02 February 2023: Articles 
A Case of Unicentric Castleman Disease with Concomitant Myasthenia Gravis and Persistent Left Superior Vena Cava
Challenging differential diagnosis, Rare coexistence of disease or pathology
Maxwell Cardwell1ABCDEF*, Marvin S. Lu2ABCDEF, Joshua Melamed23ABCDEF, John Astle24ABCDEF, Mario Gasparri25ABCDEF, David W. Johnstone26ABCDEFDOI: 10.12659/AJCR.938305
Am J Case Rep 2023; 24:e938305
Abstract
BACKGROUND: Castleman disease was first described in 1956 as mediastinal masses composed of benign lymphoid hyperplasia with germinal center formation and capillary proliferation closely resembling thymomas. It has been linked with many multi-system disorders, including myasthenia gravis. Cases of Castleman disease with corresponding myasthenia gravis have higher rates of postoperative myasthenic crisis, which are reported as high as 37.5%. We encountered a case of Castleman disease with myasthenia gravis that was discovered early and managed successfully with complete surgical resection and no postoperative myasthenic crisis.
CASE REPORT: A 25-year-old woman with an uncomplicated history presented with shortness of breath, numbness in hands, tiring with chewing, and fatigue. Myasthenia gravis was diagnosed with serology test results, and a 7.5×7.0-cm mediastinal mass was discovered in addition to the incidental finding of a persistent left superior vena cava, closely abutting the mass. Biopsy showed lymphoid proliferation, regressed germinal centers surrounded by small lymphocytes, and vascular proliferation, consistent with unicentric Castleman disease, hyaline-vascular type. The patient was successfully treated for Castleman disease with myasthenia gravis, and no postoperative myasthenic crisis occurred.
CONCLUSIONS: Castleman disease associated with myasthenia gravis can dramatically increase the risk of postoperative myasthenic crisis. Our literature review of all 16 cases of Castleman disease with myasthenia gravis since 1973 revealed that 18.75% of cases were associated with a postoperative myasthenic crisis. This association elicits the importance of prompt diagnosis of myasthenia gravis when evaluating mediastinal masses and the value of having neurology and anesthesiology staff aware of the increased risk of crisis.
Keywords: Castleman disease, Myasthenia Gravis, Female, Humans, Adult, persistent left superior vena cava, Vena Cava, Superior, Thymus Neoplasms
Background
Castleman disease was first described in 1956 by Dr. Benjamin Castleman in a case report of 13 instances of masses resembling thymomas, macroscopically and microscopically. These masses were most commonly found in the mediastinum and were histologically composed of benign lymphoid hyperplasia with germinal center formation and marked capillary proliferation, somewhat resembling a thymoma [1]. In 1972, these lesions were further histologically classified as plasma-cell, hyaline-vascular, or mixed types [2]. These lesions can also be classified as unicentric or multicentric. Recent studies have delineated an association between Castleman disease and other multi-system disorders, including myasthenia gravis [3–7]. While myasthenia gravis is rarely associated with Castleman disease, recent studies have found that postoperative myasthenic crisis occurs much more frequently in patients with both Castleman disease and myasthenia gravis than in patients with thymic epithelial tumors [8,9]. We report a case of unicentric mediastinal Castleman disease with myasthenia gravis and persistent left superior vena cava (SVC), which was resected with a left thoracic robotic approach.
Case Report
A 25-year-old woman with a history of asthma and lichen planus presented with shortness of breath, numbness of her hands, tiring with chewing, slurred speech, and extreme fatigue. The initial workup included a chest X-ray, which showed an enlarged left mediastinal mass, and computed tomography angiography imaging, which showed a homogenous 7.5×7.0-cm mediastinal mass with no apparent invasion of vascular structures and a preserved left SVC straddling the lateral side of the mass (see Figure 1). There was an absence of the innominate vein. Positron emission tomography demonstrated a maximum uptake of 3.1 at the known mass and no ectopic disease foci. A laboratory workup confirmed myasthenia gravis, with acetylcholine receptor binding antibodies of 4.92. The initial needle biopsy of the mass showed findings including immature T-cell precursors favoring a thymic lesion but was non-diagnostic and raised the possibility of T-lymphoblastic lymphoma. The patient was referred to our Cardiothoracic Surgery Department, where it was decided to repeat biopsies prior to robotic resection. A left robotic biopsy showed a non-diagnostic infiltrate of mature T cells and B cells, with no cytologic atypia. In the interim, she was hospitalized in the Intensive Care Unit for what was initially thought to be a myasthenic crisis. She improved after 5 days of plasmapheresis; however, the Neurology Department concluded that this episode of hypoxic respiratory failure was due to asthma exacerbation compounded by pneumonia. She was discharged on a steroid taper and antibiotics and had no recurrent episodes. Her surgery was further delayed, with a kidney stone requiring surgical removal and stent placement.
Due to the presence of a persistent left SVC straddling the lateral side of the mass, we considered the potential need for vascular control and pericardial access. We also considered a sternotomy versus a left transthoracic robotic approach. We ultimately decided to proceed with a left robotic approach.
In the operating room, the patient was positioned supine, and a bump was placed under her left back to expose the lateral left chest. An 8-mm camera port was placed at the halfway point between the xiphoid and the sternal notch along the midaxillary line. The right-hand port was placed at the second interspace and the left-hand port through the fifth interspace. The mass was carefully dissected free of the lung. The left SVC was visualized and retracted away to facilitate the dissection and optimize visualization. The mass was well encapsulated, extending from the pericardium to the aortic arch, subclavian artery, vagus nerve, and aortopulmonary window and was posterior to the left SVC. The mass was closely abutting the left SVC, which raised concerns for vascular connections between the two. The mass was carefully dissected free of the surrounding structures, with no large arteriovenous connections found. The 9.3×6.5×3.4-cm mass (see Figure 2) was removed intact through the assistant’s port in the lower inter-costal space. The procedure was successful, with no injury to the phrenic or vagus nerves or major vessels. The patient’s postoperative course was uneventful.
Histologic examination of the resection specimen demonstrated a prominent lymphoid proliferation, regressed germinal centers, including some with penetrating blood vessels, surrounded by mantle zones of small lymphocytes arranged in a concentric (onion-skin) pattern, vascular proliferation, and variable hyalinized vessels consistent with Castleman disease, hyaline vascular variant, with increased TdT-positive cells (see Figure 3). The diagnosis of Castleman disease was confirmed by pathologists from the National Institutes of Health.
Discussion
Castleman disease is a rare disorder characterized by lymph node enlargement and increased numbers of lymph node follicles with germinal center involution and vascular proliferation [5]. It was originally described by Dr. Benjamin Castleman in the 1950s and has since been further classified by histological analysis into plasma-cell, hyaline-vascular, and mixed-cell variants and by location as unicentric or multicentric [1,2]. The incidence in the United States is estimated to be 5940 cases per year of unicentric Castleman disease and 1756 cases per year of multicentric Castleman disease [10], although other studies have reported a lower incidence [11]. It is important to delineate unicentric Castleman disease and multi-centric Castleman disease, as unicentric Castleman disease is indolent and treated via surgical resection alone, while multi-centric Castleman disease can have significant morbidity and mortality and usually requires medical therapy. Over the years, Castleman disease has been found to be linked to pheochromocytomas [4,5], paraneoplastic pemphigus, Hodgkin disease, POEMS syndrome, and, rarely, myasthenia gravis [3,6,7].
There are 3 categories of Castleman disease, each with distinct clinical and cytopathologic features: unicentric Castleman disease, HHV-8-positive multicentric, and HHV-8-negative multicentric Castleman disease. Unicentric Castleman disease is the most common and the easiest to treat, with 94.2% of patients undergoing surgical resection. Unicentric disease affects men and women equally and is most often diagnosed in the fourth decade of life. Pathologically, this disease lies in a spectrum, as there is a hyaline vascular subtype, a plasmocytic subtype, and mixed subtype. While the disease may be unicentric, patients can still have systemic symptoms. Multicentric Castleman disease is subclassified into HHV-8-positive and -negative categories. It affects men slightly more than women and is diagnosed around the sixth decade of life [5]. These patients will often have systemic symptoms, lymphadenopathy, hepatosplenomegaly, and cytopenia, which are mediated by cytokine signaling. HHV-8-positive cases are most often found in HIV-positive or otherwise immunosuppressed patients. Immunosuppression allows HHV-8 to evade host immune control and replicate in the plasmablasts of the lymph nodes. This leads to a cytokine cascade that causes the clinical and cytopathologic features of the disease [12]. Treatment often consists of prednisone, rituximab, and newer monoclonal antibodies directed at IL-6 [11]. In severe cases, cytotoxic chemotherapy can be used. Although the etiology and pathogenesis of HHV-8-positive Castleman disease is well understood, the pathogenesis of HHV-8-negative multicentric Castleman disease is not well understood.
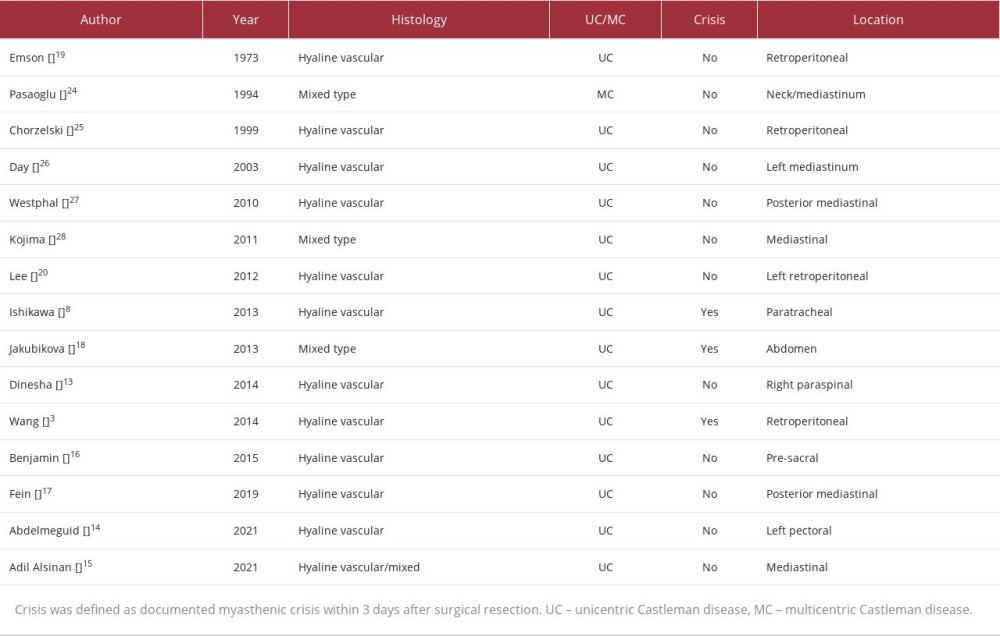
Since 1973, only 16 reported cases of Castleman disease, including the present study, have been complicated by myasthenia gravis. In a previous report, up to 37.5% of these cases were associated with myasthenic crisis after surgical resection, an association much higher than the 5.4% incidence of myasthenic crisis following surgical resection of thymic epithelial tumors [8,9]. Table 1 includes the initial cases reported by Ishikawa et al, in addition to those reported after 2013 [3,13–18]. One case of Castleman disease complicated by both myasthenia gravis and paraneoplastic pemphigus also included a myasthenic crisis; however, the crisis occurred during a hospitalization after a mucosal biopsy and not after the surgical resection of the mass [13]. Another recent case report of Castleman disease and myasthenia gravis described a postoperative myasthenic crisis, which required intubation and intensive care unit admission [3]. Two of the studies reported previously presented with myasthenic crisis 6 months and 8 months after surgery and therefore were not reported as postoperative myasthenic crises [19,20]. In total, 3 of the 16 cases (18.75%) were complicated by postoperative myasthenic crisis within 3 days of surgery.
While less than 20 cases of concomitant Castleman disease and myasthenia gravis have been reported, there may be a shared link in the development of both diseases. Interleukin (IL)-6 appears to play a role in the development of the different types of Castleman disease, as well as in myasthenia gravis. In addition to human IL-6 being elevated in multicentric Castleman disease, the HHV-8 virus encodes a homolog of IL-6 thought to be responsible for some of the pro-inflammatory symptoms seen in patients. In myasthenia gravis, IL-6 is thought to stimulate autoantibody production [21]. While initial studies found that blocking IL-6 with siltuximab in patients with multicentric Castleman disease led to a “clinical beneficial response” [22], further studies are needed to evaluate the efficacy of blocking IL-6 signaling to treat multicentric Castleman disease.
Unique to this case, the patient was also found to have a persistent left SVC. This anomaly is present in 0.3% to 0.5% of the general population without congenital heart defects. Ninety percent of those with a left SVC also have a right SVC that is functional but often narrower and smaller. Ninety-two percent of left SVCs, including in the present case, drain to the right atrium through the coronary sinus and do not pose a risk of cyanosis; the other 8% drain to the left atrium and are usually not large enough to cause cyanosis [23]. The concern in this case was that the lateral aspect of the Castleman disease mass straddled the left SVC. The additional dangers are primarily of bleeding; however, no medial collateral veins were found attached to the mass.
Conclusions
While the outcomes of unicentric Castleman disease alone are excellent, in rare cases of patients with both Castleman disease and myasthenia gravis, there is an increased risk of developing myasthenic crisis after surgery. Successful management of Castleman disease should therefore include a thorough assessment of other possible underlying disorders, such as myasthenia gravis, which if left unrecognized could lead to life-threatening complications.
Figures
References:
1.. Castleman B, Iverson L, Menendez VP, Localized mediastinal lymphnode hyperplasia resembling thymoma: Cancer, 1956; 9(4); 822-30
2.. Keller AR, Hochholzer L, Castleman B, Hyaline-vascular and plasma-cell types of giant lymph node hyperplasia of the mediastinum and other locations: Cancer, 1972; 29(3); 670-83
3.. Wang S, Chen SW, Cai SL, Jin BY, A case report of retroperitoneal pararenal Castleman’s disease associated with myasthenia gravis: World J Surg Oncol, 2014; 12; 331
4.. Maia R, Couto J, Diogo J, Unicentric Castleman disease and pheochromocytoma: Eur J Case Rep Intern Med, 2022; 9(1); 003068
5.. Dispenzieri A, Fajgenbaum DC, Overview of Castleman disease: Blood, 2020; 135(16); 1353-64
6.. Caneppele S, Picart N, Bayle-Lebey P, Paraneoplastic pemphigus associated with Castleman’s tumour: Clin Exp Dermatol, 2000; 25(3); 219-21
7.. Abdel-Reheim FA, Koss W, Rappaport ES, Arber DA, Coexistence of Hodgkin’s disease and giant lymph node hyperplasia of the plasma-cell type (Castleman’s disease): Arch Pathol Lab Med, 1996; 120(1); 91-96
8.. Ishikawa K, Kato T, Aragaki M, A case of Castleman’s disease with myasthenia gravis: Ann Thorac Cardiovasc Surg, 2014; 20(Suppl.); 585-88
9.. Fang W, Chen W, Chen G, Jiang Y, Surgical management of thymic epithelial tumors: A retrospective review of 204 cases: Ann Thorac Surg, 2005; 80(6); 2002-7
10.. Munshi N, Mehra M, van de Velde H, Use of a claims database to characterize and estimate the incidence rate for Castleman disease: Leuk Lymphoma, 2015; 56(5); 1252-60
11.. Robinson D, Reynolds M, Casper C, Clinical epidemiology and treatment patterns of patients with multicentric Castleman disease: Results from two US treatment centres: Br J Haematol, 2014; 165(1); 39-48
12.. Fajgenbaum DC, HHV-8/KSHV-associated multicentric Castleman disease: UpToDate. UpToDate2022 Available online:https://www.uptodate.com/contents/hhv-8-kshv-associated-multicentric-castleman-disease
13.. Dinesha, Padyana M, Nayak K, Castleman’s disease with paraneoplastic pemphigus: Indian J Dermatol, 2014; 59(4); 421
14.. Abdelmeguid A, Rojansky R, Berry GJ, Dewan K, Dysphagia and dysphonia, a pairing of symptoms caused by an unusual pair of diseases: Castleman’s disease and myasthenia gravis: Ann Otol Rhinol Laryngol, 2021; 130(3); 319-24
15.. Adil Alsinan T, Ahmad Robaidi H, Neamatallah Saleh W, A case report of subclinical myasthenia gravis associated with Castleman’s disease: Am J Case Rep, 2021; 22; e930948
16.. Benjamin B, Zaltzman R, Shpitz B, Presacral mass discovered during pregnancy followed by myasthenia gravis: Isr Med Assoc J, 2015; 17(5); 318-20
17.. Fein AS, Trejo Bittar HE, Castleman disease presenting with pseudo-tumour cerebri and myasthenia gravis: A case report and literature review: Neuroophthalmology, 2019; 43(3); 185-91
18.. Jakubikova M, Pitha J, Latta J, Myasthenia gravis, Castleman disease, pemphigus, and anti-phospholipid syndrome: Muscle Nerve, 2013; 47(3); 447-51
19.. Emson HE, Extrathoracic angiofollicular lymphoid hyperplasia with coincidental myasthenia gravis: Cancer, 1973; 31(1); 241-45
20.. Lee SK, Kim DH, Son BS, Castleman’s disease with myasthenia gravis: Korean J Thorac Cardiovasc Surg, 2012; 45(3); 199-201
21.. Yoshizaki K, Murayama S, Ito H, Koga T, The role of interleukin-6 in castle-man disease: Hematol Oncol Clin North Am, 2018; 32(1); 23-36
22.. Rhee Fv, Fayad L, Voorhees P, Siltuximab, a novel anti-inter-leukin-6 monoclonal antibody, for Castleman’s disease: J Clin Oncol, 2010; 28(23); 3701-8
23.. Martins VF, Hsiao A, Kligerman S, Brouha SS, Left circumflex coronary artery-to-coronary sinus fistula with coronary sinus ostial atresia and a persistent left superior vena cava in an adult patient: Radiol Cardiothorac Imag, 2022; 4(1); e210249
24.. Pasaoglu I, Dogan R, Topcu M, Gungen Y, Multicentric angiofollicular lymph-node hyperplasia associated with myasthenia gravis: Thorac Cardiovasc Surg, 1994; 42(4); 253-56
25.. Chorzelski T, Hashimoto T, Maciejewska B, Paraneoplastic pemphigus associated with Castleman tumor, myasthenia gravis and bronchiolitis obliterans: J Am Acad Dermatol, 1999; 41(3 Pt 1); 393-400
26.. Day JR, Bew D, Ali M, Castleman’s disease associated with myasthenia gravis: Ann Thorac Surg, 2003; 75(5); 1648-50
27.. Westphal FL, Lima LC, Santana LC, Castleman’s disease associated with follicular dendritic cell sarcoma and myasthenia gravis: J Bras Pneumol, 2010; 36(6); 819-23
28.. Kojima M, Shimizu K, Kaneko Y, Lymphoid hyperplasia of the thymus showing Castleman’s disease in a patient with myasthenia gravis: Rheumatol Int, 2012; 32(11); 3695-97
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133