07 October 2022: Articles 
Demodicosis as a Skin Complication in Organ Transplant Recipients: A Case Series
Rare coexistence of disease or pathology
Adriana Marquardt-Feszler1ABCDEF*, Maria Alicja Dębska-Ślizień2CDEG, Beata Imko-Walczuk1ACDEGDOI: 10.12659/AJCR.936467
Am J Case Rep 2022; 23:e936467
Abstract
BACKGROUND: Patients after organ transplantation are, due to chronic immunosuppression, prone to have many cutaneous adverse events, both infections and neoplasms. Studies show that some groups of patients under chronic immunosuppression are prone to develop demodicosis. The significance of demodicosis in the population of organ transplant recipients has not been established yet.
CASE REPORT: We present 4 cases of patients with multiply dermatological complications of immunosuppression, in which one of them is demodicosis. The presented symptoms were itch, pustules, papules, and/or telangiectasias. Age of patients varied from 64 to 79 years old. Time between transplantation and diagnosis of demodicosis varied from 6 to 10 years. Other dermatological problems that appeared were basal cell carcinoma, actinic keratosis, and seborrheic keratosis. Patients showed complete resolution after treatment with topical ivermectin 10 mg/g and topical permethrin 50 mg/g. However, the medications were prolonged to 16 weeks in 1 case to reduce persistent papules and telangiectasias. The therapy did not cause any complications or disruptions in function of transplanted kidneys in any of reported patients.
CONCLUSIONS: Demodicosis may have a significant role in the group of infections that organ transplant recipients are prone to, and may co-exist with other dermatological diseases, including neoplasms. However, larger studies in the field are needed.
Keywords: Immunosuppression, Kidney Transplantation, Aged, Humans, Ivermectin, Middle Aged, Organ Transplantation, Permethrin, Skin Neoplasms, Telangiectasis
Background
Long-term immunosuppression, which is mandatory among organ transplant recipients (OTRs), is a strong risk factor for skin complications such as infections and neoplasms [1]. The occurrence of non-melanoma cancers is much higher than in the general population and increases with time after transplantation [2,3]. Viral infections such as verrucae or herpes simplex are more common, as are bacterial and parasitic infections [4].
Demodex mites (DM) are the most common ectoparasites of humans [5].
There can be distinguished 2 clinical variants of demodicosis [5]. Primary demodicosis refers to patients who have no other inflammatory dermatoses and is associated with pityriasis folliculorum, nodulocystic demodicosis, blepharitis, peri-oral dermatitis, and auricular demodicosis. Secondary demodicosis concerns the presence of DM in patients with other skin or systemic diseases. Symptomatic demodicosis may be observed as acne rosacea, pityriasis folliculorum, papulopustular eruptions, perioral dermatitis, and blepharitis. Secondary demodicosis is more frequently found among immunosup-pressed patients. The literature contains studies evaluating the prevalence of DM in groups of patients under long-term immunosuppression for reasons other than solid organ transplantation. However, there is still only limited information and no strong conclusions are drawn about the significance of demodicosis as an infection that OTRs are prone to.
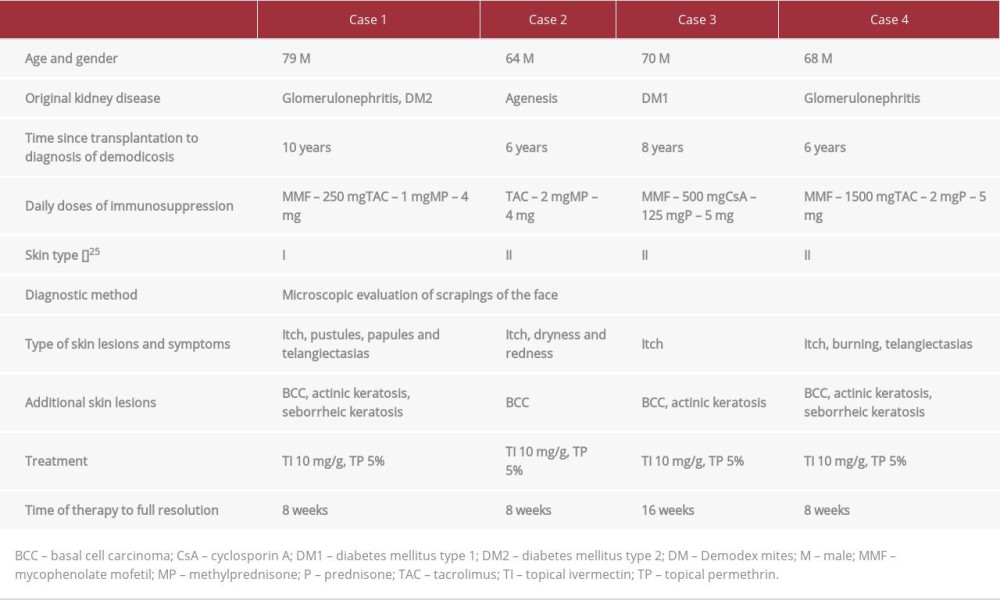
We report 4 cases of patients after kidney transplantation with skin complications of immunosuppression, in which 1 of them is demodicosis (Table 1).
Case Reports
CASE 1:
A 79-year-old man who underwent renal transplantation in 2009, with a history of glomerulonephritis and type 2 diabetes, was referred to our dermatology clinic 9 years after transplantation because of actinic keratosis and seborrheic keratosis. His immunosuppression regimen consisted of mycophenolate mofetil (MMF), tacrolimus (TAC), and methylprednisolone (MP). The patient had no medical history of skin problems before transplantation. In the first visit, actinic keratosis and seborrheic keratosis were treated with cryotherapy. During control 1 year later, several face pustules, papules, and telangiectasias were observed (Figure 1). The patient reported strong itching of the face. Microscopic evaluation of scrapings of the face revealed numerous DM. One month of treatment with topical ivermectin 10 mg/g and topical permethrin 50 mg/g led to improvement of skin lesions and reduction of itch of the face. The therapy was continued for another month. At the same time, dermoscopic evaluation of a little erosion localized on the left temple raised a suspicion of a basal cell carcinoma (BCC). Two series of cryosurgery were performed and 4 weeks of therapy with fluorouracil 50 mg/g cream was recommended afterwards, with complete recovery. Consecutive follow-ups did not show recurrence of demodicosis or BCC.
CASE 2:
A 64-year-old man who underwent kidney transplantation in 2015 was referred to our dermatology clinic because of suspicion of skin cancer on the nose. The patient has been diagnosed incidentally in 1990 with left kidney agenesis. In 2003, a progressive loss of function of the kidney began to be observed. Two years of hemodialysis before transplantation were necessary. His immunosuppression regimen consisted of TAC and MP. In the first visit 6 years after transplantation, BCC was diagnosed and 2 series of cryosurgery were performed with consecutive topical imiquimod 50 mg/g therapy for 4 weeks. In addition, he reported having dryness, redness, and itching of the face skin. No papules or pustules were observed. Microscopic evaluation of scrapings of the face revealed numerous DM. After 1 month of treatment with topical ivermectin 10 mg/g and topical permethrin 50 mg/g, a control Demodex spp. test was negative. Ivermectin 10 mg/g therapy was continued for another 4 weeks. The BCC was healed with no recurrence in follow-ups.
CASE 3:
A 70-year-old man who underwent kidney transplantation in 2009, with a history of end-stage renal disease (ESRD) secondary to type 1 diabetes mellitus, was referred to our dermatological clinic 8 years after transplantation. His immunosuppression therapy consisted of MMF, cyclosporine A (CsA), and prednisone (P). The reason for referral was suspicion of skin cancer in the medial angle of the left eye, and dermoscopic evaluation suggested BCC. He has been directed for a surgical removal in the Plastic Surgery Department. The histopathological examination of the skin tumor revealed BCC nodulare polyposum. After a few months, another BCC was confirmed in dermoscopy and was treated with cryosurgery and fluorouracil 50 mg/g for 1 month. Due to his concern about itching of the face, he was reffered to a Demodex test, which was positive. In the examination, no papules, pustules, or telangiectasias were observed. Topical ivermectin 10 mg/g and topical permethrin 50 mg/g were prescribed. After 2 months of therapy, a control test still showed presence of Demodex spp. After another 2 months of prolonged therapy, clinical improvement of the skin condition was noticed and itching of the face was significantly reduced. The BCC in the temple healed completely.
CASE 4:
A 63-year-old man who underwent kidney transplantation in 2014, with a history of glomerulonephritis, was referred to our dermatological clinic 1 year after transplantation for a routine dermatological follow-up. The immunosuppressive treatment consisted of MMF, TAC, and P. During annual examinations, actinic keratosis and seborrheic warts were treated with cryo-therapy. After 5 years of dermatological controls (6 years after transplantation), the patient reported itching and a burning feeling on the face. There were telangiectasias on skin of both cheeks (Figure 2). Microscopic evaluation of scrapings of the face revealed numerous DM. Eight weeks of treatment with topical ivermectin 10 mg/g and topical permethrin 50 mg/g resulted in disappearance of symptoms. During control 2 years later, a BCC was suspected in dermoscopy on the skin of a forearm. A photodynamic therapy preceded by photosensitizing with 5-aminolevulinic acid was planned. No recurrence of demodicosis was observed.
Discussion
Long-term immunosuppression causes multiple skin complications among OTRs [1]. Infucino et al found three-quarters of patients in the group of 812 OTRs had at least 1 cutaneous complication [12]. Gulec et al compared the prevalence of fungal infections in a group of 102 OTRs and 88 controls [13]. As the result, 65 (63.7%) patients in the group of OTRs and only 27 (30.7%) of controls were diagnosed with fungal infection [13]. Imko-Walczuk et al reported that the frequency of benign lesions in OTRs is dictated by length and dosage of immunosuppression. The authors showed that 60% out of 223 OTRs had skin fungal infection, whereas the other most frequent lesions were iatrogenic cutaneous effects such as purpura (50.2%) and xerosis (41.2%), and gingival hyperplasia as an adverse effect of cyclosporin A was noticed in 28.2% of patients [4].
Demodicosis has an unclear role in the group of skin complications after organ transplantation. Occurrence of the disease among immunosuppressed patients has been discussed only in a few studies. We aimed to find available literature to present studies on this issue. The search was conducted on 3–10 January 2022. Search terms included (demodicosis OR Demodex) AND (immunosuppression OR transplant OR transplantation). PubMed was the main search engine used. We excluded studies concerning animals. A few of articles were excluded or included only based on abstracts due to language other than English or Polish. Additional articles were identified through reference search of the articles acquired via PubMed. The selection process resulted in a total of 6 original works and 1 case report. Aydingöz et al studied DM among 30 OTRs and did not find any correlation [14]. Although the authors repeated the study 4 years later with a different method for taking skin samples, there was still no correlation found between immuno-suppression in OTRs and occurrence of DM [15]. However, the second attempt of the study consisted only of 12 patients. No more studies were conducted among more numerous groups of patients after organ transplantation. In 2016, Chovatiya and Colegio presented 4 cases of OTRs with demodicosis, suggesting that DM as a disease secondary to solid organ transplantation might be more common than previously thought [16]. The problem of demodicosis among immunocompromised patients for reasons other than organ transplantation is under discussion. Keles et al checked the density of DM among 45 patients before onset of immunosuppression (patients with psoriasis vulgaris, pemphigus vulgaris, alopecia areata, or lichen planus) and after 1 and 3 months of therapy [17]; 3 patients were diagnosed with demodicosis. The density of DM was also higher than in the control group. Dursun et al retrospectively evaluated the files of 42 patients with DLE [18]; 50% of patients were Demodex-positive. The authors suggested that DM test can be a useful routine procedure for the evaluation of DLE patients. However, there are also studies that deny the role of DM among immunosuppressed patients. Roihu et al studied the prevalence of DM in a few groups of patients, one of which consisted of patients immunosuppressed because of discoid lupus erythematosus (DLE). Talghini et al compared associations between patients with DM and those with nonmelanoma skin cancers (NMSC), DLE, and rosacea. Neither the Roihus study nor the Talghinis study showed any correlation of immunosuppression with DM [19,20].
All of the presented patients were also diagnosed with BCC. The correlation between BCC and DM is not clear. During this research, terms used on PubMed included: (demodicosis OR Demodex) AND (BCC OR NMSC OR basal cell carcinoma OR non-melanoma skin cancer). As the result, 2 studies were included. Erbagci et al showed higher prevalence and density of DM in BCC [21]. Nevertheless, the authors suggested that although DM may be a triggering factor for carcinogenesis due to its irritating effect or chronic inflammation, the higher density of DM is more likely to be secondary to, for example, higher vascularization in the BCC. Talghinis et al found no correlation between BCC and DM [20].
Diagnostic methods of DM are a topic of discussion. In 1993, Forton et al recommended SSSB as a useful tool in diagnosis of demodicosis when the density of DM is more than 5/cm2 [22]. Then, in 2017, Forton et al suggested that 2 SSSBs from the same area of the skin can increase the sensitivity of the method to 98.7% [23]. Hyun Yun et al showed that DME is also more sensitive than SSSB [24]. Evaluation of scrapings of the chosen area of the face, despite being a cheap and easy to perform method, has the limitation of low sensitivity, so a negative result does not exclude infestation of DM [11]. All of the patients were diagnosed in the same laboratory using the method of evaluation of scrapings of the changed skin of the face (Figure 3).
In the presented cases, time between transplantation and diagnosis of demodicosis varied from 6 to 10 years. However, all of the patients were referred to our clinic for reasons other than demodicosis, and one of them was diagnosed with the disease during the first visit, so the onset of the disease is unclear. Itching appears to be characteristic of patients with rosacea, telangiectasias, and other demodicosis-like lesions of the face. Appearance of pruritus of the face may be a strong factor when considering DM in OTRs. One of the patients had Fitzpatrick skin type I, whereas 3 had skin type II [25]. It is not proven that DM occurs more often among patients with skin types I and II. However, higher risk of photo-damage and blood vessels located more superficially might be a factor for stronger immunological reaction to the presence of DM [26].
Treatment of DM can consist of topical and/or oral medications. It seems that treatment of DM among OTRs does not differ from that of the general population. In our cases, topical treatment with ivermectin 10 mg/g and topical permethrin 50 mg/g was enough to cause improvement of skin lesions and reduce itching of the face. However, the medications were prolonged to 16 weeks in one case to reduce persistent papules and telangiectasias. Ivermectin, a semi-synthetic derivative of avermectin, is used in treatment of demodicosis because it has both anti-inflammatory and acaricidal activity [27,28]. Topical permethrin has a proven effectiveness on erythema and papules in rosacea [29]. The two medications can be safely combined in the therapy of demodicosis. No complications of the therapy were observed or reported. No worsening of kidney parameters was noticed at the time of therapy.
Conclusions
To sum up, the presented cases show OTRs with multiply dermatological lesions, in which one of them was DM. The role of DM as a disease secondary to immunosuppression is still under discussion and larger studies may be crucial. Because patients rarely report symptoms of DM, it may be clinically important to test OTRs for the disease, especially when patients confirm itching of the face.
Figures
References:
1.. Naldi L, Venturuzzo A, Invernizzi P, Dermatological complications after solid organ transplantation: Clin Rev Allergy Immunol, 2018; 54(1); 185-212
2.. Mittal A, Colegio OR, Skin cancers in organ transplant recipients: Am J Transplant, 2017; 17(10); 2509-30
3.. Berg D, Otley CC, Skin cancer in organ transplant recipients: Epidemiology, pathogenesis, and management: J Am Acad Dermatol, 2002; 47(1); 1-17 , quiz 18–20
4.. Imko-Walczuk B, Okuniewska A, Prędota-Głowacka A, Benign cutaneous disease among polish renal transplant recipients: Transplant Proc, 2016; 48(5); 1660-66
5.. Chen W, Plewig G, Human demodicosis: Revisit and a proposed classification: Br J Dermatol, 2014; 170(6); 1219-25
6.. Rufli T, Mumcuoglu Y: Dermatologica, 1981; 162(1); 1-11
7.. Elston CA, Elston DM, Demodex mites: Clin Dermatol, 2014; 32(6); 739-43
8.. Skonieczna D, Sławianowska P, Wesołowski R, Szewczyk-Golec K, Roztocze z rodzaju Demodex – charakterystyka, chorobotwórczość, diagnostyka: Diagn Lab, 2018; 54(1); 37-44 [in Polish]
9.. Huang HP, Hsu CK, Lee JYY, A new superficial needle-scraping method for assessing Demodex density in papulopustular rosacea: J Cosmet Dermatol, 2020; 19(4); 896-900
10.. Segal R, Mimouni D, Feuerman H, Dermoscopy as a diagnostic tool in demodicidosis: Int J Dermatol, 2010; 49(9); 1018-23
11.. Kubanov AA, Yulia G, Grevtseva A, Important aspects of Demodex diagnostics: J Surg Dermatology, 2016; 1(1); 43-51
12.. Infusino SD, Loi C, Ravaioli GM, Cutaneous complications of immunosuppression in 812 transplant recipients: A 40-year single center experience: G Ital Dermatol Venereol, 2021; 155(5); 662-68
13.. Güleç AT, Demirbilek M, Seçkin D, Superficial fungal infections in 102 renal transplant recipients: A case-control study: J Am Acad Dermatol, 2003; 49(2); 187-92
14.. Aydingöz IE, Mansur T, Dervent B, Demodex folliculorum in renal transplant patients: Dermatology, 1997; 195; 232-34
15.. Aydingöz IE, Dervent B, Demodex folliculorum in renal transplant patients revisited: Dermatology, 2001; 203; 272-73
16.. Chovatiya RJ, Colegio OR, Demodicosis in renal transplant recipients: Am J Transplant, 2016; 16(2); 712-16
17.. Keles H, Yuksel EP, Aydin F, Senturk N, Pre-treatment and post-treatment demodex densities in patients under immunosuppressive treatments: Med, 2020; 56(3); 107
18.. Dursun R, Durmaz K, Oltulu P, Ataseven A, Demodex positive discoid lupus erythematosus: Is it a separate entity or an overlap syndrome?: Dermatol Ther, 2020; 33(3); 1-7
19.. Roihu T, Kariniemi AL, Demodex mites in acne rosacea: J Cutan Pathol, 1998; 25(10); 550-52
20.. Talghini S, Fouladi DF, Babaeinejad S, Demodex mite, rosacea and skin melanoma; Coincidence or association?: Turkiye Parazitol Derg, 2015; 39(1); 41-46
21.. Erbagci Z, Erbagci I, Erkiliç S, High incidence of demodicidosis in eyelid basal cell carcinomas: Int J Dermatol, 2003; 42(7); 567-71
22.. Forton F, Seys B: Br J Dermatol, 1993; 128(6); 650-59
23.. Forton FMN, De Maertelaer V, Two consecutive standardized skin surface biopsies: An improved sampling method to evaluate demodex density as a diagnostic tool for rosacea and demodicosis: Acta Derm Venereol, 2017; 97(2); 242-48
24.. Lee JR, Chul H, Yun J, Demodex mite density determinations by standardized skin surface biopsy and direct microscopic examination and their relations with clinical types and distribution patterns: Ann Dermatol, 2017; 29(2); 137-42
25.. Fitzpatrick TB, The validity and practicality of sun-reactive skin types I through VI: Arch Dermatol, 1988; 124(6); 869-71
26.. Sarkar R, Podder I, Jagadeesan S, Rosacea in skin of color: A comprehensive review: Indian J Dermatol Venereol Leprol, 2020; 86(6); 611-21
27.. Ebbelaar CCF, Venema AW, Van Dijk MR, Topical ivermectin in the treatment of papulopustular rosacea: A systematic review of evidence and clinical guideline recommendations: Dermatol Ther (Heidelb) Sep, 2018; 8(3); 379-87
28.. Benkali K, Rony F, Graeber M, Clinical pharmacokinetics profile of ivermectin 1% cream after dermal applications on the face: World J Dermatol, 2016; 5(1); 57-64
29.. Raoufinejad K, Mansouri P, Rajabi M, Efficacy and safety of permethrin 5% topical gel vs. placebo for rosacea: A double-blind randomized controlled clinical trial: J Eur Acad Dermatology Venereol, 2016; 30(12); 2105-17
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133