01 February 2023: Articles 
Alveolar Hemorrhage Following Positron Emission Tomography/Computed Tomography in 2 Separate Episodes 5 Months Apart
Unknown etiology, Challenging differential diagnosis, Unexpected drug reaction
Jasmin KhateebDOI: 10.12659/AJCR.938668
Am J Case Rep 2023; 24:e938668
Abstract
BACKGROUND: Positron emission tomography/computed tomography (PET/CT) has become one of the most prominent modalities worldwide for the diagnosis and surveillance of malignancies. Current clinical imaging guidelines report adverse reactions following PET/CT, especially due to contrast-induced toxicities, such as contrast-induced nephropathy and other rare reactions attributed to a hypersensitivity immune response, such as bronchospasm. Other rare lung toxicities were reported in a few case reports. Herein, we report repeated episodes of alveolar hemorrhage, a novel adverse response to PET/CT, occurring on 2 separate occasions 5 months apart.
CASE REPORT: A 57 year-old female patient with breast carcinoma managed by mastectomy, adjuvant chemotherapy, irradiation, and hormonal therapy presented with massive alveolar hemorrhage following PET/CT performed for surveillance 13 years after completion of chemotherapy and irradiation. An additional episode of massive alveolar hemorrhage occurred 5 months later following PET/CT, with respiratory failure requiring mechanical ventilation. Fluorine-18 fluorodeoxyglucose ([¹⁸F] FDG) and iohexol were used for imaging on both occasions. Common causes of alveolar hemorrhage, including malignancy, were excluded.
CONCLUSIONS: The repeated episodes immediately following PET/CT and the earlier and more intense respiratory failure following the second event raise the possibility of an immune-mediated alveolar hemorrhage in response to either the administration of iodinated radiocontrast agent or to [¹⁸F] FDG.
Keywords: Contrast Media, Fluorodeoxyglucose F18, Hemoptysis, Positron Emission Tomography Computed Tomography, Hypersensitivity, Respiratory Insufficiency, Female, Humans, Middle Aged, Breast Neoplasms, Mastectomy, Hemorrhage
Background
Hemoptysis is a worrisome infrequent clinical presentation. Its differential diagnosis ranges from severe acute left-heart failure with pulmonary congestion, through various forms of acute or chronic infections and congenital or acquired structural changes in the pulmonary vasculature, to chemical-related injuries or malignancies affecting the lungs [1]. It can also appear in thrombo-emboli with pulmonary consolidation or infarction or in various forms of immune-mediated or inflammatory lesions affecting the pulmonary parenchyma and blood vessels and in hemostatic disorders [2,3].
Herein, we present a 57-year-old female patient with a history of breast cancer, under surveillance for breast carcinoma, who developed twice alveolar hemorrhage shortly following Fluorine-18 fluorodeoxyglucose ([18F] FDG) positron-emission tomography, coupled with radiocontrast-enhanced computerized tomography (PET/CT). We propose that this repeated presentation with hemoptysis reflect a rare and hardly reported adverse response to the imaging procedure, related to [18F] FDG and/or iodinated radiocontrast medium.
Case Report
A 57-year-old non-smoking female patient with a history of locally invasive ductal carcinoma of the breast managed by left mastectomy, adjuvant chemotherapy, local radiation, trastuzumab, and letrozole had been in remission for 13 years; additional medical history included type II diabetes mellitus, dyslipidemia, hypertension, obesity (body mass index, 32), and long-term treatment with aspirin for primary prevention of atherosclerotic complications.
The patient underwent a follow-up PET/CT performed with the use of [18F] FDG and iohexol, a low-osmolar radiocontrast medium, as part of a newly discovered bone mass.
Seventy-two minutes after the completion of the imaging procedure, she presented in the Emergency Department (ED) with massive hemoptysis. She denied bleeding diathesis, the use of anticoagulants or additional anti-platelet medications, recent fever or respiratory complains, previous allergic reactions, chest wall trauma, and interventional procedures.
On admission, she had dyspnea without tachypnea, with a room oxygen saturation of 88%, normalized with the administration of oxygen. She was afebrile, sinus tachycardia of 110 beats/min was recorded, and her blood pressure was 170/110 mmHg (Table 1A).
On examination, decreased breath sounds above both lung bases were noted; there were no coetaneous or mucosal petechial lesions or telangiectasis. Upper airway and gastrointestinal bleeding were ruled out. Complete blood count, coagulation profile, renal and hepatic blood tests results were all normal. Autoimmune profiles, including anti nuclear antibodies, antineutrophil cytoplasmic antibodies, and anti-glomerular basement membrane antibodies, were all negative (Table 1B).
The already performed total body PET/CT scan detected ground-glass opacities with concurrent overlapping high radioactive uptake in the right upper and right middle lobes, compatible with diffuse alveolar hemorrhage, and ruled out metastatic disease, segmental pulmonary embolism, vascular and anatomical abnormalities, and other space-occupying lesions. No radioactive uptake was detected elsewhere (Figure 1).
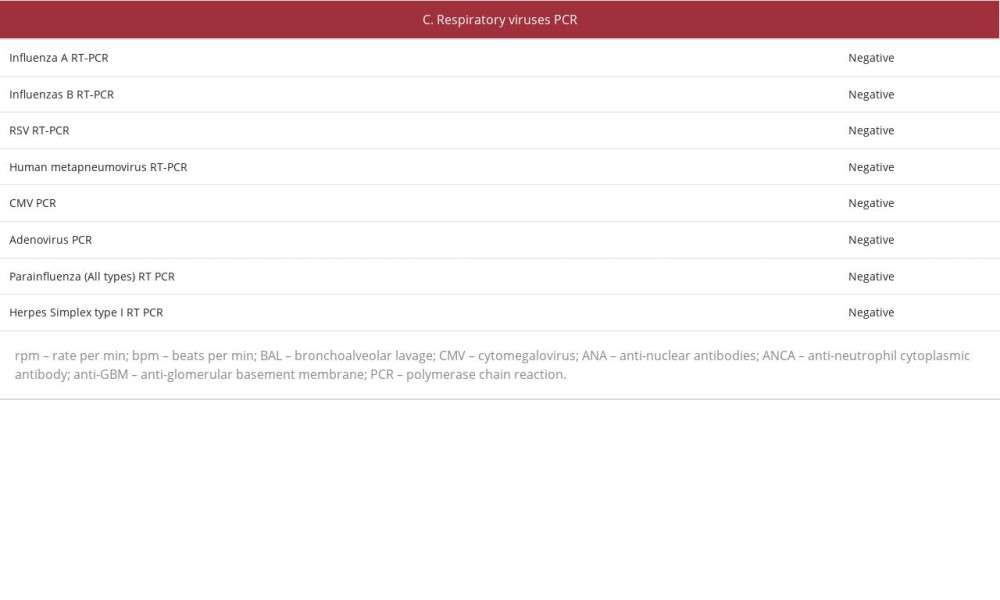
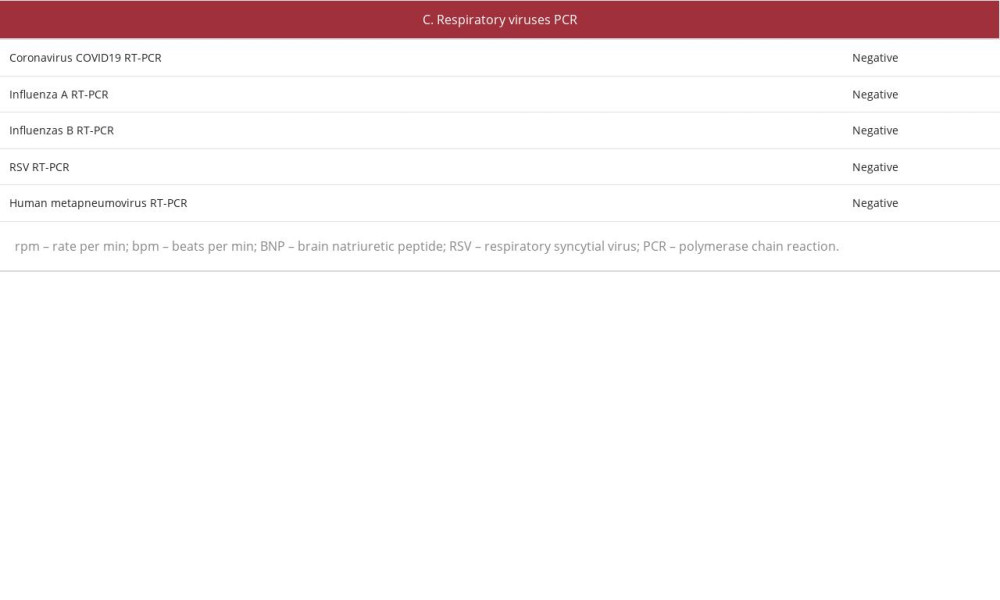
A fiberoptic bronchoscopy, which was performed 48 h after the initial hemoptysis episode, showed mucosal petechias at the right main bronchus, without active bleeding or purulent secretions. Bronchoalveolar lavage cytology confirmed diffuse alveolar hemorrhage by the presence of hemosiderin-loaded alveolar macrophages. Various known etiologies of hemoptysis were excluded, including infections (negative bacterial and fungal cultures, Gram and acid-fast stains, and PCR for respiratory viruses), pulmonary emboli (imaging), and immune-mediated disorders (negative autoantibodies) (Table 1C). Eventually, hemoptysis resolved spontaneously, and the patient was discharged from the hospital without a defined diagnosis or treatment.
In the meantime, a biopsy specimen was taken from the bone mass, and pathology revealed benign tenosynovial giant cell tumor.
Five months later, the patient underwent a repeated PET/CT scan using the same protocol with [18F] FDG and iohexol. Fifty-two minutes later, she presented to the ED with acute hypoxemic respiratory failure and massive hemoptysis (Table 2A–2C). Emergent treatment with intravenous epinephrine and hydro-cortisone was initiated for a presumed hypersensitivity reaction. Emergent fiberoptic examination of the upper airways excluded laryngeal edema and upper airway bleeding. Gastric aspiration with the use of nasogastric tube excluded upper gastrointestinal bleeding. Due to respiratory compromise, the patient required endotracheal intubation and was mechanically ventilated.
The PET/CT scan performed prior to this second episode of hemoptysis showed pulmonary infiltrates suggestive for diffuse alveolar hemorrhage, similar to the prior PET/CT scan 5 months earlier, without evidence of metastases. Chest X-ray upon admission showed bilateral alveolar pulmonary infiltrates, compatible with alveolar bleeding. CT angiography with iohexol performed 4 h later for the exclusion of thrombemboli demonstrated bilateral alveolar ground-glass opacities, suggestive of diffuse alveolar hemorrhage, and was otherwise normal (Figure 2).
A fiberoptic bronchoscopy revealed diffuse alveolar hemorrhage, without purulent secretions (Figure 3). The patient was admitted to the Intensive Care Unit and received supportive care in addition to empiric antibiotic therapy with levofloxacin. Two days later, the patient’s condition stabilized, and she was extubated and later discharged. She remained well, free of hemoptysis, and without evidence of recurring malignancy over the following 18 months. It is noteworthy that the patient underwent previously PET/CT once only, uneventfully, a year before she developed the first episode of hemoptysis.
Discussion
We present an extremely rare case of acute hemoptysis as a manifestation of alveolar hemorrhage following PET/CT. In this case, 2 separate episodes were reported, with the second episode showing an accelerated and more severe reaction that resulted in hypoxemic respiratory failure and the need of mechanical ventilation, suggesting an acute hypersensitivity reaction.
The source of hemoptysis was established by ruling out upper airway and gastrointestinal bleeding and by the typical finding of hemosiderin-laden macrophages in alveolar wash fluids.
Our patient has been previously treated with trastuzumab, letrozole, and radiation therapy, which are known to cause diverse manifestations of lung injury, especially pneumonitis, and can occasionally present with alveolar hemorrhage [4,5].
However, these agents were administered 10 years previously, and lung injury was not noted at that time. Extensive laboratory evaluation of other causes were negative, including coagulation profile, a search for viral and bacterial pathogens, and autoimmune serologic tests, ruling out lung infection and autoimmune-mediated diseases as the cause of alveolar hemorrhage. Echocardiography ruled out mitral stenosis.
The setting and timing of hemoptysis, which occurred twice shortly following repeated PET/CT scans, suggest that intravenous iodinated contrast media, or [18F] FDG, was the trigger to hemoptysis.
[18F] FDG is a positron-emitting radiopharmaceutical agent used in PET/CT scan for the diagnosis and surveillance of various malignancies, inflammatory lesions, and infections. Adverse reactions to [18F] FDG are relatively uncommon, with mainly minor reactions, such as rash, erythema, pruritus, and dysgeusia [6]. More serious adverse events have rarely been reported following the administration of [18F] FDG, including exfoliative dermatitis, sympathetic overactivity with hypertension and tachycardia, or anaphylactic responses with shock, angioedema, seizures, diaphoresis, vomiting, and diarrhea. A few lethal events were also reported [7–11]. As opposed to the very rare occurrence of immediate adverse responses to radiopharmaceuticals (up to a few patients per 100 000 procedures), acute IgE-mediated and non-IgE mediated adverse reactions to iodinated radiocontrast agents administered during CT are quite common. Self-limited hot flushes and erythema occur almost invariably, reflecting cutaneous vasodilation. More worrisome are features of anaphylaxis, with shock, angioedema, bronchospasm, and respiratory failure, as well as cardiac arrhythmias and pulmonary edema [12]. More delayed serious adverse reactions include contrast-induced nephropathy among patients with advanced renal failure, related to hypoxic and oxygen-free radical-mediated tubular injury [13,14], as well as thyrotoxicosis and parotitis [12,15].
Reported pulmonary adverse reactions following intravascular administration of radiocontrast media include bronchospasm or upper airway compromise due to laryngeal edema, with significant hypoxia [16]. American College of Radiology guidelines for the management of allergic-like contrast-induced adverse events include the administration of oxygen, antihista-mines, systemic steroids, intravenous fluids, bronchodilators, and epinephrine [17].
Respiratory complications related to anaphylaxis were very rarely reported also following the administration of [18F] FDG [9]. Yet, alveolar hemorrhage with hemoptysis has never been noticed. By contrast, a few case reports do indicate an association of hemoptysis with the administration of radiocontrast materials. Two patients concurrently treated with amiodarone developed fatal alveolar hemorrhage an hour following intravenous iodinated contrast agents administered during pulmonary angiography, in the evaluation of hypoxemia and pleuritic pain, suggesting an existing pulmonary pathology dissimilar to our case [18]. In another well-documented case report resembling our patient, an 81-year-old patient underwent 2 repeated coronary angiographic studies and interventions, ending with recurrent alveolar hemorrhage and hemoptysis [19]. As with our patient, the second episode occurred faster and was associated with a more intense respiratory compromise requiring intubation and mechanical ventilation; however, the hemoptysis developed days and not hours after the first exposure to radiocontrast media in contrast to our case. Other causes for hemoptysis were excluded, and the patient convalesced with the administration of corticosteroids [19].
The repeated temporal association of alveolar hemorrhage and PET/CT favors a cause-and-effect relationship with the administered materials, and the more prompt and intense reaction in the second episode suggests an immune-mediated reaction, possibly affecting the alveolar capillaries. The causative agent remains speculative. On one hand, alveolar hemorrhage has been encountered following the administration of iodinated contrast agent but not following [18F] FDG. On the other hand, several clues favor a role for [18F] FDG. First, CT anomalies showing ground-glass opacities were detected already on the initial PE/CT study (Figure 1). This procedure is initiated by PET with a protracted period of emission data acquisition, followed by CT, performed immediately after the injection of contrast material. Therefore, it is more likely that the imaging abnormalities were generated by [18F] FDG. Furthermore, detected PET anomalies developed before the administration of the radiocontrast agent. Finally, CT angiography performed during the second event was not associated with detected clinical deterioration, although the patient was mechanically ventilated and after the administration of steroids, which could mask a repeated episode of alveolar hemorrhage.
With all that in mind, we favor the possibility that alveolar hemorrhage occurred in our patient in response to [18F] FDG and was evident 72 min and 52 min in the first and second events, respectively, following the completion of CT, the last component of the PET/CT (Figure 4).
Conclusions
This case report suggests that alveolar hemorrhage can be a rare adverse effect of injected [18F] FDG, unreported, to date. The nature of this life-threatening complication is yet to be defined, but its extreme rarity and intensification on repeated exposure likely indicate a hypersensitivity-mediated reaction rather than a predicted toxic alveolar capillary injury. A possible role for the administration of radiocontrast material in the development of alveolar hemorrhage, especially as a “second hit”, cannot be unequivocally excluded.
Figures
References:
1.. Jean-Baptiste E, Clinical assessment and management of massive hemoptysis: Crit Care Med, 2000; 28(5); 1642-47
2.. Earwood JS, Thompson TD, Hemoptysis: evaluation and management: Am Fam Physician, 2015; 91(4); 243-49
3.. Krause ML, Cartin-Ceba R, Specks U, Peikert T, Update on diffuse alveolar hemorrhage and pulmonary vasculitis: Immunol Allergy Clin North Am, 2012; 32(4); 587-600
4.. Hackshaw MD, Danysh HE, Singh J, Incidence of pneumonitis/inter-stitial lung disease induced by HER2-targeting therapy for HER2-positive metastatic breast cancer: Breast Cancer Res Treat, 2020; 183(1); 23-39
5.. Peddi PF, Shatsky RA, Hurvitz SA, Noninfectious pneumonitis with the use of mTOR inhibitors in breast cancer: Cancer Treat Rev, 2014; 40(2); 320-26
6.. Meher BR, Agrawal K, Gnanasegaran G, Review of adverse reactions associated with the use of common diagnostic radiopharmaceuticals: Indian J Nucl Med, 2021; 36(2); 163-67
7.. Matsuda H, Uehara T, Okazawa H, Full report on a survey of adverse reactions to radiopharmaceuticals from 1975 to 2017 in Japan: Ann Nucl Med, 2020; 34(4); 299-304
8.. Kennedy-Dixon TG, Gossell-Williams M, Cooper M, Evaluation of radiopharmaceutical adverse reaction reports to the British Nuclear Medicine Society from 2007 to 2016: J Nucl Med, 2017; 58(12); 2010-12
9.. Schreuder N, Koopman D, Jager PL, Adverse events of diagnostic radio-pharmaceuticals: A systematic review: Semin Nucl Med, 2019; 49(5); 382-410
10.. Laroche ML, Quelven I, Mazère J, Merle L, Adverse reactions to radiopharmaceuticals in France: Analysis of the national pharmacovigilance database: Ann Pharmacother, 2015; 49(1); 39-47
11.. Pérez-Iruela JA, Pastor-Fructuoso P, De Gracia-Rodríguez C, Adverse reactions to radiopharmaceuticals: Farm Hosp, 2021; 45(3); 142-49
12.. Andreucci M, Solomon R, Tasanarong A, Side effects of radiographic contrast media: Pathogenesis, risk factors, and prevention: Biomed Res Int, 2014; 2014; 741018
13.. Heyman SN, Rosen S, Rosenberger C, Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy: Clin J Am Soc Nephrol, 2008; 3(1); 288-96
14.. Heyman SN, Rosen S, Khamaisi M, Reactive oxygen species and the pathogenesis of radiocontrast-induced nephropathy: Invest Radiol, 2010; 45(4); 188-95
15.. Mehran R, Nikolsky E, Contrast-induced nephropathy: Definition, epidemiology, and patients at risk: Kidney Int Suppl, 2006(100); S11-15
16.. Chiu TM, Chu SY, Hypersensitivity reactions to iodinated contrast media: Biomedicines, 2022; 10(5); 1036
17.. Kodzwa R, ACR Manual on contrast media: 2018 updates: Radiol Technol, 2019; 91(1); 97-100
18.. Wood DL, Osborn MJ, Rooke J, Holmes DR, Amiodarone pulmonary toxicity: Report of two cases associated with rapidly progressive fatal adult respiratory distress syndrome after pulmonary angiography: Mayo Clin Proc, 1985; 60(9); 601-3
19.. Ugajin M, Ishiguro N, Kani H, Hattori H, A case of alveolar haemorrhage induced by iodinated contrast medium use: Eur J Case Rep Intern Med, 2020; 7(9); 001744
Figures
Tables
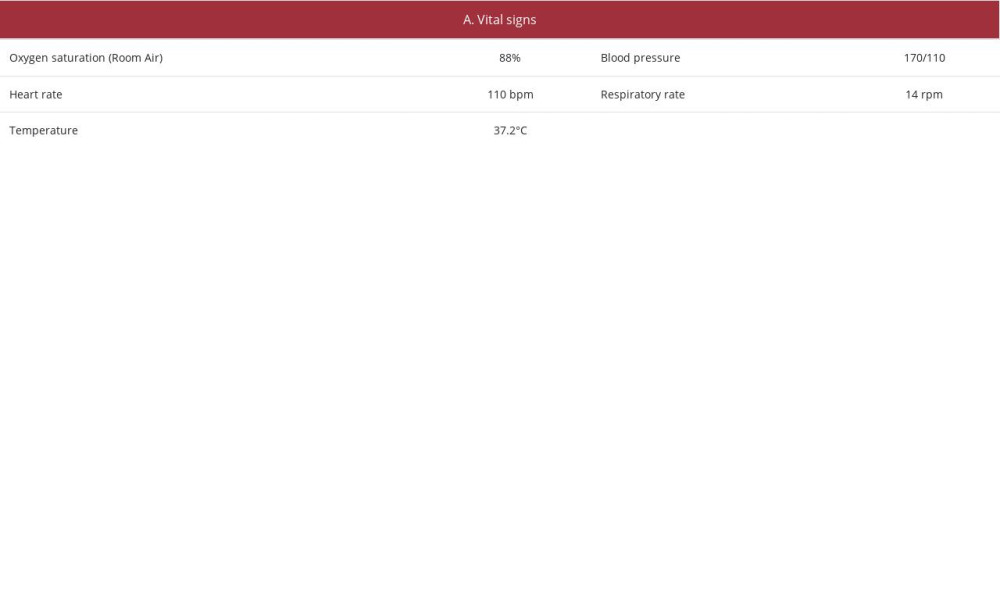 Table 1.. Patient’s parameters and laboratory evaluation at first hemoptysis episode.
Table 1.. Patient’s parameters and laboratory evaluation at first hemoptysis episode.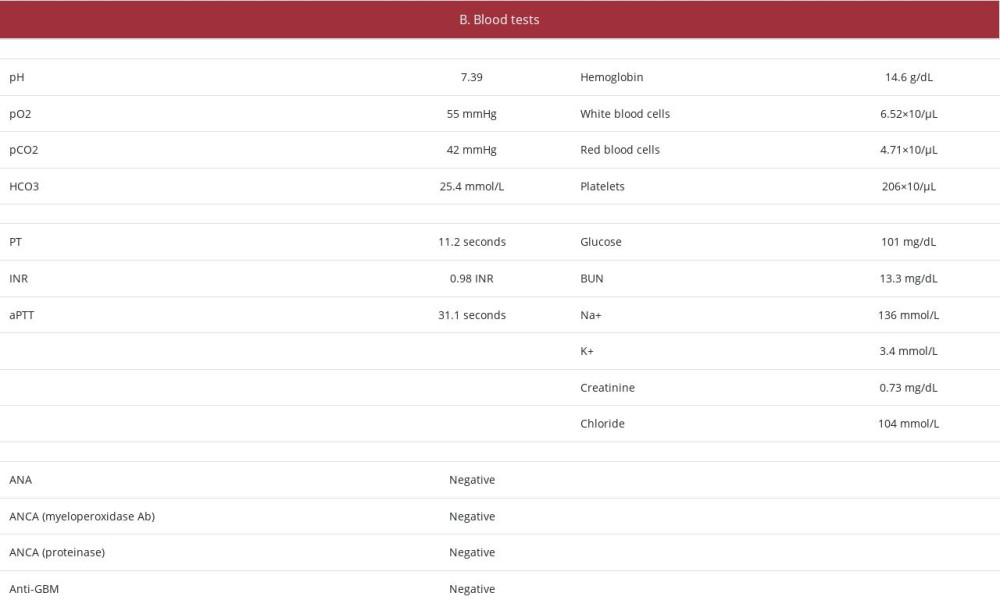 .
.  .
. 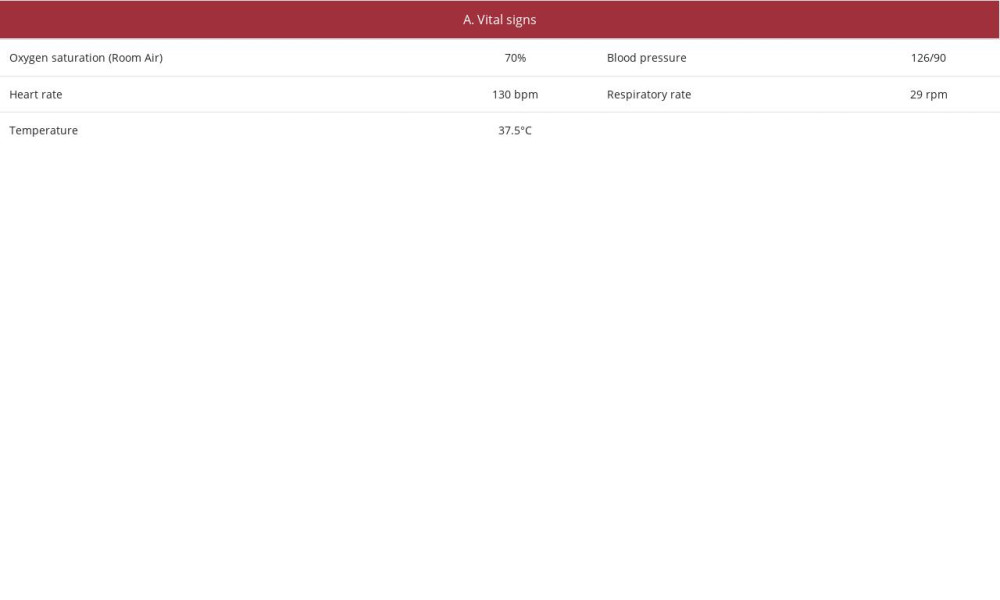 Table 2.. Patient’s parameters and laboratory evaluation at second hemoptysis episode
Table 2.. Patient’s parameters and laboratory evaluation at second hemoptysis episode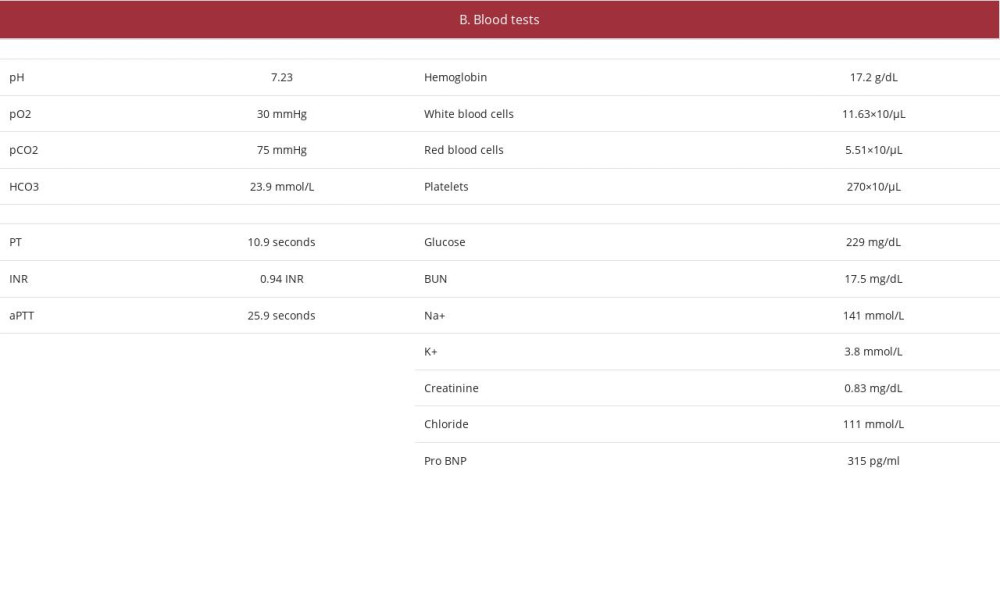 .
.  .
.  Table 1.. Patient’s parameters and laboratory evaluation at first hemoptysis episode.
Table 1.. Patient’s parameters and laboratory evaluation at first hemoptysis episode. .
.  .
.  Table 2.. Patient’s parameters and laboratory evaluation at second hemoptysis episode
Table 2.. Patient’s parameters and laboratory evaluation at second hemoptysis episode .
.  .
. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








