27 February 2022: Articles 
Acute Hemorrhagic Leukoencephalitis – A Rare but Fatal Form of Acute Disseminated Encephalomyelitis – Complicated by Brain Herniation: A Case Report and Literature Review
Unusual clinical course, Challenging differential diagnosis, Management of emergency care, Rare disease, Rare coexistence of disease or pathology
Hamza M. Alsaid12ABCDEFG*, Mohammad A.A. Atawneh1ABCDEFG, Sadi Abukhalaf1ABCDEFG, Amro Daoud3EFG, Abdurrahman Hamadah4CDEF, Kamel Gharaibeh1ABCDEFGDOI: 10.12659/AJCR.935636
Am J Case Rep 2022; 23:e935636
Abstract
BACKGROUND: Acute hemorrhagic leukoencephalitis (AHLE) is a very rare fulminant post-infectious demyelinating disease of the CNS. We report an atypical presentation of AHLE involving unique brain areas 2 weeks following a viral upper-respiratory tract infection (URTI). Early diagnosis and proper management improve the prognosis of this disease, and AHLE can have a very poor prognosis and high mortality rate.
CASE REPORT: A 52-year-old male patient was referred for deteriorating consciousness 2 weeks after a viral URTI. An initial brain CT scan showed multiple patchy bilateral and diffuse hypodense areas including the cerebellar, occipital, parietal, and frontal lobes. The diagnostic workup also included CSF analysis and MRI of the brain, which revealed multiple areas of hemorrhagic involvement. Management included broad-spectrum antibiotics, acyclovir, mannitol, steroids, and plasmapheresis. On the fifth day of admission, brain CT showed severe diffuse edema and brain herniation. Unfortunately, despite prompt aggressive treatment measures, within 48 hours the patient died due to centrally-mediated hemodynamic instability.
CONCLUSIONS: We report a rare case of AHLE with a unique presentation and extensive unusual involvement of regions of periventricular and subcortical white matter, cerebellum, and midbrain. Early diagnosis along with appropriate management measures and intensive care can help decrease morbidity and mortality; therefore, prompt referral and high-level care should be sought for all patients who present with acute deteriorating consciousness. We hope that this report can help future studies to better characterize this rare disease and provide further guidance regarding prognosis and management.
Keywords: Encephalomyelitis, Acute Disseminated, Leukoencephalitis, Acute Hemorrhagic, Brain, Humans, Magnetic Resonance Imaging, Male, Neuroimaging
Background
Acute hemorrhagic leukoencephalitis (AHLE), also known as Hurst’s disease, is a very rare fulminant post-infectious demyelinating disease of the central nervous system (CNS), and is usually reported to follow acute respiratory illnesses or vaccinations. Frequently, AHL presents with meningismus, seizures, multiple focal neurological deficits, and impaired consciousness that rapidly progresses to coma [1]. The diagnosis is largely made clinically with suggestive cerebrospinal fluid (CSF) analysis and magnetic resonance imaging (MRI) findings.
However, the definitive diagnosis is confirmed by brain biopsy [1,2]. Although white matter of The frontal and parietal lobes are usually the affected areas of the brain, the posterior fossa structures are rarely involved [2,3]. The outcome is usually devastating, with poor prognosis, although prompt recognition and management can improve outcomes [3–6]. Herein, we report a 52-year-old male patient who presented with acute encephalopathy after 2 weeks of a viral upper-respiratory tract infection that was complicated by emerging neurological deficits with involvement of posterior fossa structures, which rapidly resulted in brain herniation and death.
Case Report
A 52-year-old previously healthy male patient was referred to our tertiary center for deteriorating consciousness and confusion. The patient was in his usual healthy state until 2 weeks prior to admission, when he developed odd behavior and altered level of consciousness following an upper-respiratory tract illness that required a 2-day stay in a periphery local hospital before his referral to our center; otherwise, the patient had a nonsignificant past medical history. At the outside hospital, the patient required intubation and mechanical ventilation for airway protection due to decreased consciousness. The initial brain computed tomography (CT) scan (Figure 1) showed multiple patchy bilateral and diffuse hypodense areas at cortical and subcortical regions of the brain, including the cerebellar, occipital, parietal, and frontal lobes, lacking contrast enhancement.
Upon his arrival to our intensive care unit, the patient was lethargic, opened eyes to pain stimuli, localized to pain, with stable vital signs, and was afebrile. A neurologic examination revealed brisk right extensor planter reflex (Babinski reflex), his pupils were reactive bilaterally with anisocoria, and the right pupil was smaller than the left. The patient’s respiratory and physical examinations were within normal limits, and no skin changes were noted. A non-traumatic lumbar puncture revealed cerebrospinal fluid (CSF) pleocytosis with white blood cell count of 250 cells/mL, of which 70% were lymphocytes, red blood cell count was 400 cells/mL, increased protein level 156 mg/dl, and a normal CSF-to-serum glucose ratio 74/151. Human immunodeficiency virus (HIV), Herpes Simplex Virus (HSV), and Epstein-Barr virus (EBV) viral titers were negative, no other viral titers were obtained, and bacterial blood cultures were obtained and were negative.
Magnetic resonance imaging (MRI) of the brain (Figure 2) revealed bilateral multiple areas of abnormal hyper-intensities with associated edema and effacement of sulci on FLAIR and T2-weighted images. Susceptibility-weighted imaging showing multiple areas of hemorrhagic components bilaterally in the involved portions of the cerebral hemispheres.
The clinical, neuroimaging, and CSF features suggested the diagnosis of AHLE. The patient was promptly managed with broad-spectrum antibiotics, including vancomycin along with acyclovir, pulse steroids (methylprednisolone 1 gram i.v. daily), and mannitol. He was also started on plasmapheresis and received 2 sessions. However, the patient’s neurological state continued to deteriorate. A brain CT scan was repeated on the hospital day 5 and showed severe diffused brain edema with brain herniation (Figure 3); 24 hours later, he became hemodynamically unstable, with severe bradycardia and hypotension, which was thought to be centrally mediated. Subsequently, upon extensive discussion with his family and all care team members, the patient’s care was transitioned to a comfort measures approach and he died within 48 hours. Brain biopsy and autopsy were not done due to family preference.
Discussion
Acute hemorrhagic leukoencephalitis was first described by Edward Weston Hurst in 1941 and is also known as Hurst’s disease. Hurst and colleagues reported 2 adult patients who developed severe focal neurological signs after respiratory illnesses [7]. AHLE is a very rare demyelinating disease mainly affecting children [5].
AHLE is usually a post-infectious disease, which might be associated with viral or bacterial infections or can occur after vaccination. Brain biopsies of patients with AHLE can show infective agents such as Herpes simplex virus, Varicella zoster virus, and Human herpesvirus-6 [8]. Other agents have also been described to be associated with AHLE, such as Epstein-Barr virus (EBV), Influenza A virus, and
The frequency of AHLE is unclear, with more than 90 cases reported, and 19 of these cases included reported MRI findings [3,7]. The clinical picture of AHLE is usually characterized by a progressive deteriorating neurological condition. Patients can have headache, mental status changes, focal neurological deficits, and fever. Less commonly, AHLE can present with psychological symptoms [11]. This clinical picture is on the contrary of acute disseminated encephalomyelitis (ADEM), which is a CNS demyelinating disease with a distinct tendency to a perivenous localization of pathological changes. ADEM tends to have a milder clinical presentation and better outcomes with less occurrence of hemorrhage on MRI. As with AHLE, the etiology of ADEM can have a temporal link with viral infections (post-infection encephalomyelitis) [12,13]. Therefore, AHL is considered the hyper-acute form of ADEM [5–7].
Other pathologic entities can be considered in the differential diagnosis: Herpes simplex encephalitis, which usually involves the limbic system, including the medial temporal lobes, insular cortices, and inferolateral frontal lobes on MRI, sparing the basal ganglia [14]; and multiple sclerosis, which can have various presentations depending on the areas affected (MRI findings in multiple sclerosis include hypo-intense or hyper-intense multifocal plaque lesions, which can be infratentorial, periventricular, within the deep white matter, juxtacortical, or mixed white matter/gray matter lesions [15]. Another differential diagnosis is CNS vasculitis, which includes a wide variety of diseases that can have a non-specific presentation. On MRI, vasculitis is characterized by multiple infarctions, usually bilateral, affecting different vascular territories of variable size, and in various stages of healing [16].
Elevated erythrocyte sedimentation rate, leukocytosis, and elevation of CSF protein and white blood cell count can also be seen [5]. Our patient’s CSF evaluation revealed pleocytosis with a white blood cell count of 250 cells/mL, of which 70% were lymphocytes, red blood cell count of 400 cells/mL, and increased protein level of 156 mg\dl. These findings are compatible with a diagnosis of AHLE.
Typical features of AHLE on radiologic workup include focal hemorrhage, diffuse demyelination, perivascular necrosis, and inflammatory meningeal infiltrates [17]. AHLE has a variable scope of magnetic resonance imaging (MRI) findings, including multifocal bilateral white matter hyper-intense lesions, hyper-intense white matter edema, and punctate hemorrhagic hypo-intense lesions on T2-weighted MRI. These lesions can be extensive and include areas of the brainstem, internal capsules, parietal lobes, and cerebellum [18]. The present case had extensive abnormal hyper-intense lesions affecting the periventricular and subcortical white matter, cerebellum, and midbrain.
Management of AHLE mainly focuses on early diagnosis and intensive care. It largely depends on immunosuppression, which is achieved by combinations of corticosteroids and cyclophosphamide. Plasmapheresis may have profound effects on improvement [3]. Measures against increased intracranial pressure should also be considered [3]. Our patient was started on mannitol, acyclovir, and methylprednisolone in addition to plasmapheresis.
The prognosis and outcome of patients suffering AHLE can be fulminant and poor, as in most patients, with death occurring an average of 6 days after symptoms onset [2]. Our patient’s fulminant neurological status unfortunately remained unchanged and, shortly after transitioning to comfort measures, the patient died.
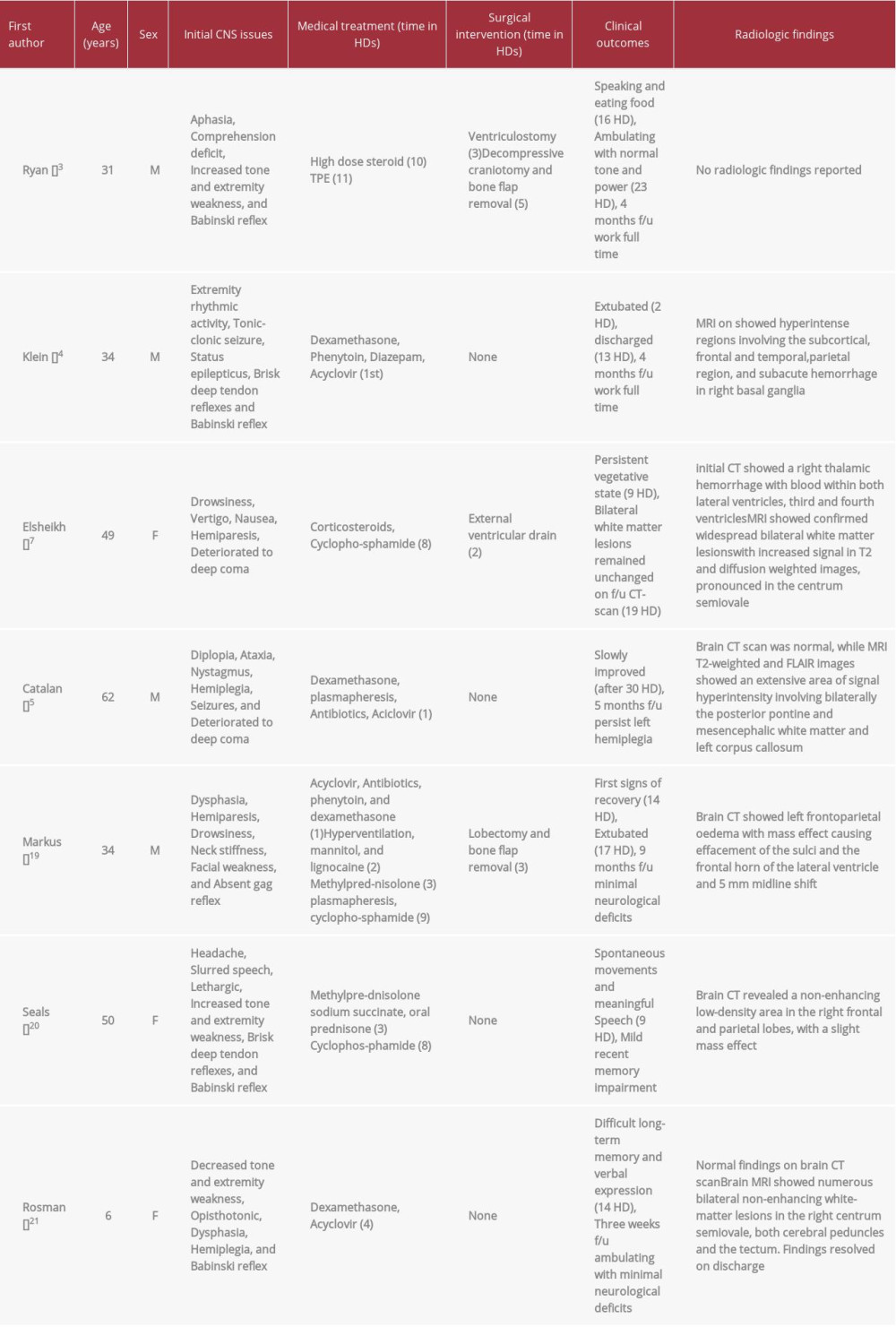
Table 1 includes 7 cases of AHLE, along with their clinical presentation, treatment, outcomes, and radiologic findings. In clinical presentation, a progressive reduced level of consciousness almost always followed by focal neurological deficits is the earliest feature of the disease. Survival of the patients relied mainly on immune suppression and lowering intracranial pressure. Immune suppression achieved by either a combination of corticosteroids, plasmapheresis, and cyclophosphamide, or corticosteroids with plasmapheresis, or even corticosteroid alone, as in a single surviving patient. Managing the increased intracranial pressure is a major component as well, and is best achieved either medically or surgically. In these surviving patients we believe that early surgical intervention against increased ICP was a key to survival. Clinical outcome is variable for the patients who survived, with some of the focal neurologic deficits persisting on follow-up [18–22].
Conclusions
AHLE should be considered in patients with acute deteriorating level of consciousness after a recent respiratory illness. AHLE typically presents with headache, mental status changes, fever and multifocal neurological deficits. The diagnosis is largely made clinically, with suggestive CSF analysis and MRI findings. Neuroimaging with MRI and CT scan allows for prompt diagnosis. Management largely depends on immunosuppression with corticosteroids and cyclophosphamide in addition to plasma-pheresis. AHLE usually has a very poor prognosis and outcome; and, based on the limited available literature, we believe the mortality and morbidity rates can be decreased by early detection and treatment with immunosuppression and plasma-pheresis; therefore, prompt referral and high-level care should be sought for all patients who present with acute deteriorating level of consciousness. There are still large gaps of knowledge about this rare disease, and more studies are needed to better characterize it and to provide further information and guidance regarding its prognosis and management.
Figures
References:
1.. Pinto P, Taipa R, Moreira B, Acute hemorrhagic leukoencephalitis with severe brainstem and spinal cord involvement: MRI features with neuro-pathological confirmation: J Magn Reson Imaging, 2011; 33(4); 957-61
2.. Vartanian TK, de la Monte S, Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 1-1999. A 53-year-old man with fever and rapid neurologic deterioration: N Engl J Med, 1999; 340(2); 127-35
3.. Ryan L, Bowman R, Zantek N, Use of therapeutic plasma exchange in the management of acute hemorrhagic leukoencephalitis: A case report and review of the literature: Transfusion, 2007; 47(6); 981-86
4.. Klein C, Wijdicks E, Earnest IV F, Full recovery after acute hemorrhagic leukoencephalitis (Hurst’s disease): J Neurol, 2000; 247(12); 977-79
5.. Catalan M, Naccarato M, Grandi F, Acute hemorrhagic leukoencephalitis with atypical features: Neurol Sci, 2009; 30(1); 55-57
6.. Friedman D, Neuroradiology case of the day. Unilateral acute hemorrhagic leukoencephalitis (AHL) complicated by venous thrombosis: Radiographics, 1998; 18(1); 246-50
7.. ElShikh S, Schumacher M, Mader I, Acute hemorrhagic leukoencephalitis (Hurst disease): A case report and review of recent literature: European Journal of Radiology Extra, 2006; 59(2); 43-47
8.. An S, Groves M, Martinian L, Detection of infectious agents in brain of patients with acute hemorrhagic leukoencephalitis: J Neurovirol, 2002; 8(5); 439-46
9.. Francisci D, Sensini A, Fratini D, Acute fatal necrotizing hemorrhagic encephalitis caused by Epstein-Barr virus in a young adult immunocompetent man: J Neurovirol, 2004; 10(6); 414-17
10.. Payne E, Rutka J, Ho T, Treatment leading to dramatic recovery in acute hemorrhagic leukoencephalitis: J Child Neurol, 2007; 22(1); 109-13
11.. Nasr J, Andriola M, Coyle P, ADEM: Literature review and case report of acute psychosis presentation: Pediatr Neurol, 2000; 22(1); 8-18
12.. Ferraro A, Roizin L, Neuropathologic variations in experimental allergic excephalomyelitis: J Neuropathol Exp Neurol, 1954; 13(1); 60-104
13.. Russell D, The nosological unity of acute haemorrhagic leucoencephalitis and acute disseminated encephalomyelitis: Brain, 1955; 78(3); 369-76
14.. Bonnici-Mallia M, Kanodia A, Rae N, Marwick C, Herpes simplex encephalitis: Unusual imaging appearances: BMJ Case Rep, 2016; 2016; bcr2016214993
15.. Nesbit G, Forbes G, Scheithauer B, Multiple sclerosis: histopathologic and MR and/or CT correlation in 37 cases at biopsy and three cases at autopsy: Radiology, 1991; 180(2); 467-74
16.. Abdel Razek A, Alvarez H, Bagg S, Imaging spectrum of CNS vasculitis: Radiographics, 2014; 34(4); 873-94
17.. Fontoura P, Mendes A, Correira M, Melo Pires M, [Weston Hurst acute haemorrhagic leukoencephalitis. Neuropathological study of one case]: Rev Neurol, 2002; 35(4); 328-31
18.. Kuperan S, Ostrow P, Landi M, Bakshi R, Acute hemorrhagic leukoencephalitis vs ADEM: FLAIR MRI and neuropathology findings: Neurology, 2003; 60(4); 721-22
19.. Markus R, Brew B, Turner J, Pell M, Successful outcome with aggressive treatment of acute haemorrhagic leukoencephalitis: J Neurol Neurosurg Psychiatry, 1997; 63(4); 551-52
20.. Seales D, Greer M, Acute hemorrhagic leukoencephalitis: A successful recovery: Arch Neurol, 1997; 48(10); 1086-88
21.. Rosman N, Gottlieb S, Bernstein C, Acute hemorrhagic leukoencephalitis: Recovery and reversal of magnetic resonance imaging findings in a child: J Child Neurol, 1997; 12(7); 448-54
22.. Hofer M, Weber A, Haffner K, Acute hemorrhagic leukoencephalitis (Hurst’s disease) linked to Epstein-Barr virus infection: Acta Neuropathol, 2004; 109(2); 226-30
Figures
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943420
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942824
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943118
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942826
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250