakayama H, Raaschou HO, Outcome and time course of recovery in stroke. Part II: Time course of recovery. The Copenhagen Stroke Study: Arch Phys Med Rehabil, 1995; 76(5); 406-12
23.. Masliah E, Diez-Tejedor E, The pharmacology of neurotrophic treatment with Cerebrolysin: Brain protection and repair to counteract pathologies of acute and chronic neurological disorders: Drugs Today (Barc), 2012; 48(Suppl. A); 3-24
24.. Zhang L, Chopp M, Wang C, Prospective, double blinded, comparative assessment of the pharmacological activity of Cerebrolysin and distinct peptide preparations for the treatment of embolic stroke: J Neurol Sci, 2019; 398; 22-26
Figures
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942966
05 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942032
06 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942937
12 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943244
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250