22 October 2021: Articles 
Vulvar Extramammary Paget Disease Detected by Cytology for Cervical Cancer Screening: A Case Report and Literature Review
Challenging differential diagnosis, Rare disease
Kotaro Takahashi1ACDEF*, Akira Kikuchi1BDEF, Ayano Horiuchi2DEF, Ryo Tamura1DEF, Kazufumi Haino3DEF, Masayuki Yamaguchi1DEF, Takashi Kawasaki4BDEFDOI: 10.12659/AJCR.933655
Am J Case Rep 2021; 22:e933655
Abstract
BACKGROUND: Vulvar extramammary Paget disease (EMPD) with abnormal cervical cytology is extremely rare. We encountered a case of secondary EMPD derived from urothelial carcinoma diagnosed after cytological examination for cervical cancer screening. We diagnosed the case promptly owing to suspicion based on the patient’s medical history and vulvar appearance. We report the case and present a review of published cases of EMPD with abnormal cervical cytology.
CASE REPORT: A 77-year-old Japanese woman visited a hospital because cervical cancer screening raised the suspicion of adenocarcinoma. Findings of the cytological examinations of the cervix, endometrium, and urethral meatus corresponded to those of other malignant neoplasms of the Bethesda system. The patient had undergone total urethral cystectomy for urothelial carcinoma 5 years earlier. In our hospital, we found erythema extending from the urethral meatus to the vulva and performed a vulvar biopsy based on the suspicion of recurrence of the urothelial carcinoma. We diagnosed secondary EMPD derived from the urothelial carcinoma based on the findings of Paget cells in the epithelium and immunohistochemistry.
CONCLUSIONS: A review of all the reported cases of EMPD with abnormal cervical cytology shows that the frequency of primary lesions is high in primary EMPD and secondary EMPD derived from urothelial carcinoma. These cases demonstrated the difficulty of suspecting EMPD based on cervical cytology alone. It should be considered that the cells derived from vulvar EMPD can be observed in cervical cytology, particularly in patients with a history of primary EMPD or urothelial carcinoma and with vulvar symptoms.
Keywords: Cervix Uteri, Paget Disease, Extramammary, Vaginal Smears, Carcinoma, Transitional Cell, Early Detection of Cancer, Female, Humans, Vulva
Background
Extramammary Paget disease (EMPD) is rare, accounting for 5.7% of all vulvar malignancies according to registry data from the National Cancer Institute [1]. The disease usually involves the anogenital area and most commonly the vulva. EMPD can be classified into primary and secondary forms, with the latter representing intraepithelial spread of an underlying carcinoma arising in the urogenital or gastrointestinal tracts [2]. Additionally, primary EMPD is subdivided into type I (primary EMPD intraepithelial neoplasm), type II (intraepithelial neoplasm with invasion), and type III (manifestation of an underlying primary adenocarcinoma of a skin appendage or subcutaneous vulvar gland). Secondary EMPD is subclassified into type I (secondary to anorectal or urothelial neoplasia) and type II (Paget disease secondary to adenocarcinomas or related tumors of other sites) [1]. There are reports of vulvar primary EMPD and secondary EMPD derived from the bladder or rectum spreading to the cervix through the epithelium, but only 11 cases of vulvar EMPD with abnormal cervical cytology have been reported in the English literature [3–12]. Herein, we present a case of type I secondary EMPD derived from urothelial carcinoma diagnosed after cytological examination for cervical cancer screening. In this case, we could diagnose the case immediately by performing a vulvar biopsy because we suspected secondary EMPD based on the patient’s medical history and vulvar appearance. We also reviewed the literature on primary and secondary EMPD with abnormal cervical cytology for the purpose of clarifying their characteristics. This information will be helpful in diagnosing EMPD promptly, which will lead to appropriate treatment.
Case Report
A 77-year-old Japanese woman visited a hospital because of a suspicion of adenocarcinoma after cervical cancer screening. Cytologic specimens were obtained from the cervix, endometrium, and urethral meatus, and their findings corresponded not to adenocarcinoma but to other malignant neoplasms of the Bethesda system. The patient was referred to our hospital for further examination. She gave a past history of total urethral cystectomy for urothelial carcinoma 5 years earlier. On examination, erythema extending from the urethral meatus to the vulva was found and was accompanied by pruritus. Cervical colposcopy showed no obvious abnormal findings, and cervical curettage and endometrial biopsy revealed only atypical glandular cells with mild nuclear atypia. We performed a biopsy of the left labia minora because we suspected recurrence of urothelial carcinoma based on macroscopic vulvar findings and the patient’s medical history. On histopathological examination, many Paget cells with large nuclei, prominent nucleoli, and abundant pale cytoplasm were found in the epithelium (Figure 1A, 1B). Immunohistochemistry revealed that tumor cells were positive for cytokeratin (CK) 7, CK20, GATA3, and uroplakin II and negative for gross cystic disease fluid protein 15 (GCDFP15) (Figure 2A–2E), indicating that the tumor was a secondary EMPD derived from the urothelial carcinoma.
We retrospectively examined the cytologic features of the cervix, endometrium, and urethral meatus, and the findings were similar to those of the biopsy specimen (Figure 3A–3D). The specimen revealed clusters of atypical cells with a high nuclear/ cytoplasm ratio, irregular nuclear membranes, and prominent nucleoli. There was no feature that contradicted a diagnosis of adenocarcinoma, but adenocarcinoma could not be confirmed because no glandular structure or mucus was observed. These findings are consistent with those of urothelial carcinoma.
Thoracoabdominal computed tomography and pelvic magnetic resonance imaging showed no other findings suggestive of recurrence. Therefore, we made a diagnosis of type I secondary EMPD derived from urothelial carcinoma and referred the patient back to the hospital that had treated her for urothelial carcinoma.
Discussion
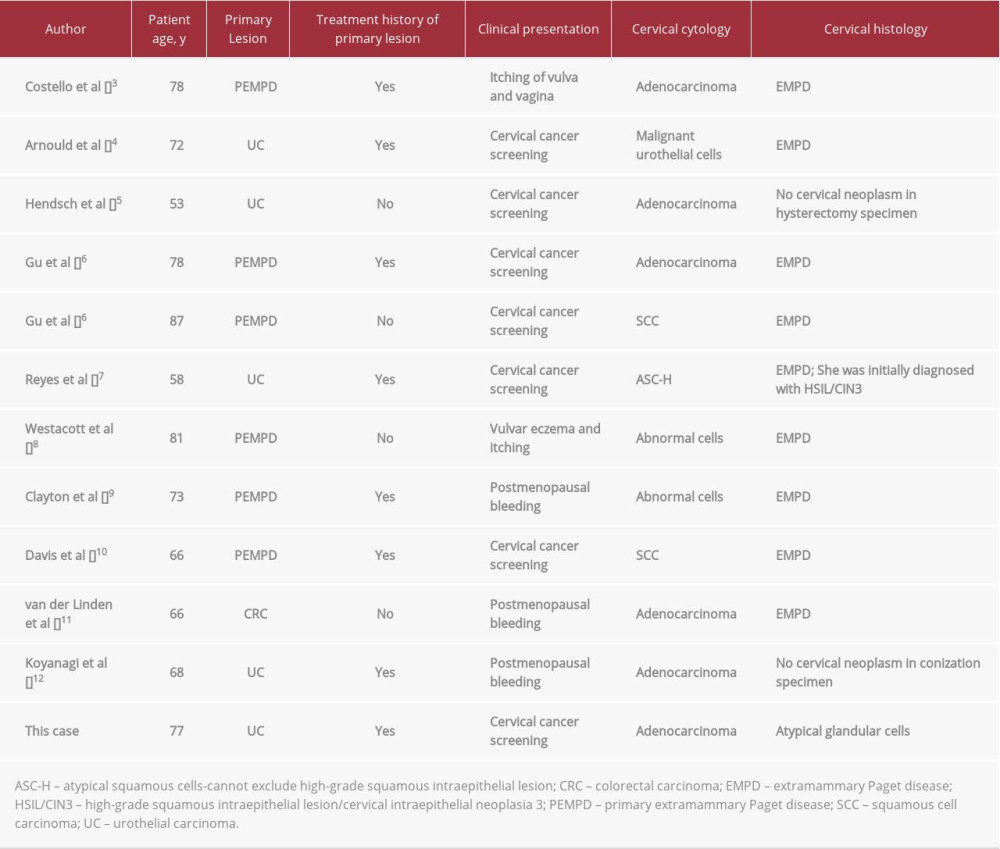
We encountered a case of secondary EMPD derived from urothelial carcinoma and were able to recognize it immediately based on the patient’s medical history and appearance of the vulva. Because it was a rare case, we conducted a literature review by searching PubMed using the terms “Paget disease, extramammary” [Mesh] AND (“cervix uteri” [Mesh] OR “vaginal smears” [Mesh] OR “uterine cervical neoplasms” [Mesh]) between 1980 and 2020. Articles eligible for the study included primary studies that reported EMPD with abnormal cervical cytology. From these, we reviewed 10 articles describing 11 cases that matched the above (Table 1) [3–12]. It should be noted that among the 12 cases, 6 were primary EMPD, 5 were secondary EMPD derived from urothelial carcinoma, and 1 was secondary EMPD derived from a rectal carcinoma. The frequency of primary EMPD and secondary EMPD derived from urothelial carcinoma was high. Here, we mainly discuss the cervical cytological findings, histological findings, and clinical presentation of our case along with previously reported cases.
Regarding the cervical cytology, there were 6 cases of adeno-carcinoma, 2 of squamous cell carcinoma (SCC), 1 with atypical squamous cells-cannot exclude high-grade squamous intraepithelial lesion (ASC-H), 2 cases with abnormal cells, and only 1 case with suspected malignant urothelial cells. In the 6 cases diagnosed as adenocarcinoma, the parameters for identification were findings such as cluster-like appearance, large cells, high nuclear/cytoplasm ratio, irregular nuclear membranes, or prominent nucleoli, and findings not positive for adenocarcinoma such as glandular structure or mucus. However, 2 cases had cytoplasmic vacuoles, which may be described as mucus [3,6]. Furthermore, in cases diagnosed as adenocarcinoma, atypical cells from EMPD seemingly mimic atypical cells from adenocarcinoma. Similarly, the 2 cases diagnosed as SCC were identified from cell atypia, as described above, and were not inferred from findings that are commonly found in SCC, such as presence of keratinization and orange G-stained cells [6,10]. In addition, in the case of the ASC-H, the authors stated that it appeared to be similar to an intraepithelial lesion, including high-grade squamous intraepithelial lesions (HSILs) because the EMPD lesion spread to the epithelium [7]. Thus, atypical cells of EMPD are frequently misdiagnosed as adenocarcinoma or SCC despite the cytological features of EMPD being different from those of adenocarcinoma or SCC. This is seemingly because the main histologic types of cervical cancer are SCC and adenocarcinoma. Therefore, it is difficult to suspect EMPD using cervical cytology alone.
In addition, there have been reports of cases in which no histo-logical lesions were found in the cervix despite abnormal cervical cytology, resulting in overtreatment. One patient underwent conization [12] and another underwent total abdominal hysterectomy [5]. These cases were finally diagnosed by biopsy of the vulva, but it took several months to reach a correct diagnosis. In addition, there was a case that was first misdiagnosed as HSIL/cervical intraepithelial neoplasia 3 and was finally diagnosed as secondary EMPD derived from urothelial carcinoma in another hospital [7].
To reach a correct diagnosis, appropriate histological analysis is necessary. The histological findings of EMPD are as follows. Although diagnosing primary and secondary EMPD using only hematoxylin and eosin staining is difficult, the following findings may be helpful. The former is characterized by Paget cells with abundant pale cytoplasm and rare mitoses, whereas biopsies may be required [1]. Therefore, it is important to suspect EMPD based on medical history and vulvar appearance. In this review, 7 of 11 patients had a history of treatment for the primary tumor, especially in the case of primary and secondary EMPD derived from urothelial carcinoma. Regarding the reasons for seeing a doctor, there were 7 patients who consulted a doctor because of abnormal cervical cancer screening results and 5 patients who presented with vulvar symptoms such as postmenopausal bleeding or vulvar itching. Most of the cases were identified during cervical cancer screening, but histological examination of the vulva was finally performed in 11 of the 12 cases, which led to the diagnosis. In 1 review article, about 90% of patients with EMPD had vulvar symptoms such as pruritus, erythematous skin patch, and focal swelling [14]. In our review, the symptoms were clearly described in only 6 cases, but considering that 11 cases had a histological examination of the vulva, they probably displayed symptoms the latter usually displays severe disarrangement of neoplastic cells with mitotic activity, often showing pleomorphism/ anaplasia [7]. Our case also showed the above characteristics of secondary EMPD with hematoxylin and eosin staining. For an appropriate diagnosis, immunohistochemistry is required in 75% of cases [1]. The following immunostaining is widely used. Primary EMPD is CK7+/CK20–/GCDFP15+ and positive for GATA3. Secondary EMPD derived from urothelial carcinoma is CK7+/CK20+/GCDFP15– and positive for GATA3 and uroplakin II or III. Secondary EMPD derived from colorectal carcinoma is CK7–/CK20+/GCDFP15− and positive for CDX2 [2,12,13]. Our patient was positive for CK7, CK20, GATA3, and uroplakin II and negative for GCDFP15; therefore, we were able to diagnose secondary EMPD derived from urothelial carcinoma.
For a representative analysis, obtaining a biopsy from a representative lesion is important. In many instances, multiple in the vulva. If patients with abnormal cervical cytology have a history of primary EMPD or urothelial carcinoma, clinicians should systematically pay attention to vulvar appearance and consider vulvar biopsy. In our case, we could diagnose the patient promptly by performing vulvar biopsy because we considered secondary EMPD based on her medical history and vulvar appearance.
Discussing the subtype of EMPD was not possible in our review because some studies did not report sufficient information to allow for recognition of the subtype. We have mainly discussed the process of diagnosis here because the prognosis differs between primary and secondary EMPD. We assume that making a prompt diagnosis is important because it can lead to appropriate treatment.
Conclusions
In conclusion, we encountered a case of secondary EMPD derived from urothelial carcinoma diagnosed after cytology for cervical cancer screening. Any abnormal cervical cytology should prompt a careful history assessment and physical examination. This process may guide clinicians toward the diagnosis of rare malignancies that can be associated with abnormal cervical cytology.
Figures
References:
1.. Wohlmuth C, Wohlmuth-Wieser I, Vulvar malignancies: An interdisciplinary perspective: J Dtsch Dermatol Ges, 2019; 17; 1257-76
2.. Konstantinova AM, Kazakov DV, Extramammary Paget disease of the vulva: Semin Diagn Pathol, 2021; 38(1); 50687
3.. Costello TJ, Wang HH, Schnitt SJ, Paget’s disease with extensive involvement of the female genital tract initially detected by cervical cytosmear: Arch Pathol Lab Med, 1988; 112; 941-44
4.. Arnould L, Chalabreysse L, Belichard C, Genital carcinoma secondary to pagetoid spread from a pagetoid urothelial carcinoma in-situ: Histopathology, 1998; 32; 575-77
5.. Hendsch SA, Glover SD, Otis CN, Donovan JT, Atypical glandular cells of undetermined significance from extramammary Paget’s of the bladder: Obstet Gynecol, 2002; 99; 912-14
6.. Gu M, Ghafari S, Lin F, Pap smears of patients with extramammary Paget’s disease of the vulva: Diagn Cytopathol, 2005; 32; 353-57
7.. Reyes MC, Park KJ, Lin O, Urothelial carcinoma involving the gyneco-logic tract: A morphologic and immunohistochemical study of 6 cases: Am J Surg Pathol, 2012; 36; 1058-65
8.. Westacott LS, Cominos D, Williams S, Primary cutaneous vulvar extramammary Paget’s disease involving the endocervix and detected by Pap smear: Pathology, 2013; 45; 426-28
9.. Clayton EF, Rubin SC, Dumoff KL, Paget cells in endometrial and endocervical curettings in a patient with recurrent vulvar Paget’s disease: Int J Surg Pathol, 2014; 22; 374-77
10.. Davis G, Anderson L, Pather S, Extramammary Paget’s disease mimicking localized malignancy on cervical cytology: Diagn Cytopathol, 2016; 44; 931-34
11.. van der Linden M, Bult P, Nagtegaal ID, Cervical metastases originating from a primary rectal adenocarcinoma due to a pagetoid spread: Hum Pathol, 2017; 68; 184-88
12.. Koyanagi Y, Kubo C, Nagata S, Detection of pagetoid urothelial intraepithelial neoplasia extending to the vagina by cervical screening cytology: A case report with renewed immunochemical summary: Diagn Pathol, 2019; 14(1); 9
13.. Primo W, Primo GRP, Basilio DB, Vulvar Paget disease secondary to high-grade urothelial carcinoma with underlying massive vascular embolization and cervical involvement: Case report of unusual presentation: Diagn Pathol, 2019; 14(1); 125
14.. Phyo AK, Mun KS, Kwan KC, Genitourinary extramammary Paget’s disease: Review and outcome in a multidisciplinary setting: Int J Clin Exp Pathol, 2020; 13; 2369-76
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133