06 December 2022: Articles 
A Novel Case Report of Iatrogenic Tracheal Rupture Caused by a Laser-Resistant Endotracheal Tube Under Transoral Laser Laryngeal and Nasopharyngeal Surgery
Diagnostic / therapeutic accidents
Yoko Shimamoto1ABDEF*, Michiyoshi Sanuki1ABDEF, Shigetaki Kurita1BE, Masaya Ueki1B, Yoshie Kuwahara1BDOI: 10.12659/AJCR.938357
Am J Case Rep 2022; 23:e938357
Abstract
BACKGROUND: Intraoperative tracheal rupture due to endotracheal intubation is a rare but serious complication that requires prompt responses. Transoral laser microsurgery is effective for dissection of laryngeal and nasopharyngeal lesions, and a laser-resistant endotracheal tube is therefore commonly used under general anesthesia.
CASE REPORT: We present the case of a 69-year-old man in whom a rare complication involving endotracheal tube tip dislodgement during transoral laser surgery led to iatrogenic tracheal rupture. We used a Laser-Flex cuffed endotracheal tube, which is a non-inflammable, armored stainless-steel tube with a Murphy eye. Repeated mobilization of the laryngoscope blade and excessive neck extension for adequate laryngeal exposure during surgery may have led to significant soft tissue swelling and tube tip displacement, inducing tracheal rupture with the keen edge of the Murphy eye. At the end of the surgical procedure, subcutaneous emphysema was observed in the right anterior neck. Computed tomography revealed subcutaneous emphysema and pneumomediastinum without esophageal injury or mediastinitis. The injury was 1 cm in length, with wall involvement to a depth to the muscular wall in the membranous trachea at a point 2 cm proximal to the carina, in which we could position the alternative endotracheal tube distal to the tracheal rupture. After conservative treatment, the patient was extubated and mechanical ventilation was ceased.
CONCLUSIONS: During transoral laser laryngeal and nasopharyngeal surgery, dislodgement of the laser-resistant endotracheal tube tip can lead to iatrogenic tracheal rupture. In this case, injury during application of a Laser-Flex cuffed endotracheal tube with a Murphy eye, was followed by conservative treatment. This treatment achieved a successful outcome.
Keywords: Laser Therapy, Postoperative Complications, Subcutaneous Emphysema, Male, Humans, Aged, Trachea, Rupture, Intubation, Intratracheal, Tracheal Diseases, Lasers, Iatrogenic Disease
Background
Intraoperative tracheobronchial rupture is a rare but serious complication requiring prompt diagnosis, management, and treatment. Most cases of intraoperative tracheobronchial rupture are caused by tracheal intubation, for which the estimated incidence is 0.05–0.37% [1]. This clinical diagnosis is made when cervical subcutaneous emphysema is accompanied by snowball crepitation, pneumomediastinum, or respiratory failure. Differential diagnoses include other causes of rapid-onset cervical swelling such as hemorrhage, hematoma, or allergic reactions, and a rapid diagnosis of tracheal rupture is essential to avoid the above-mentioned complications. Furthermore, the source of air leakage should be correctly identified using fiberoptic bronchoscopy and computed tomography (CT) scans. Decisions regarding surgical or conservative treatment should be made based on the patient’s general state in addition to analysis of the available clinical, radiological, and endoscopic data. The main problems encountered by anesthesiologists include airway management for ventilation and fast recovery of spontaneous breathing without worsening tracheal lacerations.
Transoral laser microsurgery has been established as an effective option in the treatment of glottic, supraglottic, and hypopharyngeal tumors, and is usually performed under general anesthesia with a laser-resistant endotracheal tube. Careful airway management and ventilation are required. Here we present a case of intraoperative tracheal rupture in a patient with pulmonary emphysema, pulmonary hypertension, and liver cirrhosis, with a higher risk of bleeding, who underwent transoral laser microsurgery for both laryngeal and nasopharyngeal tumors.
Case Report
A 69-year-old man (height, 157 cm; body weight, 47 kg) underwent resection of both laryngeal and nasopharyngeal tumors. His medical history included hypertension, current smoking, pulmonary emphysema, pulmonary hypertension (combined groups 1 and 3), and alcoholic liver cirrhosis with portal hyper-tension, for which he was prescribed furosemide 20 mg orally once a day, spironolactone 25 mg orally once a day, tolvaptan 7.5 mg orally once a day, macitentan 10 mg orally once a day, ambroxol hydrochloride sustained-release 45 mg orally once a day, tiotropium bromide hydrate 2.5 μg/olodaterol hydrochloride 2.5 μg inhaler, 1 puff once a day, theophylline 200 mg orally twice a day, and ursodeoxycholic acid 100 mg orally 3 times a day.
Echocardiography revealed a left ventricular ejection fraction of 66%, aortic regurgitation of 2/4, mitral regurgitation of 2/4, tricuspid regurgitation of 3/4, left atrial dilation (left atrial volume index 45 ml/m2), and right atrial and ventricular dilations, indicative of right ventricular heart failure and pulmonary hypertension. His estimated right ventricular pressure gradient on Doppler echocardiography was 75 mmHg. Preoperative spirometry showed a vital capacity of 2600 mL (% vital capacity: 80.7%), forced expiratory volume (FEV)1.0 1260 mL, and FEV1.0% 51.8%, compatible with chronic obstructive pulmonary disease (Hugh-Jones classification III, SpO2 94% on room air). Abdominal computed tomography and preoperative CT scan of the chest both revealed pulmonary emphysema, with multiple blebs in the upper lobes, pulmonary artery dilation, liver cirrhosis, splenomegaly, and ascites. He was thus considered to have American Society of Anesthesiologists Physical Status Class III for surgery under general anesthesia.
General anesthesia was induced with an intravenous bolus of remimazolam at 6 mg, remifentanil at 0.5 μg/kg/min, and rocuronium at 0.8 mg/kg. Following loss of consciousness, the patient subsequently received remimazolam at infusion rates of 0.3 to 1.0 mg/kg/h as the maintenance dose, which was adjusted to maintain adequate sedation using a pressure-regulated volume control mode. After induction, for the use of carbon dioxide (CO2) lasers, a Laser-Flex cuffed endotracheal tube with a Murphy eye as a side hole (inner diameter 5.5 mm, Covidien Products, Tokyo, Japan) was inserted under video laryngoscopy on the first pass, and was secured 22 cm at the left angle of the mouth. Endotracheal tube introducers were not used in this procedure. When direct laryngoscopy was performed in an extended neck position for adequate laryngeal exposure during surgery, the laryngoscope blade was mobilized several times. This induced soft tissue injuries, including mucosal lacerations and bleeding in both the sublingual portion and the upper lip. A further extended neck position enabled sufficient exposure and resection of the laryngeal tumor. Subsequently, rigid nasopharyngoscopy was performed to confirm the nasopharyngeal tumor and induce bleeding due to the fragility of the nasal mucosa. After laser resection of the nasopharyngeal tumor, a massive hemorrhage in the oral cavity required reoperation to arrest bleeding. Blood leakage from the nasopharynx was treated with Bellocq tamponade and the epiglottis was coagulated using an electrocauterizer. Inspection and palpation at the end of the surgery revealed snowball crepitation, indicating subcutaneous emphysema around the right anterior neck. CT confirmed subcutaneous emphysema (Figure 1) and pneumomediastinum (Figure 2), with a 1-cm injury on the right side of the membranous trachea at a point 2 cm proximal to the carina (Figure 3), without concurrent esophageal injury or mediastinitis. This tracheal injury was also observed by video bronchoscopy. According to the classification of Cardillo et al, depending on the depth of the wall involvement, this lesion was staged as Level II, for which conservative treatment is proposed [2]. Using a tube exchanger (12 Fr), the Laser-Flex cuffed endotracheal tube was replaced with a standard endotracheal tube (Taper Guard endotracheal tube, Covidien Products, Tokyo, Japan; inner diameter 7.0 mm), which was secured 23 cm at the left angle of the mouth. Video bronchoscopy confirmed the positioning of the tip of the endotracheal tube distal to the tracheal laceration.
The patient was transferred to the intensive care unit (ICU) with postoperative bleeding, severe lingual edema, and tracheal injury in an unfavorable state, and was maintained under volume-controlled synchronized intermittent mandatory ventilation for postoperative ventilation management (VC-SIMV; tidal volume 400 mL, Peep 5 cm H2O, FiO2 0.5, respiratory rate 14, pressure support 15 cm H2O). Table 1 shows postoperative blood gas test data in the ICU. Postoperative sedation was maintained with propofol at 2 mg/kg/h and dexmedetomidine at 0.4 μg/kg/h. Noradrenaline was administered at 0.05–0.07 μg/kg/min as a vasoconstrictor, to maintain coronary perfusion pressure in response to the blood pressure-lowering effect of propofol, accompanied by systemic vascular resistance (Table 2). Twenty-four hours after surgery, CT revealed a slight improvement in both subcutaneous emphysema and pneumomediastinum, and confirmed no further extension of the lesion by air leaks (Figure 4). Administration of inotropes, including dopamine at 2–3 μg/kg/min and dobutamine at 1–4 μg/kg/ min, was initiated to improve the contractility of the right and left ventricles. Forty-eight hours after surgery, the mechanical ventilatory weaning was prepared. This preparation included analgosedation with fentanyl at 2 μg/kg/h under spontaneous-continuous positive airway pressure support (SPN-CPAP/PS, Peep 5 cm H2O, pressure support 10 cm H2O, FiO2 0.4, respiratory rate 14). Seventy-two hours after surgery, video bronchos-copy confirmed healing of the tracheal laceration (Figure 5).
One hundred and twenty hours after the surgery, the patient was extubated and released from mechanical ventilation, under oxygen supplementation at 5 L/min. During the postoperative period, spirometry showed no loss of tidal volume or no differences between inspiratory and expiratory volumes. Table 3 shows blood test results before and after surgery.
Discussion
Transoral laryngopharyngeal surgery is associated with severe postoperative complications. Although rare complications, these are potentially life-threatening, and include postoperative bleeding, cervical subcutaneous emphysema, and/or pneumomediastinum, which may compress the trachea [3–6]. The pathogenic mechanisms underlying postoperative cervical subcutaneous emphysema can be categorized into 2 groups. The first potential mechanism involves intubation. Iatrogenic rupture occurs along the tracheobronchial tree during endotracheal tube passage or overinflation of the endotracheal tube cuff. Air may further enter the peritracheal or perialveolar tissues. Moreover, excessive positive pressure ventilation or increased intrathoracic pressure may induce rupture of the alveolar bleb, causing air to enter the mediastinum via the perivascular sheath. Furthermore, leaked air may ascend into the subcutaneous tissues of the neck. Surgical procedures, the second pathogenic mechanism, can give rise to mucosal and muscular laceration of the larynx and/or pharyngeal rupture, especially in the pyriform sinus, due to complications after endoscopic laryngopharyngeal surgery. Kishimoto et al [3] also previously demonstrated that the resection of lesions in the pyriform sinus is associated with subcutaneous emphysema.
In our case, postoperative subcutaneous emphysema was caused by a tracheal laceration induced by a laser-resistant endotracheal tube. Most laryngeal surgeries are performed using CO2 lasers with laser-resistant endotracheal tubes to reduce the risk of airway fires. Laser-resistant endotracheal tubes have larger external diameters than the given internal diameter. This is because of the need for laser-resistant coatings or materials, either made of metal or of a nonmetal tube core with a metallic overlay. We used a Laser-Flex cuffed endotracheal tube, which is a non-inflammable, armored stainless-steel tube with a Murphy eye, the edge of which is sharp and hard, with dual extra-large-diameter cuffs (Figure 6).
During laryngeal procedures, the endotracheal tube sometimes interferes with visualization, making it difficult to dissect the laryngeal lesions. The endotracheal tube was positioned according to the surgeon’s preference to facilitate the introduction of surgical instruments for laryngeal manipulation. Laryngoscopy causes abrupt pressure primarily at the base of the tongue and hypopharynx with intense stimulation [7–9]. Local edema or hematoma created by concealed local bleeding may obstruct the airway postoperatively and require emergency management in the operating room and ICU. In this case, we hypothesized that repeated mobilization of the laryngoscope blade and excessive neck extension for adequate laryngeal exposure may have resulted in displacement of the tube tip and induced tracheal rupture at the keen edge of the Murphy eye. Among the currently available endotracheal tubes for CO2 laser surgery, only the Laser-Flex has a Murphy eye as a side hole, which remains a universal feature of endotracheal tubes to provide air passage in situations where the tube tip is obstructed by mucus [10]. Hall et al [11] used computational fluid dynamics modeling to simulate an endotracheal tube and investigated the role of the Murphy eye. Notably, the Murphy eye is designed so that a high gas velocity through the endotracheal tube results in no flowthrough until the tip is almost occluded. When the tip is fully occluded, all the flow passes through the Murphy eye as an alternative flow path, which prevents immediate harm and has saved countless lives.
Patients with hemorrhagic tendencies have a higher risk of significant injury [12,13]. Our patient regularly consumed alcohol, which induced alcoholic liver cirrhosis and portal hypertension with thrombocytopenia (preoperative platelet level 88×109/L). He was also an active smoker, with a tobacco consumption of 50 pack-years and pulmonary emphysema. Furthermore, he had pulmonary hypertension (combined groups 1 and 3: group 1, pulmonary arterial hypertension associated with portal hypertension; group 3, pulmonary hypertension due to lung diseases and/or hypoxia), which may have also led to a higher risk of bleeding [14]. Owing to hypoxemic vasoconstriction and microthrombus, reduced pulmonary circulation at the pulmonary arteriole induced engorged bronchial arteries [15]. This condition can easily lead to rupture and bleeding.
The treatment decision for iatrogenic tracheal rupture depends on the location and extent of the rupture, in addition to the injury size (length and depth) and the patient’s respiratory status. Several comprehensive algorithms have been utilized for the management of intraoperative tracheobronchial ruptures. Regarding injury size, some authors have promoted conservative treatment in patients with injury lengths less than 2 cm [16], while others have stressed the importance of measuring injury depth independently of length and proposed shallow injury as the condition to guide the selection of conservative treatment [2,17–19]. In general, conservative management may be considered sufficient for stable patients with small lacerations, regardless of whether air leaks are present, whereas invasive surgical therapy may be more appropriate for patients with large defects with significant air leaks and instability. Our patient had a tracheal laceration 1 cm in length, at a depth sufficient to induce tracheal wall involvement up to the muscular wall, with subcutaneous emphysema and pneumomediastinum without esophageal injury or mediastinitis. According to the classification of Cardillo et al, this lesion was classified as Level II, for which conservative treatment is recommended [2]. Furthermore, we positioned the endotracheal tube distal to the tracheal rupture, which was located 2 cm proximal to the carina. Although subcutaneous emphysema and pneumomediastinum existed, they did not progress during mechanical ventilation, no loss of tidal volume was seen, and the inspiratory and expiratory volumes remained unchanged on spirometry. While the optimal method for repair and protection is extubation, most patients have no choice but to remain intubated postoperatively. From an anesthesiological point of view, the minimal work required involves airway management to guarantee breathing and oxygenation without worsening the tracheal laceration. We positioned the tip of the orotracheal tube distal to the tracheal lesion with ventilator settings in low Peep (5 cm H2O) because high-pressure support can negatively affect tracheobronchial rupture.
Conclusions
Here we present the first case of a patient in whom laser-resistant endotracheal tube tip dislodgement during transoral laser surgery led to iatrogenic tracheal rupture. Application of a Laser-Flex cuffed endotracheal tube with a Murphy eye, the edge of which is sharp and hard, led to the rupture. Conservative treatment of the rupture achieved a successful outcome.
Figures
References:
1.. Prokakis C, Koletsis EN, Dedeilias P, Airway trauma: A review on epidemiology, mechanisms of injury, diagnosis and treatment: J Cardiothorac Surg, 2014; 9; 117
2.. Cardillo G, Carbone L, Carleo F, Tracheal lacerations after endotracheal intubation: A proposed morphological classification to guide non-surgical treatment: Eur J Cardiothorac Surg, 2010; 37; 581-87
3.. Kishimoto Y, Sogami T, Uozumi R, Complications after endoscopic laryngopharyngeal surgery: Laryngoscope, 2018; 128; 1546-50
4.. Chiesa Estomba CM, Reinoso FA, Velasquez AO, Complications in CO2 laser transoral microsurgery for larynx carcinomas: Int Arch Otorhinolaryngol, 2016; 20; 151-55
5.. Ambrosch P, Gonzalez-Donate M, Fazel A, Transoral laser microsurgery for supraglottic cancer: Front Oncol, 2018; 8; 158
6.. Chiesa-Estomba CM, Sistiaga-Suarez JA, González-García JA, Intraoperative surgical complications in transoral laser CO2 microsurgery of the larynx: An observational, prospective, single-center study: Ear Nose Throat J, 2021; 100; 456S-61S
7.. Shribman AJ, Smith G, Achola KJ, Cardiovascular and catecholamine responses to laryngoscopy with and without tracheal intubation: Br J Anaesth, 1987; 59; 295-99
8.. Hassan HG, el-Sharkawy TY, Renck H, Mansour G, Fouda A, Hemodynamic and catecholamine responses to laryngoscopy with vs. without endotracheal intubationr: Acta Anaesthesiol Scand, 1991; 35; 442-47
9.. Kahl M, Eberhart LH, Behnke H, Stress response to tracheal intubation in patients undergoing coronary artery surgery: Direct laryngoscopy versus an intubating laryngeal mask airway: J Cardiothorac Vasc Anesth, 2004; 18; 275-80
10.. Murphy FJ, Two improved intratracheal catheters: Anesth Analges, 1941; 20; 102-25
11.. Hall EJ, Burns AD, Ng AC, Lumb AB, Does the Murphy’s eye perform its role?: Anaesthesia, 2015; 70; 1326-29
12.. Loh KS, Irish JC, Traumatic complications of intubation and other airway management procedures: Anesthesiol Clin North Am, 2002; 20; 953-69
13.. Weymuller EA, Prevention and management of intubation injury of the larynx and trachea: Am J Otolaryngol, 1992; 13; 139-44
14.. Chin KM, Channick RN, de Lemos JA, Hemodynamics and epoprostenol use are associated with thrombocytopenia in pulmonary arterial hypertension: Chest, 2009; 135; 130-36
15.. Liebow AA, Hales MR, Lindskog GE, Enlargement of the bronchial arteries, and their anastomoses with the pulmonary arteries in bronchiectasis: Am J Pathol, 1949; 25; 211-31
16.. Fiorelli A, Cascone R, Di Natale D, Endoscopic treatment with fibrin glue of post-intubation tracheal laceration: J Vis Surg, 2017; 3; 102
17.. Deja M, Menk M, Heidenhain C, Strategies for diagnosis and treatment of iatrogenic tracheal ruptures: Minerva Anestesiol, 2011; 77; 1155-66
18.. Conti M, Pougeoise M, Wurtz A, Management of postintubation tracheobronchial ruptures: Chest, 2006; 130; 412-18
19.. Schneider T, Storz K, Dienemann H, Hoffmann H, Management of iatrogenic tracheobronchial injuries: A retrospective analysis of 29 cases: Ann Thorac Surg, 2007; 83; 1960-64
Figures
Tables
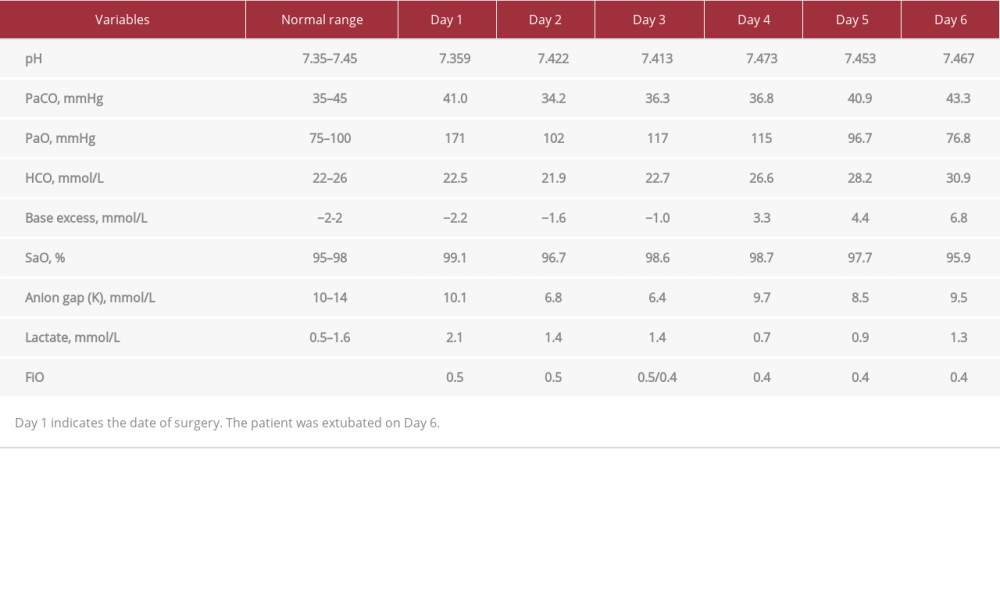 Table 1.. Blood gas analysis after surgery in the Intensive Care Unit.
Table 1.. Blood gas analysis after surgery in the Intensive Care Unit.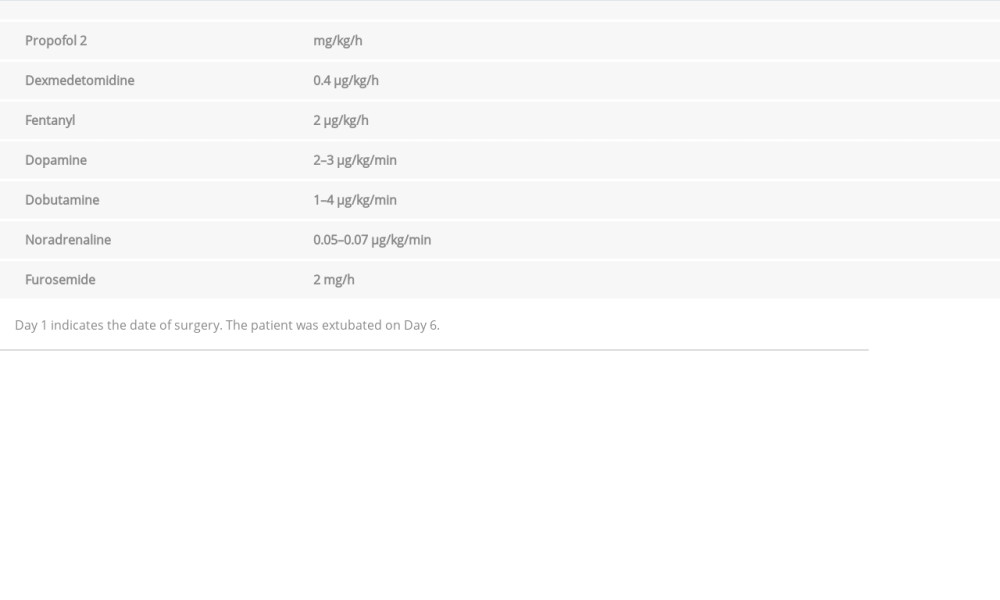 Table 2.. Intravenous medication after surgery in the Intensive Care Unit.
Table 2.. Intravenous medication after surgery in the Intensive Care Unit.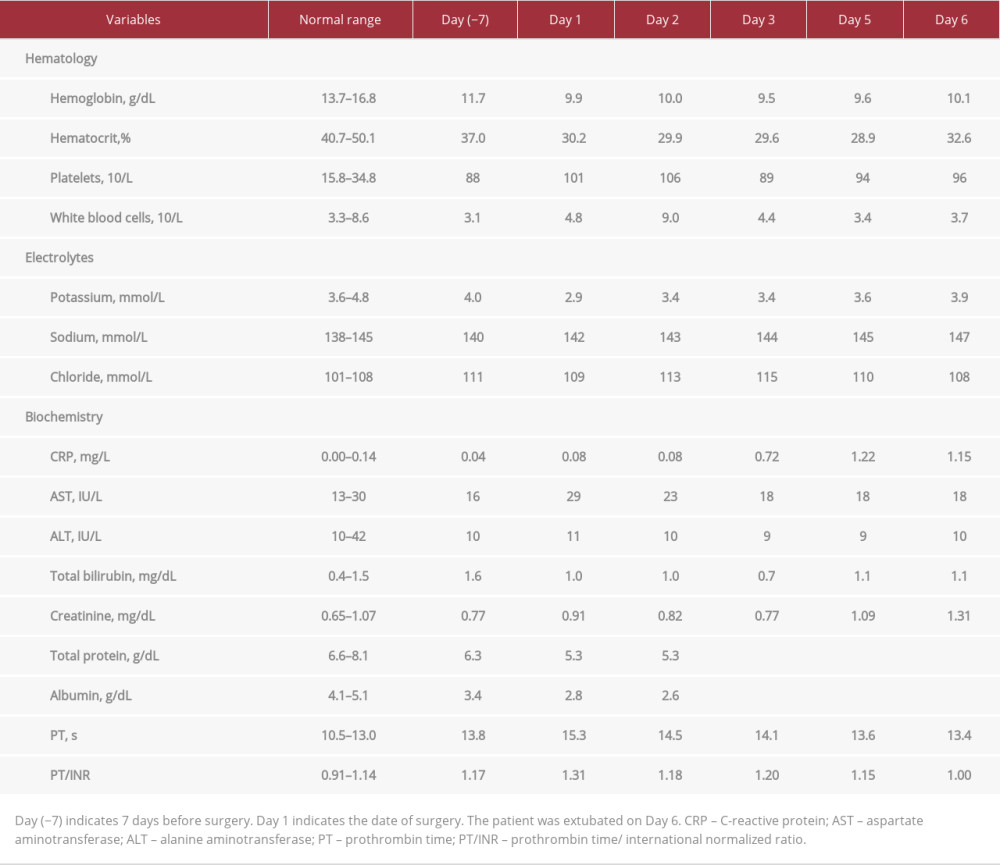 Table 3.. Blood laboratory test results before and after surgery.
Table 3.. Blood laboratory test results before and after surgery. Table 1.. Blood gas analysis after surgery in the Intensive Care Unit.
Table 1.. Blood gas analysis after surgery in the Intensive Care Unit. Table 2.. Intravenous medication after surgery in the Intensive Care Unit.
Table 2.. Intravenous medication after surgery in the Intensive Care Unit. Table 3.. Blood laboratory test results before and after surgery.
Table 3.. Blood laboratory test results before and after surgery. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








