21 December 2021: Articles 
A Neonate with a Diagnosis of Pontocerebellar Hypoplasia Type 6 Treated with Biotin and Developed Biotin Interference with Laboratory Thyroid Function Tests
Unusual clinical course
Motomichi Nagafuji1ABCEF*, Daisuke Hitaka1EF, Atsushi Iwabuchi12DEF, Yayoi MiyazonoDOI: 10.12659/AJCR.934417
Am J Case Rep 2021; 22:e934417
Abstract
BACKGROUND: The interference of biotin administration with thyroid function tests has been reported; however, it remains unclear in clinical practice. In this report, we present the case of a neonate with a diagnosis of pontocerebellar hypoplasia type 6 (PCH6) treated with biotin who developed biotin interference with laboratory thyroid function tests.
CASE REPORT: A 1-day-old male infant with hypothermia, tachypnea, and lactic acidosis had a suspected diagnosis of mitochondrial disease. Biotin and several vitamins were administered to improve his condition. On day 14, his laboratory tests revealed a free triiodothyronine level of 4.7 pg/mL, free thyroxine level of 3.7 ng/dL, thyroid-stimulating hormone level of 0.07 μIU/mL, and thyroid-stimulating hormone receptor antibody (TRAb) level of 37.6 IU/L, suggesting Graves’ disease. No goiter or tachycardia developed. The maternal thyroid function was not measured during pregnancy, while the maternal TRAb was negative on the same day. After methimazole administration, the patient’s thyroid function normalized, and methimazole was therefore discontinued. All thyroid function tests were conducted using immunoassay methods with avidin and biotin. Later, reduced oxygen consumption under aerobic conditions in skin fibroblasts and compound heterozygous variants of the mitochondrial arginine tRNA synthetase gene were identified, and the patient was diagnosed with PCH6.
CONCLUSIONS: In this case, the clinical symptoms and physical findings were incompatible with the thyroid function. These laboratory findings could have mimicked Graves’ disease due to the biotin interference with immunoassays. Therefore, caution is required when evaluating similar cases.
Keywords: Biotin, Graves Disease, Mitochondrial Diseases, Female, Humans, Infant, Newborn, Laboratories, Male, Olivopontocerebellar Atrophies, Pregnancy, Thyroid Function Tests
Background
Neonatal hyperthyroidism is mostly associated with maternal Graves’ disease due to the transplacental passage of the maternal thyroid-stimulating hormone receptor antibody (TRAb) [1]. Other causes of neonatal hyperthyroidism include genetic defects of the thyroid-stimulating hormone (TSH) receptor. Thus, this rare disease is referred to as non-autoimmune hyperthyroidism [2]. Thyroid dysfunction complications in mitochondrial disease have not been thoroughly studied. Hypothyroidism has been reported as a complication, while autoantibody-positive hyperthyroidism has not [3]. Laboratory test result interference due to excessive biotin intake has been previously reported [4–8]. However, it remains clinically unrecognized; therefore, caution is required. We herein report the case of a neonate who received biotin for pontocerebellar hypoplasia type 6 (PCH6), one of the mitochondrial respiratory chain disorders (MRCDs), and who exhibited laboratory findings suggestive of Graves’ disease during treatment.
Case Report
A 1-day-old boy, born at 38 weeks and 5 days of gestation with a birth weight of 2704 g, was referred to our hospital after developing hypothermia and tachypnea. Metabolic acidosis associated with hyperlactic acidemia was observed, and mitochondrial disease was suspected. Biotin (5 mg/day), carnitine, and coenzyme Q10 were administered intravenously in addition to various vita-mins. On day 1, results of the thyroid function tests were within normal limits. Because his activity and feeding ability gradually declined, he was re-examined on day 14. The results of laboratory tests performed using the Cobas e801 immunoassay analyzer (Roche, Switzerland) were suggestive of Graves’ disease: free triiodothyronine (fT3) 4.7 pg/mL (normal range: 2.4–4.8 pg/mL), free thyroxine (fT4) 3.7 ng/dL (normal range: 1.4–3.4 ng/dL), TSH 0.07 μIU/mL (normal range: 1.2–3.0 μIU/mL), and TRAb 37.6 IU/L (normal range: <2.0 IU/L). All thyroid function tests, including TRAb, were conducted using electro-chemiluminescence immunoassay utilizing avidin and biotin. The patient had no goiter or tachycardia, and his thyroid echo findings were normal. The maternal thyroid function was not measured during pregnancy, and the maternal TRAb result was negative on the day of assessment. Because the patient received fresh frozen plasma on day 1, the TRAb level in the donor blood, stored in a Red Cross blood bank, was also measured to confirm a negative result. After methimazole (MMI) administration, the patient’s thyroid function normalized, and his TRAb levels decreased (Figure 1). MMI was then discontinued on day 49 because of elevated TSH levels. Biotin therapy was continued, although the patient showed normal thyroid function even after the discontinuation of MMI.
Later, the respiratory chain enzyme activity was measured in skin fibroblasts, which was found to be normal. Oxygen consumption under aerobic conditions in fibroblasts was significantly reduced, leading to the establishment of the diagnosis of MRCD. Genetic analysis for the genes associated with MRCD identified a compound heterozygous mutation in the RARS2 gene, which together with clinical features, led to the diagnosis of PCH6 in this patient.
Discussion
We report the case of a neonate with MRCD treated with biotin. During treatment, his laboratory findings suggested Graves’ disease. There are no reports of Graves’ disease in newborns with MRCD. Biotin administration possibly interfered with the laboratory tests.
Thyroid function analysis using avidin and biotin may be affected by biotin administration [9]. In competitive immunoas-says based on biotin–streptavidin interaction, excess biotin in the serum derived from supplemental biotin competed with the biotinylated components in the assays, resulting in falsely elevated values in the fT3, fT4, and TRAb assays. In contrast, the TSH assay employs a sandwich method, enabling excess biotin to displace the antigen–antibody complex, resulting in falsely decreased TSH values. This case showed that the laboratory findings could mimic those of Graves’ disease.
Wijeratne et al showed that the interference peaked at approximately 2 h after biotin ingestion and persisted for up to 24 h [5]. Therefore, according to the recommendations of the Roche Diagnostics TSH kit, patients, including adults receiving high biotin doses (ie, >5 mg/day), should have sample collection at least 8 h after the last biotin administration. In this case, biotin was administered twice a day, and it likely interfered with the thyroid function tests.
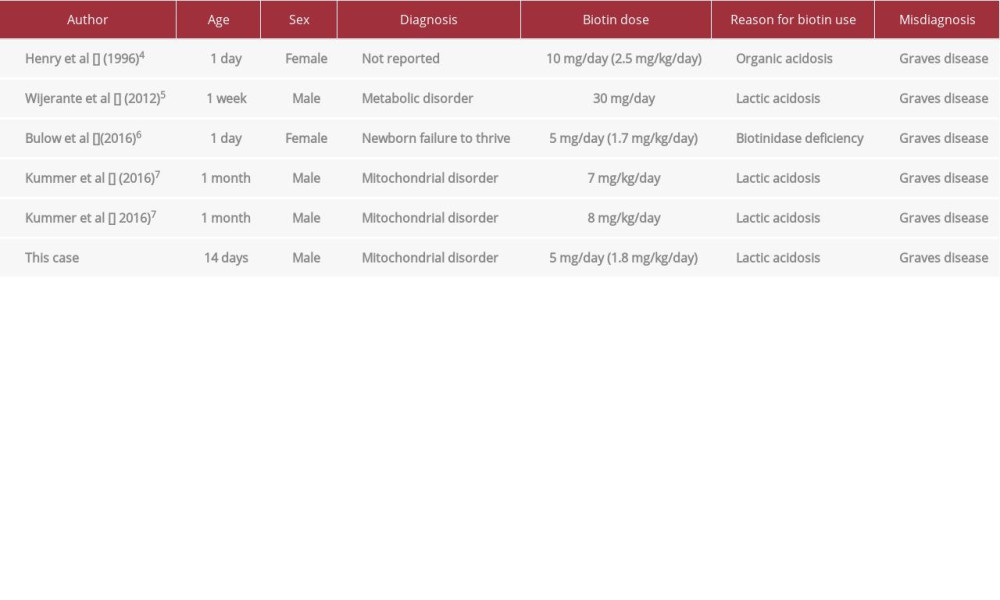
The degree of biotin interference was related to the amount of biotin administered [9]. A higher biotin concentration over the interference threshold caused a more severe interference. When methimazole administration was discontinued in our patient, he gained weight, from 2704 g to 3720 g. When the biotin dose was decreased (from 1.8 to 1.3 mg/kg/day), the test interference of biotin was reduced. Table 1 shows previous neonatal reports, wherein biotin likely interfered with the clinical laboratory test values. A similar biotin dose was administered in a previously reported neonate case, and the test values were possibly influenced by biotin [6].
In previous studies, thyroid function was evaluated using various assays and was re-evaluated after biotin discontinuation to identify false-positive results. In the present case, thyroid function was not evaluated using other assays that did not use avidin and biotin.
Serum biotin concentration, rather than biotin dose, is the major factor causing interference in thyroid function assays [10–12]. In this case, the biotin concentration was not measured because no samples were left when abnormal results in thyroid function were obtained, and no additional measurements could be performed. Therefore, a genuinely abnormal thyroid function could not be excluded. However, the patient had no other symptoms related to Graves’ disease. Thus, the clinical symptoms and physical findings were incompatible with the thyroid function. Therefore, the test results were conclusively false, and anti-thyroid drug treatment was unnecessary. Moreover, the patient did not exhibit hypothyroidism symptoms during anti-thyroid drug administration. Incorrect test results due to test interference can cause adverse events in children, and clinicians should always be aware of any interference with the test.
Currently, MRCDs that are responsive to treatment with biotin are only biotinidase deficiency and holocarboxylase synthetase deficiency. However, because the diagnosis of these 2 diseases is time consuming, biotin may be administered to newborns who have a suspected diagnosis of MRCDs in the acute phase. When biotin is administered in neonates, relatively high doses may be recommended, and clinicians need to be aware that biotin can affect laboratory values due to interference.
Conclusions
Clinicians must be aware of the possibility of biotin interference in immunoassays of patients with clinical presentations that are not consistent with the laboratory findings.
References:
1.. McKenzie JM, Zakarija M, Fetal and neonatal hyperthyroidism and hypothyroidism due to maternal TSH receptor antibodies: Thyroid, 1992; 2; 155-59
2.. Duprez L, Parma J, Van Sande J, Germline mutations in the thyrotropin receptor gene cause non-autoimmune autosomal dominant hyperthyroidism: Nat Genet, 1994; 7; 396-401
3.. Schaefer AM, Walker M, Turnbull DM, Taylor RW, Endocrine disorders in mitochondrial disease: Mol Cell Endocrinol, 2013; 379; 2-11
4.. Henry JG, Sobki S, Arafat N, Interference by biotin therapy on measurement of TSH and FT4 by enzyme immunoassay on Boehringer Mannheim ES700 analyser: Ann Clin Biochem, 1996; 33; 162-63
5.. Wijeratne NG, Doery JC, Lu ZX, Positive and negative interference in immunoassays following biotin ingestion: A pharmacokinetic study: Pathology, 2012; 44; 674-75
6.. Pedersen I, Laurberg P, Biochemical hyperthyroidism in a newborn baby caused by assay interaction from biotin intake: Eur Thyroid J, 2016; 5; 212-15
7.. Kummer S, Hernsen D, Distelmaier F, Biotin treatment mimicking Graves’ disease: N Eng J Med, 2016; 375; 704-6
8.. Li D, Radulescu A, Shrestha RT, Association of biotin ingestion with the performance of hormone and nonhormone assays in healthy adults: JAMA, 2017; 318; 1150-60
9.. Li D, Ferguson A, Cervinski MA, Lynch KL, Kyle PB, AACC guidance document on biotin interference in laboratory tests: J Appl Lab Med, 2020; 5; 575-87
10.. Paketçi A, Köse E, Gürsoy Çalan Ö, Serum level of biotin rather than the daily dosage is the main determinant of interference on thyroid function assays: Horm Res Paediatr, 2019; 92; 92-98
11.. Bowen R, Benavides R, Colón-Franco JM, Best practices in mitigating the risk of biotin interference with laboratory testing: Clin Biochem, 2019; 74; 1-11
12.. Samarasinghe S, Meah F, Singh V, Biotin interference with routine clinical immunoassays: Understand the causes and mitigate the risks: Endocr Pract, 2017; 23; 989-98
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133