20 February 2022: Articles 
Relapsing Infectious Mononucleosis-Like Symptoms Associated with Liver Insufficiency in a Chronic Hepatitis B Patient with Common Variable Immunodeficiency
Unusual clinical course, Challenging differential diagnosis, Unusual or unexpected effect of treatment
Akira Sato1ABCDEF*, Fumiaki Sano2CD, Hideaki Takahashi1B, Nobuyuki Matsumoto1BDOI: 10.12659/AJCR.934003
Am J Case Rep 2022; 23:e934003
Abstract
BACKGROUND: Common variable immunodeficiency (CVID) is a rare disease. Infectious mononucleosis-like symptoms due to Epstein-Barr virus reactivation in adulthood are also rare. Here, we aimed to report a case of Epstein-Barr virus reactivation presenting with relapsing infectious mononucleosis-like symptoms with liver failure in common variable immunodeficiency with chronic hepatitis B virus infection.
CASE REPORT: A 36-year-old Japanese woman with chronic hepatitis B virus infection developed relapsing fever, lymphadenopathy with marked splenomegaly, and ascites 6 months after treatment with propagermanium, a nonspecific immune modulator, and subsequent treatment with entecavir and pegylated interferon sequential therapy. Although the hepatitis B virus load was controlled, Epstein-Barr virus deoxyribose nucleic acid was detected in her serum. Seven months later, her symptoms improved following corticosteroid treatment. Prior to sequential therapy, she developed pneumonia 4 times in 2 months and exhibited consistent hypoimmunoglobulinemia before corticosteroid treatment. Further examinations showed low amounts of switched memory B cells, and absence or barely detectable levels of isohemagglutinins. Subsequently, she was diagnosed with common variable immunodeficiency.
CONCLUSIONS: Epstein-Barr virus reactivation with relapsing infectious mononucleosis-like symptoms can occur following immune modulation therapy in patients with common variable immunodeficiency, and this can affect the patient’s primary disease. Therefore, immunoglobulin screening along with the consideration of CVID in all patients is required before immune modulation therapy is planned.
Keywords: Common Variable Immunodeficiency, Epstein-Barr Virus Infections, Hepatitis B, Proxigermanium, Adult, Female, Hepatic Insufficiency, Hepatitis B, Chronic, Herpesvirus 4, Human, Humans, Infectious Mononucleosis
Background
Epstein-Barr virus (EBV) infection is extremely common worldwide, and approximately 90% of adults are infected before 30 years of age [1]. Following primary infection, EBV persists for the remainder of an individual’s life. It can reactivate under physical and psychological stress and in individuals with a variety of autoimmune diseases and cancers [2]. Moreover, reactivation sometimes results in severe or fatal outcomes [3].
Common variable immunodeficiency (CVID) is a primary immunodeficiency disease characterized by hypogammaglobulinemia, recurrent infections, and various complications, with a prevalence of 0.001–3.374 per 100 000 population, and the prevalence tends to be higher in countries with a high Human Development Index [4]. It is very rare for patients with CVID to present with relapsing infectious mononucleosis (IM)-like symptoms.
Propagermanium (PG) is an organic germanium that is used to treat chronic hepatitis B in Japan owing to its immune modulatory effects; it can reduce levels of hepatitis B e (HBe) antigen, hepatitis B virus (HBV) deoxyribose nucleic acid (DNA) polymerase, and alanine aminotransferase (ALT), as well as increase the HBe antibody titer [5]. In recent years, attempts have been made to use PG for the treatment of various diseases owing to its multiple effects, including anti-inflammatory, antitumor, and antiviral effects [6–8].
Here, we report a case of relapsing IM-like symptoms associated with liver insufficiency due to the reactivation of EBV after PG treatment in a patient with chronic HBV infection accompanied by CVID.
Case Report
A 36-year-old Japanese woman was diagnosed with chronic HBV infection through a medical examination and was followed up for 5 years. Her prior medical history revealed only a diagnosis of mild sinusitis when she was 24 years old. One year ago, her HBe antigens converted to HBe antibodies. However, her HBV DNA levels fluctuated between 4.0 and 8.1 log copies/mL, and her ALT levels fluctuated as well. Recent abdominal ultrasonography revealed chronic hepatitis with marked splenomegaly (Figure 1). Upper gastrointestinal endoscopy revealed no signs of portal hypertension. She was started on 30 mg of PG for the disease in February 2010. Laboratory data prior to PG administration are shown in Table 1. One month after the commencement of PG therapy, the patient developed pneumonia (positivity for
Discussion
Our patient presented with 2 unique clinical manifestations. The first was relapsing IM-like symptoms due to EBV reactivation. The second was CVID that became apparent after the commencement of PG treatment.
EBV infection is one of the most common infections in humans, and approximately 90% of humans acquire EBV infection by young adulthood [1]. Following infection, EBV remains latent in infected B cells in healthy individuals; however, it occasionally reactivates and sometimes causes severe illness. Even when EBV reactivation is asymptomatic, it influences the severity of a patient’s primary disease [12]. Our patient presented with relapsing IM-like symptoms with positivity for serum EBV DNA, which was suspected to be a result of chronic active EBV infection [13].
However, 2 examinations of lymph node biopsy specimens only revealed the vascular transformation of the lymph node sinuses, posing a clinical diagnostic challenge. The consistently low Ig levels seen prior to PSL administration led to the suspicion of CVID, and subsequent findings of decreased switched memory B cells and lack of isohemagglutinins were consistent with this diagnosis [14]; further, this condition was thought to be an important cause of the atypical IM-like symptoms in this case.
CVID is a primary immunodeficiency characterized by reduced serum levels of IgG, IgA, and sometimes IgM, with reduced or absent specific antibody production [14]. The prevalence of CVID in Japan is estimated to be 0.25 per 100 000 population [15]. The clinical features of CVID vary and can include acute and chronic infections, inflammatory diseases, autoimmune diseases, and increased incidence of cancer and lymphoma. There are several known atypical forms of EBV infection in patients with primary immune deficiencies [16]; however, to the best of our knowledge, reactivation of EBV in patients with CVID has not yet been reported.
The hallmark of CVID is the loss of B-cell function, but CVID is a heterogenous disease, and some patients may have additional hidden abnormalities in T cells or natural killer (NK) cells [17], which play an important role in EBV latency [18].
PG was formerly used in Japan for the treatment of chronic hepatitis B and had ameliorating effects on biochemical and virological markers. The mechanism underlying these effects is thought to be immunomodulation, including activation of cytotoxic T cells and NK cells [5]. Furthermore, PGs have also been reported to stimulate and proliferate B-cell lines [19].
In this case, we thought it was highly likely that CVID, which had been asymptomatic until then, stimulated the immune system including NK cells, T cells, and B cells via PG, which destabilized the latent state of EBV and reactivated EBV, leading to the onset and recurrence of IM-like symptoms.
With regard to chronic HBV, although the HBV DNA level was increased, exacerbation of hepatitis did not occur after discontinuation of PG. However, liver insufficiency occurred in association with relapsing IM-like symptoms, suggesting the influence of EBV reactivation. Recently, Goh et al reported that EBV reactivation in sepsis caused by pneumonia is associated with increased morbidity [12]. Additionally, Hu et al reported that EBV infection is associated with a high Child-Pugh score in patients with liver cirrhosis and that it has an influence on the liver function of patients with HBV-related acute-on-chronic liver failure [20,21]. These studies also suggest the influence of EBV reactivation on liver insufficiency.
Since PG has various immunomodulatory effects, the use of PG in patients with CVID with a latent EBV infection may lead to immune imbalance and reactivation of EBV. As PG can be used for the treatment of various diseases in the future, we recommend screening for CVID by testing immunoglobulin levels prior to PG administration.
Conclusions
We present a case of relapsing IM-like symptoms due to EBV reactivation associated with liver insufficiency after PG treatment in a patient with chronic hepatitis B, and CVID was diagnosed during the subsequent clinical course. EBV reactivation can occur in patients with CVID after immune modulation therapy, and it can affect the patient’s primary disease; therefore, immunoglobulin levels should be checked along with the consideration of CVID before administering this therapy.
Figures
Tables
Table 1.. Laboratory data before treatment with propagermanium (Feb. 2010).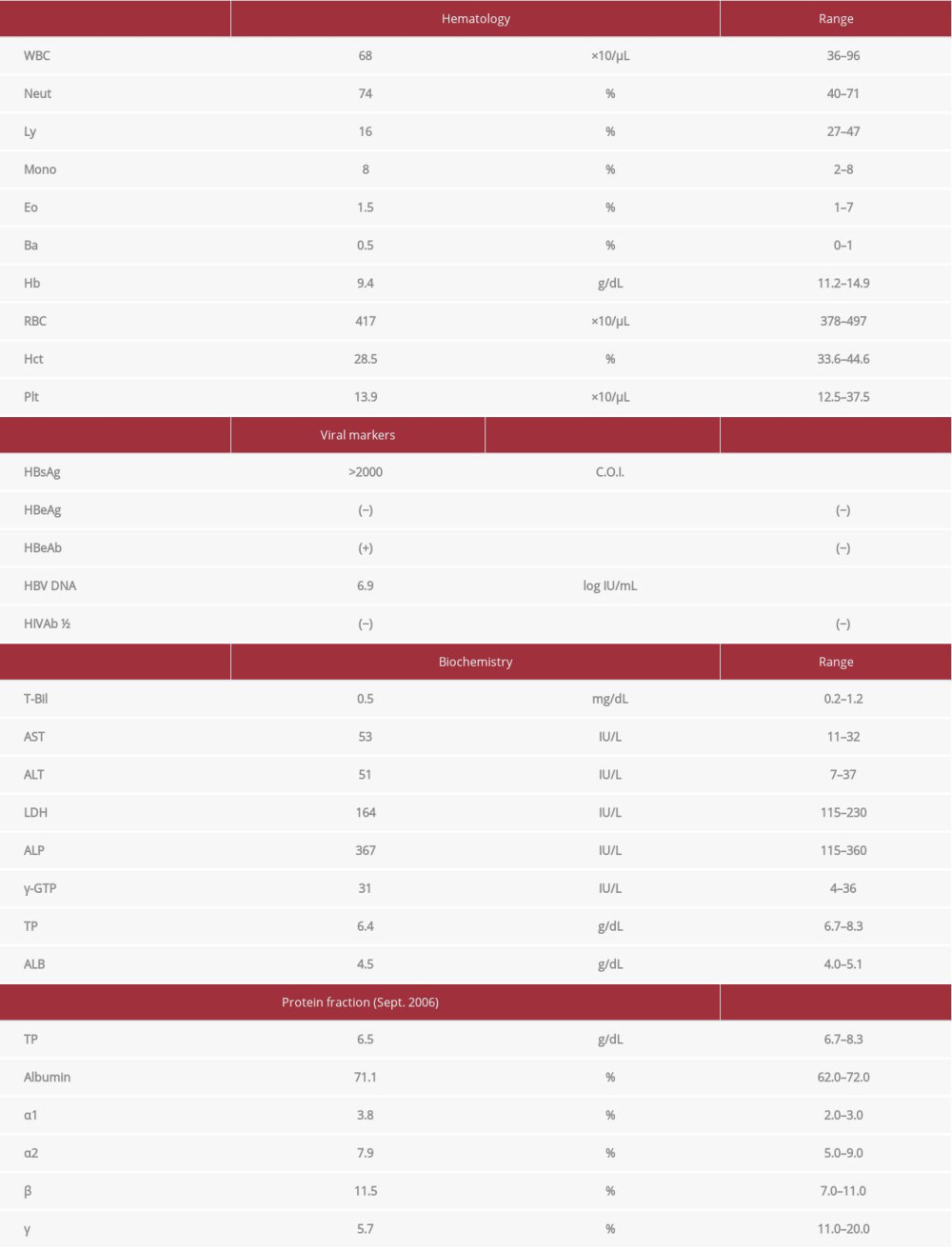 Table 2.. Viral markers before prednisolone administration (June 2012).
Table 2.. Viral markers before prednisolone administration (June 2012).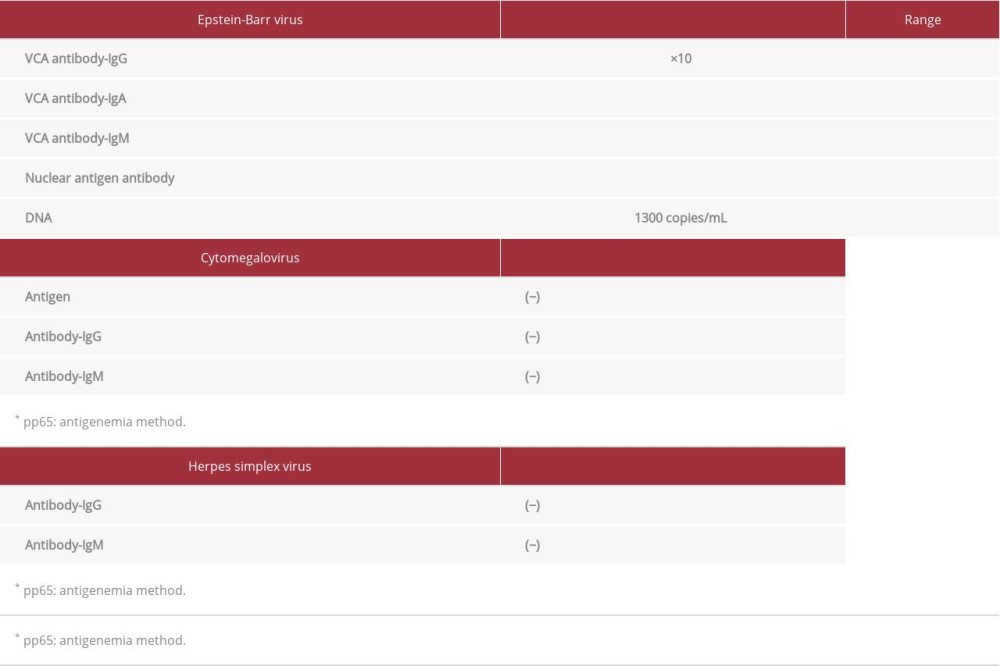 Table 3.. Immunoglobulin levels before and after prednisolone administration.
Table 3.. Immunoglobulin levels before and after prednisolone administration.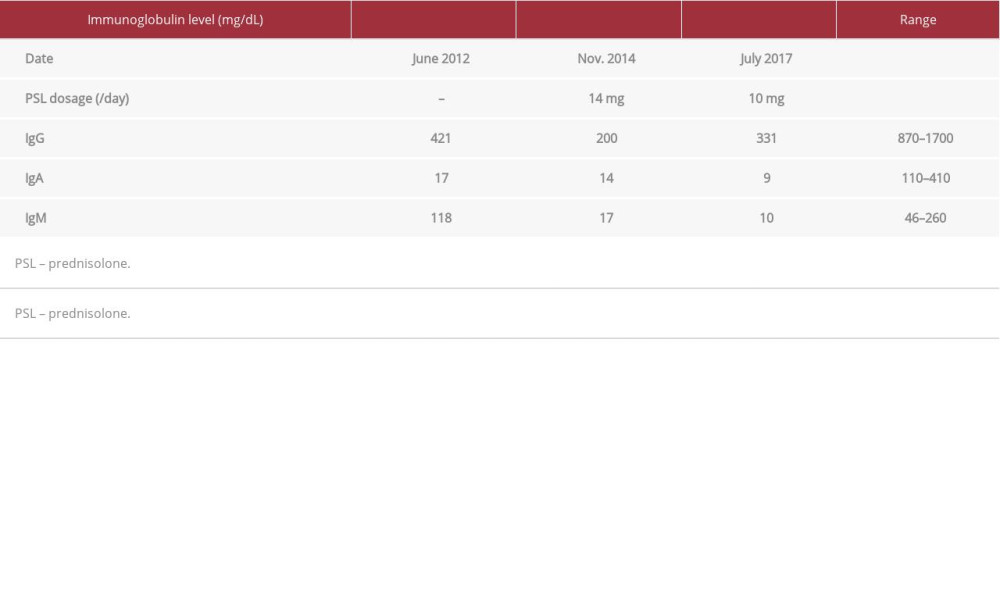 Table 4.. The titers of isohemagglutinins.(Aug. 2017, Taking prednisolone10 mg/day).
Table 4.. The titers of isohemagglutinins.(Aug. 2017, Taking prednisolone10 mg/day).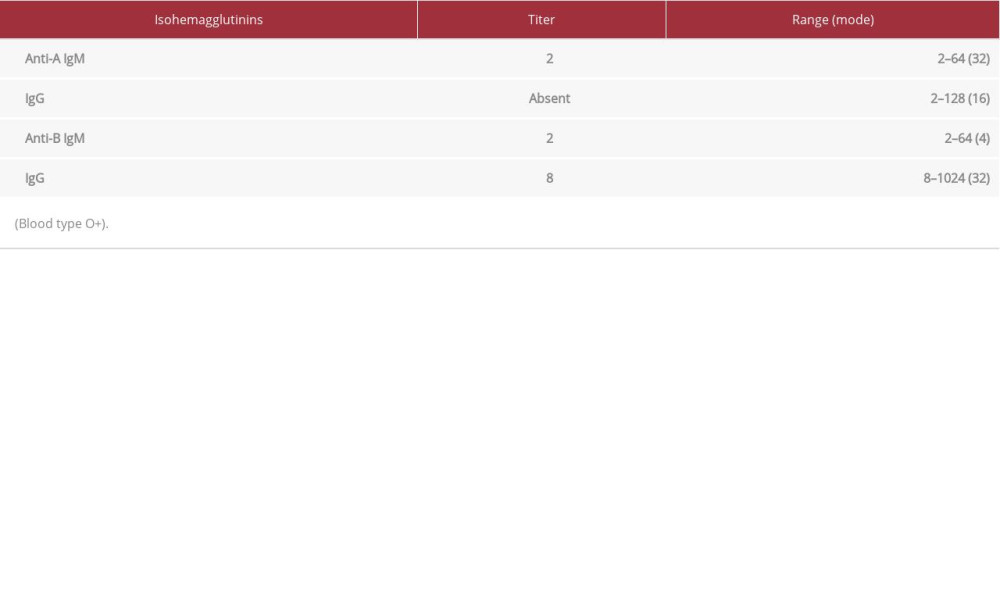 Table 5.. Lymphocyte subpopulations in peripheral blood by flowcytometry.(July 2015, Taking prednisolone 10 mg/day)
Table 5.. Lymphocyte subpopulations in peripheral blood by flowcytometry.(July 2015, Taking prednisolone 10 mg/day)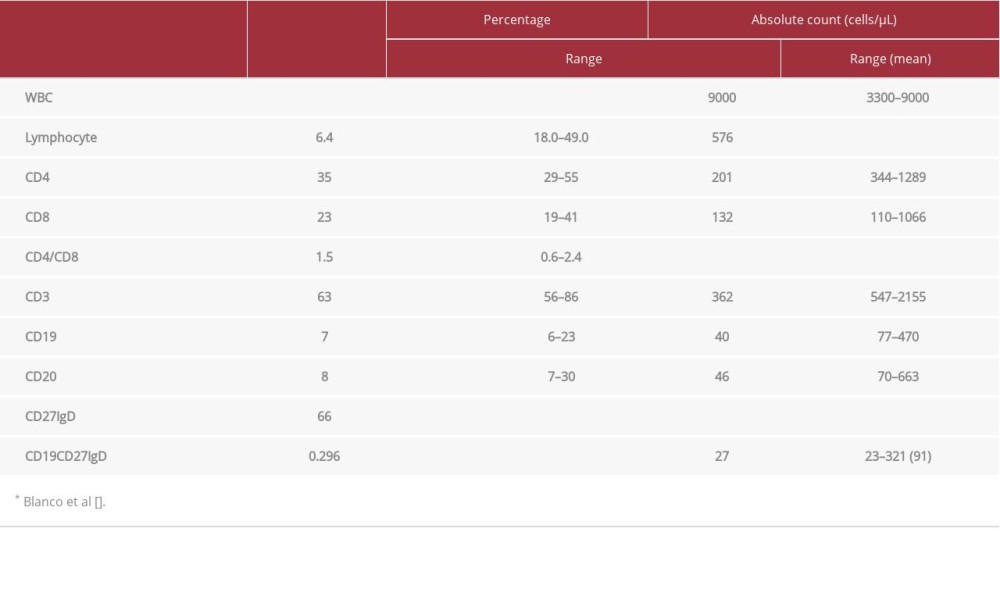
References:
1.. Dunmire SK, Hogquist KA, Balfour HH, Infectious mononucleosis: Curr Top Microbiol Immunol, 2015; 390(Pt 1); 211-40
2.. Kerr JR, Epstein-Barr virus (EBV) reactivation and therapeutic inhibitors: J Clin Pathol, 2019; 72; 651-58
3.. Takahashi T, Maruyama Y, Saitoh M, Fatal Epstein-Barr virus reactivation in an acquired aplastic anemia patient treated with rabbit antithymocyte globulin and cyclosporine: Case Rep Hematol, 2015; 2015; 926874
4.. Weifenbach N, Schneckenburger AAC, Lötters S, Global distribution of Common Variable Immunodeficiency (CVID) in the light of the UNDP Human Development Index (HDI): A Preliminary perspective of a rare disease: J Immunol Res, 2020; 2020; 8416124
5.. Hirayama C, Suzuki H, Ito M, Oda T, Propagermanium: A nonspecific immune modulator for chronic hepatitis B: J Gastroenterol, 2003; 38; 525-32
6.. Mulder P, van den Hoek AM, Kleemann R, The CCR2 inhibitor propagermanium attenuates diet-induced insulin resistance, adipose tissue inflammation and non-alcoholic steatohepatitis: PLoS One, 2017; 12; e0169740
7.. Kikuchi S, Noguchi K, Wakai K, Propagermanium induces NK cell maturation and tends to prolong overall survival of patients with refractory cancer: Anticancer Res, 2019; 39; 4687-98
8.. Ishiwata Y, Suzuki E, Yokochi S, Studies on the antiviral activity of propagermanium with immunostimulating action: Arzneimittelforschung, 1994; 44; 357-61
9.. Theise ND, Liver biopsy assessment in chronic viral hepatitis: A personal, practical approach: Mod Pathol, 2007; 20(Suppl. 1); S3-14
10.. Blanco E, Pérez-Andrés M, Arriba-Méndez S, Age-associated distribution of normal B-cell and plasma cell subsets in peripheral blood: J Allergy Clin Immunol, 2018; 141; 2208-19
11.. Kucybała I, Ciuk S, Tęczar J, Spleen enlargement assessment using computed tomography: Which coefficient correlates the strongest with the real volume of the spleen?: Abdom Radiol, 2018; 43; 2455-61
12.. Goh C, Burnham KL, Ansari A, Epstein-Barr virus reactivation in sepsis due to community acquired pneumonia is associated with increased morbidity and an immunosuppressed host transcriptomic endotype: Sci Rep, 2020; 10; 9838
13.. Accessed 25 Nov. 2021. Available from: URL: https://rarediseases.info.nih.gov/diseases/9534/chronic-active-epstein-barr-virus-infection#ref_8879
14.. Seidel MG, Kindle G, Gathmann B, The European Society for Immunodeficiencies (ESID) Registry working definitions for the clinical diagnosis of inborn errors of immunity: J Allergy Clin Immunol Pract, 2019; 7; 1763-70
15.. Ishimura M, Takada H, Doi T, Nationwide survey of patients with primary immunodeficiency diseases in Japan: J Clin Immunol, 2011; 31; 968-76
16.. Dropulic LK, Cohen JI, Severe viral infections and primary immunodeficiencies: Clin Infect Dis, 2011; 53; 897-909
17.. Yazdani R, Habibi S, Sharifi L, Common Variable Immunodeficiency: Epidemiology, pathogenesis, clinical manifestations, diagnosis, classification, and management: J Investig Allergol Clin Immunol, 2020; 30; 14-34
18.. Tangye SG, Palendira U, Edwards ES, Human immunity against EBV-lessons from the clinic: J Exp Med, 2017; 214; 269-83
19.. Cho JM, Chae J, Jeong SR, Immune activation of Bio-Germanium in a randomized, double-blind, placebo-controlled clinical trial with 130 human subjects: Therapeutic opportunities from new insights: PLoS One, 2020; 15; e0240358
20.. Hu J, Zhang X, Yu G, Epstein-Barr virus infection is associated with a higher Child-Pugh score and may predict poor prognoses for patients with liver cirrhosis: BMC Gastroenterol, 2019; 19; 94
21.. Hu J, Zhao H, Lou D, Human cytomegalovirus and Epstein-Barr virus infections, risk factors, and their influence on the liver function of patients with acute-on-chronic liver failure: BMC Infect Dis, 2018; 18; 577
Figures
Tables
 Table 1.. Laboratory data before treatment with propagermanium (Feb. 2010).
Table 1.. Laboratory data before treatment with propagermanium (Feb. 2010). Table 2.. Viral markers before prednisolone administration (June 2012).
Table 2.. Viral markers before prednisolone administration (June 2012). Table 3.. Immunoglobulin levels before and after prednisolone administration.
Table 3.. Immunoglobulin levels before and after prednisolone administration. Table 4.. The titers of isohemagglutinins.(Aug. 2017, Taking prednisolone10 mg/day).
Table 4.. The titers of isohemagglutinins.(Aug. 2017, Taking prednisolone10 mg/day). Table 5.. Lymphocyte subpopulations in peripheral blood by flowcytometry.(July 2015, Taking prednisolone 10 mg/day)
Table 5.. Lymphocyte subpopulations in peripheral blood by flowcytometry.(July 2015, Taking prednisolone 10 mg/day) Table 1.. Laboratory data before treatment with propagermanium (Feb. 2010).
Table 1.. Laboratory data before treatment with propagermanium (Feb. 2010). Table 2.. Viral markers before prednisolone administration (June 2012).
Table 2.. Viral markers before prednisolone administration (June 2012). Table 3.. Immunoglobulin levels before and after prednisolone administration.
Table 3.. Immunoglobulin levels before and after prednisolone administration. Table 4.. The titers of isohemagglutinins.(Aug. 2017, Taking prednisolone10 mg/day).
Table 4.. The titers of isohemagglutinins.(Aug. 2017, Taking prednisolone10 mg/day). Table 5.. Lymphocyte subpopulations in peripheral blood by flowcytometry.(July 2015, Taking prednisolone 10 mg/day)
Table 5.. Lymphocyte subpopulations in peripheral blood by flowcytometry.(July 2015, Taking prednisolone 10 mg/day) In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








