11 October 2021: Articles 
Toxic Epidermal Necrolysis Caused by Allopurinol: A Serious but Still Underestimated Adverse Reaction
Rare disease, Adverse events of drug therapy
Daniel Hoyer1ACDEF*, Carlo Atti1ABCDE, Sebastian Nuding1E, Alexander Vogt1E, Daniel G. Sedding1E, Artjom Schott1ABCDEDOI: 10.12659/AJCR.932921
Am J Case Rep 2021; 22:e932921
Abstract
BACKGROUND: Allopurinol is the first-line therapy for the treatment of symptomatic hyperuricemia (gout). In clinical practice, there is a tendency to overmedicate asymptomatic patients who have elevated serum urate. Because of this practice, serious and life-threatening reactions such as Stevens-Johnson syndrome (SJS) or the more dramatic toxic epidermal necrolysis (TEN), both frequently caused by uricostatics, may occur. To increase awareness of these complications, we present a case with fulminant TEN caused by allopurinol.
CASE REPORT: A 75-year-old woman noticed a mildly itching skin rash accompanied by fever, shivering, and weakness approximately 3 weeks after taking newly prescribed allopurinol. The initial clinical examination revealed a generalized maculopapular exanthema. An adverse drug reaction was recognized, and allopurinol was discontinued. Ambulatory supportive therapy using prednisolone and cetirizine was started but failed. The patient developed a progressive exanthema with painful widespread blistering, skin peeling, and mucosal and conjunctival lesions. After recurrent presentations to the Emergency Department, the patient was transferred to our Intensive Care Unit (ICU). The clinical picture confirmed the suspected diagnosis of TEN. Massive fluid replacement, prednisolone, and cyclosporine were used as anti-inflammatory therapy. Polyhexanide and octenidine were applied for local treatment. All treatment measures were guided daily by a multidisciplinary team. After 7 days in the ICU, the patient was transferred to the Dermatology Department and was discharged from the hospital 42 days later.
CONCLUSIONS: With the prescription of allopurinol, there should be awareness of potentially life-threatening complications such as SJS or TEN. Patients with SJS or TEN should be immediately transferred to an ICU with dermatological expertise and multidisciplinary therapy.
Keywords: Allopurinol, Critical Care, Intensive Care, Stevens-Johnson Syndrome, Blister, Cyclosporine, Exanthema, Female, Humans
Background
Almost 100 years ago, in 1922, Stevens-Johnson syndrome (SJS) was described for the first time. Thirty-four years later, Lyell [1] established the term toxic epidermal necrolysis (TEN) for a skin-peeling disease characterized by widespread erythema and epidermal blistering and associated with high mortality. In his report of 4 cases, he assumed an adverse event due to medication. SJS and TEN are now known to refer to the same underlying disease, with an overlapping stage defined by the extent of affected dermal surface [2]. In the ensuing decades, a clear correlation between TEN and an increasing number of medications has been found and progress in therapy has occurred [3]. However, there is still an unacceptably high mortality rate of up to 45% [4]. Allopurinol is one of the possible TEN-inducing drugs [3], but it is often prescribed to treat asymptomatic hyperuricemia without necessary caution. Due to this usage, TEN remains a highly dangerous and serious adverse event, despite its rare occurrence of about 2 cases per 1 million people [5].
Case Report
A 75-year-old woman was admitted to the Emergency Department of our tertiary university hospital with a mildly itching skin rash for 2 days, fever, shivering, and weakness. The rash was initially noticed on the torso, and it spread out to the extremities. Self-medication with cetirizine led to no improvement. There were no signs of respiratory or gastrointestinal disorders. A SARS-CoV-2 polymerase chain reaction test was negative. A chest X-ray did not show any signs of pulmonary infiltrates. According to her past medical history, the patient was prescribed allopurinol for hyperuricemia about 3 weeks earlier. Apart from long-term antihypertensive drugs (amlodipine, valsartan), no other medication had been ingested.
On presentation, the patient showed a maculopapulous exanthema covering the whole body, which was particularly prominent on the back, abdomen, and between the breasts. Oral lesions were not apparent. From a dermatological point of view, the skin lesions appeared to be caused by a viral infection. However, an adverse drug reaction induced by allopurinol was taken into account as well, and allopurinol was discontinued immediately. Furthermore, oral prednisolone (0.9 mg/kg/d for 2 days) and cetirizine (10 mg/d) were prescribed as anti-inflammatory, immunosuppressive, and symptomatic therapy. This treatment was carried out on an outpatient basis as the patient declined hospitalization. However, despite the treatment, the patient developed a worsening exanthema, leading to hospital admission 2 days later. At this time, the maculopapulous exanthema presented as a highly painful widespread blistering and skin peeling covering more than 85% of the body surface (Figure 1A, 1B). Oral involvement was also present, with mucosal ulceration and erythema of the conjunctiva (Figure 2). The fulminant progression of the clinical picture confirmed the suspected diagnosis of TEN, most likely caused by allopurinol. After dermatological consultation, prednisolone intravenously (3.7 mg/kg/d for 3 days) and cyclosporine (5 mg/kg/d for 10 days) were started. A skin biopsy for histopathological examination showed typical signs of TEN (Figure 3). Considering the high mortality rate of TEN, the patient was immediately transferred to our Intensive Care Unit (ICU). The clinical course was reviewed daily by a multidisciplinary supportive team including clinical experts from several specialties (dermatology, ophthalmology, gynecology, urology, otorhinolaryngology, and anesthesiology). The severity of TEN was assessed according to the severity-of-illness score for toxic epidermal necrolysis (SCORTEN), a scale of 7 prognosis-relevant items calculated on admission and daily thereafter to monitor therapeutic success. The initial SCORTEN predicted hospital mortality of 90% and showed a gradual decrease after 3 days in the ICU (Table 1, Figure 4). We guided our therapy according to the current UK guidelines for the management of SJS and TEN in adults [6]. The main goal of therapy was sufficient fluid replacement to prevent end-organ hypoperfusion and shock. The requirement of fluid was adjusted daily based on urine output, serum lactate, and clinical signs of pulmonary edema. A conservative approach was taken for skin management. Large blisters were decompressed to avoid pressure-induced enlargement. Warm sterile lotions of polyhexanide and octenidine were used for daily wound cleaning. Afterward, the lesions were covered with a nonadhesive gauze, antiseptic gel, and sterile compresses. Metalline foil was used to prevent shearing forces to the skin. Oral lesions were rinsed several times per day with saline and antiseptic lotions to manage mucositis. Despite intensive analgesia including opiates, the patient experienced intense pain. Only patient-controlled analgesia ensured adequate comfort at rest. As oral nutrition was precluded by oral involvement, enteral nutrition was achieved by nasogastric feeding. Prophylactic anticoagulation was applied as recommended by the UK guidelines for TEN. Urine samples showed bacteriuria, which was treated by antibiotics. Ophthalmologic consultations confirmed rapid recovery using anti-infective and lubricating eye drops including dexamethasone and ofloxacin. Gynecological consultations revealed signs of involvement with superficial necrolysis at the labia minora and introitus, without evidence of a superinfection. Further clinical signs and laboratory findings did not confirm other organ involvement such as hepatitis or thyroiditis.
Due to multidisciplinary supportive care, the patient’s skin lesions and general condition gradually improved. Laboratory findings showed regression of renal failure and inflammatory markers. After 7 days in the ICU, the patient was transferred to the Dermatology Department. After 42 days in the hospital, the patient recovered completely and was discharged. The adverse drug reaction was documented in the patient’s notes and communicated to her general practitioner.
Discussion
TEN, first described in 1956 by Lyell [1], is a serious and life-threatening immunological disease, most often caused by an adverse reaction to various medications [1,3,7–9]. It is one of the most severe dermatological emergency conditions and represents the worst manifestation of epidermal necrolysis within a disease complex including SJS, TEN, and an overlapping syndrome between them that depends on the extent of affected dermal surface [2,10]. The literature indicates that TEN has a mortality rate of about 30% up to 45%, with a rare incidence of 1 to 2 per 1 million per year [4,5,11,12]. In addition to the dermal symptoms, various other manifestations of TEN, such as inflammation of internal gastrointestinal or respiratory mucosa, anemia, or neutropenia, cause a mortality rate that is as high as that associated with metabolic disorders, sepsis, and bleeding [13]. In most cases, intensive care and invasive interventions are necessary. Potentially causative drugs for TEN include certain antibiotics, anticonvulsives, nonsteroidal anti-inflammatory drugs, and allopurinol [3,11,12].
The clinical onset of TEN is often seen a few days to several weeks after intake of the inducing drug, and it begins with nonspecific symptoms and signs, such as fatigue, fever, pruritus, and mucosal irritation [14,15]. Late occurrence of dark erythematous to purple-colored patches, such as atypical target-oid lesions, blisters, erosions, or ulcers with a positive Nikolsky sign, is reported in the literature [8,16]. Concomitant oral, ocular, genital, or rectal mucosal effects are typical for TEN [16]. The blistering separates the epidermis from the dermis and detachment from the dermis develops. In our case, the initial symptoms were a mild pruritic rash on the torso 3 weeks after the first ingestion of allopurinol. The initial maculopapulous exanthema then worsened to a blistering, skin-peeling condition affecting almost 85% of the body surface as the condition developed.
Clinically, TEN has to be differentiated from other dermatoses, which is difficult, especially in the early phase. Viral or bacterial exanthema, autoimmune diseases, and drug-induced cutaneous reactions are potential differential diagnoses for TEN [17]. In our case, a viral exanthema or adverse drug reaction was initially included in the differential diagnosis and required ruling out.
One of the most important severe differential diagnoses is a drug reaction with eosinophilia and systemic symptoms (DRESS), a maculopapular or morbilliform drug reaction similar to that associated with allopurinol but primarily affecting the trunk, face, and extremities [18]. Along with dermal effects, fever, facial edema, lymphadenopathy, eosinophilia, and further laboratory abnormalities of various organ systems (hepatitis, nephritis, pneumonitis, carditis, and thyroiditis) may occur as a reactivation of a human herpes group virus. The incidence is estimated to be 10 per million, with a mortality rate of up to 10%, so DRESS occurs more often than TEN but is much less deadly.
Histopathological analysis of skin biopsies is necessary for the differentiation and diagnosis of TEN. With TEN, full-thickness epidermal necrolysis can be seen, whereas DRESS shows a non-specific picture with spongiotic or necrotic keratinocytes [8,18]. Necrolysis in TEN is due to a T-cell-mediated cytotoxic apoptosis and release of mediators and cytokines such as granulolysin, perforin, granzyme B, tumor necrosis factor-α, interferon-γ, or FAS-receptor-ligand by a T-cell type IV-like reaction of the body [8,19,20]. Granulolysin seems to be the leading cytokine [21]. Several HLA alleles and variants have been found to be associated with drug-induced TEN. The HLA–B*58: 01 allele is related to the occurrence of TEN induced by allopurinol. In summary, a combination of the drug structure, drug metabolism, and genetic factors are relevant in the development of TEN [8]. The pathophysiology of DRESS is not yet understood, but an autoimmune genesis due to cross-reactive T cells from viral antigens is suspected. Certain HLA alleles determine the occurrence and severity of DRESS [18,22,23].
Prognostic assessment of TEN can be achieved based on the SCORTEN (Table 2), with the most predictive value at day 3 of hospitalization [24,25]. Depending on the distribution of parameters, there is a wide range of mortality from 3.2% to 90% [24]. Table 1 shows the development of the SCORTEN in our patient, with a calculated mortality of 90%, 12%, and 12% on days 1, 3, and 5, respectively. In contrast, diagnosis of DRESS takes place through fulfilling the RegiSCAR criteria [26] or the modified criteria of Shiohara et al [27].
The best treatment for TEN remains unclear because data based on randomized controlled trials are lacking [14] owing to the rareness of the disease. Predominantly symptomatic therapy and supportive measures in the ICU are recommended, such as prevention of temperature loss, administering volume and albumin, sufficient analgosedation, renal replacement therapy as far as needed, antibiotic medication, and topical therapy with polyhexanide [8]. Treatment in our case initially adhered to the Parkland formula, and further fluid administration was guided by the specific individual needs and the continuous evaluation of clinical parameters of fluid balance to prevent organ failure and ensure adequate oxygen delivery to all organs. Moreover, we had to initiate patient-controlled analgesia to achieve pain relief. Withdrawal of the causative agent is elementary and was of course the first step and initiated at the first outpatient presentation [14]. There is no benefit in survival or re-epithelization between different dermatological therapeutic approaches such as debridement versus “anti-shear wound care” [28]. Furthermore, the involvement of the mucosal tissue has to be kept in mind when catheter insertion (eg, urogenital catheterization) is necessary [14,29]. Early interdisciplinary involvement from multiple fields (eg, ophthalmology, dermatology, plastic surgery, urology, gynecology) is recommended [8]. In our case, a daily interdisciplinary ward round was instituted. Rapid treatment through early admission to special burn centers is associated with a beneficial outcome. Complications are most often infections caused by large dermal defects and wounds, which progress to sepsis in about 20% of TEN patients [29]. Therapeutic principles in DRESS are quite similar to TEN, with discontinuation of the causative drug, supportive measures, and topical or systemic anti-inflammatory medications such as corticosteroids or calcineurin inhibitors [18].
In addition to the basic treatment, corticosteroids and intravenous immunoglobulin represent more specific and targeted therapies for TEN. Analysis of registry data did not show a clear survival benefit associated with either. Only high-dose steroids lowered mortality in different case series and the EuroSCAR registry, whereas analysis of the RegiSCAR registry showed no influence on mortality in TEN [6,14]. In contrast, cyclosporine, infliximab, and etanercept were found to lower SCORTEN-predicted mortality, but this outcome has only been noted in case reports or case series so far [6,8,14]. Thalidomide plays no role in treatment because of higher mortality rates, and therapeutic plasma exchange had no clear benefit on mortality [8]. Bearing this in mind, we chose to combine prednisolone and cyclosporine, which finally led to a good recovery.
Conclusions
TEN is a rare immunological condition that is associated with an adverse drug reaction caused by an everyday medication, which in some cases is prescribed prematurely. It is a life-threatening condition, with a mortality rate of 30% to 45%, despite therapy. Unfortunately, with exception of the British guidelines of 2016, there are no current European guidelines and no randomized controlled trials to back up the management. Moreover, recommendations on therapy are not based on randomized controlled trials. In our view, diagnosis and treatment should be based on a multidisciplinary approach. Therefore, we recommend the early transfer of possible TEN patients to a tertiary medical center that has all required disciplines. Progression of mild dermal symptoms and signs of TEN could be prevented by training as well as a more restrictive prescribing behavior in causative medications. Especially for asymptomatic mild hyperuricemia without existing or supposed gout, prescription of allopurinol should be evaluated critically.
Figures
References:
1.. Lyell A, Toxic epidermal necrolysis: An eruption resembling scalding of the skin: Br J Dermatol, 1956; 68(11); 355-61
2.. Bastuji-Garin S, Rzany B, Clinical classification of cases of toxic epidermal necrolysis, Stevens-Johnson syndrome, and erythema multiforme: Arch Dermatol, 1993; 129(1); 92-96
3.. Mockenhaupt M, Viboud C, Dunant A, Stevens-Johnson syndrome and toxic epidermal necrolysis: Assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study: J Invest Dermatol, 2008; 128(1); 35-44
4.. Sekula P, Dunant A, Mockenhaupt M, Comprehensive survival analysis of a cohort of patients with Stevens-Johnson syndrome and toxic epidermal necrolysis: J Invest Dermatol, 2013; 133(5); 1197-204
5.. Rzany B, Mockenhaupt M, Baur S, Epidemiology of erythema exsudativum multiforme majus, Stevens-Johnson syndrome, and toxic epidermal necrolysis in Germany (1990–1992): Structure and results of a population-based registry: J Clin Epidemiol, 1996; 49(7); 769-73
6.. Creamer D, Walsh SA, Dziewulski P, U.K. guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in adults 2016: Br J Dermatol, 2016; 174(6); 1194-227
7.. Kinoshita Y, Saeki H, A review of the pathogenesis of toxic epidermal necrolysis: J Nippon Med Sch, 2016; 83(6); 216-22
8.. Schneider JA, Cohen PR, Stevens-Johnson syndrome and toxic epidermal necrolysis: A concise review with a comprehensive summary of therapeutic interventions emphasizing supportive measures: Adv Ther, 2017; 34(6); 1235-44
9.. Roujeau JC, Huynh TN, Bracq C, Genetic susceptibility to toxic epidermal necrolysis: Arch Dermatol, 1987; 123(9); 1171-73
10.. Roujeau JC, Stevens-Johnson syndrome and toxic epidermal necrolysis are severity variants of the same disease which differs from erythema multi-forme: J Dermatol, 1997; 24(11); 726-29
11.. Mockenhaupt M, Stevens-Johnson syndrome and toxic epidermal necrolysis: Clinical patterns, diagnostic considerations, etiology, and therapeutic management: Semin Cutan Med Surg, 2014; 33(1); 10-16
12.. Heng YK, Lee HY, Roujeau JC, Epidermal necrolysis: 60 years of errors and advances: Br J Dermatol, 2015; 173(5); 1250-54
13.. Pereira FA, Mudgil AV, Rosmarin DM, Toxic epidermal necrolysis: J Am Acad Dermatol, 2007; 56(2); 181-200
14.. Harris V, Jackson C, Cooper A, Review of toxic epidermal necrolysis: Int J Mol Sci, 2016; 17(12); 2135
15.. Somkrua R, Eickman EE, Saokaew S, Association of HLA–B*5801 allele and allopurinol-induced Stevens-Johnson syndrome and toxic epidermal necrolysis: A systematic review and meta-analysis: BMC Med Genet, 2011; 12; 118
16.. Chung WH, Wang CW, Dao RL, Severe cutaneous adverse drug reactions: J Dermatol, 2016; 43(7); 758-66
17.. Zakrzewski JL, Lentini G, Such U, Toxic epidermal necrolysis: differential diagnosis of an epidermolytic dermopathy in a hematopoietic stem cell transplant recipient: Bone Marrow Transplant, 2002; 30(5); 331-33
18.. Cardones AR, Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome: Clin Dermatol, 2020; 38(6); 702-11
19.. Wang F, Ma Z, Wu X, Liu L, Allopurinol-induced toxic epidermal necrolysis featuring almost 60% skin detachment: Medicine (Baltimore), 2019; 98(25); e16078
20.. Lee HY, Pang SM, Thamotharampillai T, Allopurinol-induced Stevens-Johnson syndrome and toxic epidermal necrolysis: J Am Acad Dermatol, 2008; 59(2); 352-53
21.. Chung WH, Hung SI, Yang JY, Granulysin is a key mediator for disseminated keratinocyte death in Stevens-Johnson syndrome and toxic epidermal necrolysis: Nat Med, 2008; 14(12); 1343-50
22.. Cho YT, Yang CW, Chu CY, Drug reaction with eosinophilia and systemic symptoms (DRESS): An interplay among drugs, viruses, and immune system: Int J Mol Sci, 2017; 18(6); 1243
23.. Aihara M, Pharmacogenetics of cutaneous adverse drug reactions: J Dermatol, 2011; 38(3); 246-54
24.. Bastuji-Garin S, Fouchard N, Bertocchi M, SCORTEN: A severity-of-illness score for toxic epidermal necrolysis: J Invest Dermatol, 2000; 115(2); 149-53
25.. Guégan S, Bastuji-Garin S, Poszepczynska-Guigné E, Performance of the SCORTEN during the first five days of hospitalization to predict the prognosis of epidermal necrolysis: J Invest Dermatol, 2006; 126(2); 272-76
26.. Chen YC, Cho YT, Chang CY, Chu CY, Drug reaction with eosinophilia and systemic symptoms: A drug-induced hypersensitivity syndrome with variable clinical features: Dermatologica Sinica, 2013; 31(4); 196-204
27.. Shiohara T, Iijima M, Ikezawa Z, Hashimoto K, The diagnosis of a DRESS syndrome has been sufficiently established on the basis of typical clinical features and viral reactivations: Br J Dermatol, 2007; 156(5); 1083-84
28.. Dorafshar AH, Dickie SR, Cohn AB, Antishear therapy for toxic epidermal necrolysis: an alternative treatment approach: Plast Reconstr Surg, 2008; 122(1); 154-60
29.. Rajaratnam R, Mann C, Balasubramaniam P, Toxic epidermal necrolysis: Retrospective analysis of 21 consecutive cases managed at a tertiary centre: Clin Exp Dermatol, 2010; 35(8); 853-62
Figures
Tables
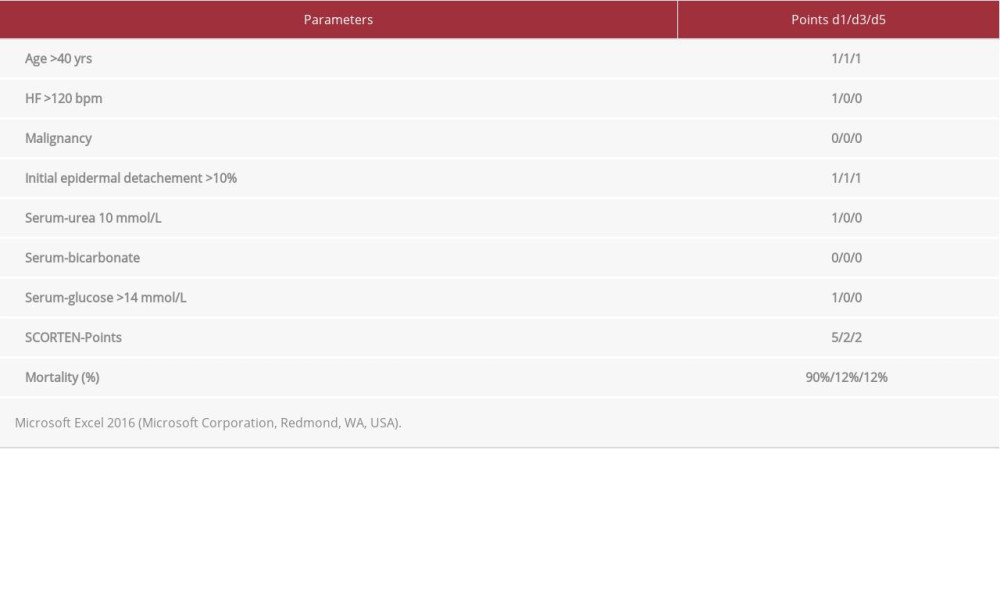 Table 1.. Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN) at Day 1, 3, and 5.
Table 1.. Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN) at Day 1, 3, and 5.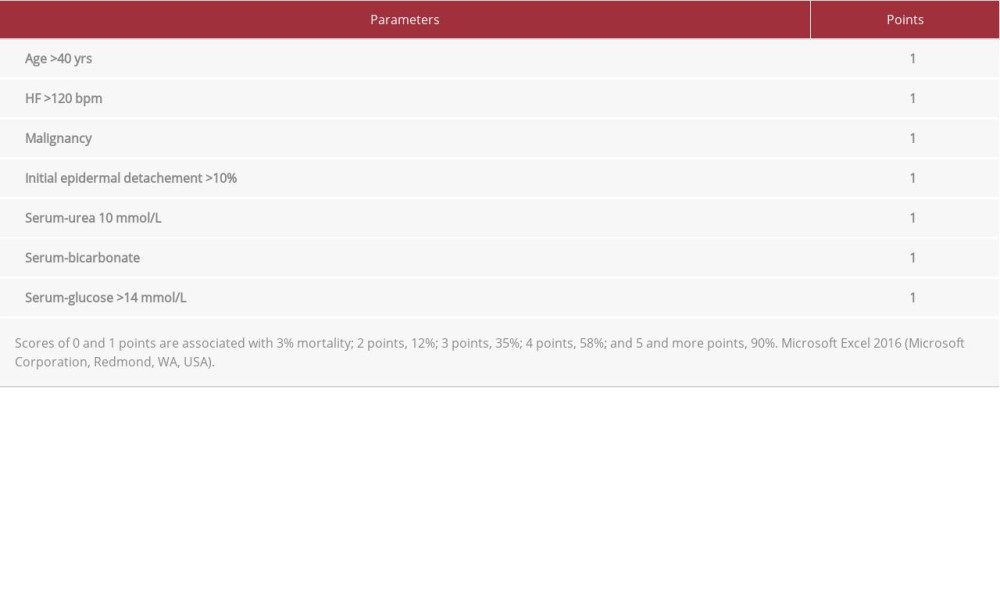 Table 2.. Predicted mortality based on the Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN).
Table 2.. Predicted mortality based on the Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN). Table 1.. Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN) at Day 1, 3, and 5.
Table 1.. Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN) at Day 1, 3, and 5. Table 2.. Predicted mortality based on the Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN).
Table 2.. Predicted mortality based on the Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN). In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
18 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943803
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250








