19 September 2021: Articles 
Insulin-Induced Edema in a Patient with Type 2 Diabetes Mellitus
Challenging differential diagnosis, Unusual or unexpected effect of treatment, Diagnostic / therapeutic accidents, Unexpected drug reaction, Rare disease, Clinical situation which can not be reproduced for ethical reasons
Mohammed E. Elmahal1ABEF, Mahmoud M. Ramadan2BF*DOI: 10.12659/AJCR.931960
Am J Case Rep 2021; 22:e931960
Abstract
BACKGROUND: The strong association between type 2 diabetes and obesity has been well recognized. Insulin treatment is usually needed at some point in the treatment of patients with type 2 diabetes and obesity to achieve the targeted glycemic control goal.
CASE REPORT: A 35-year-old woman who had gastric banding for morbid obesity 1 year prior presented to the outpatient clinic with recent onset of shortness of breath and bilateral ankle edema, which were thought to be of cardiac origin. Band slippage occurred a few months after the procedure, and the patient gained weight rapidly thereafter. She had been diagnosed with type 2 diabetes mellitus 8 years earlier, which was treated with oral hypoglycemic medication. Thyroid function tests done 4 months prior to her current presentation revealed normal results. A cardiac assessment showed normal cardiac function with no evidence of heart failure. Repeated thyroid function testing showed new-onset subclinical hypothyroidism. The patient had started insulin therapy 2 weeks before her current presentation, which was probably the main cause of her edema. While continuing insulin, the patient’s general condition improved with diuretics therapy, following which she had gastric bypass surgery.
CONCLUSIONS: Insulin-induced edema is an under-diagnosed problem. It should be considered whenever a patient with uncontrolled diabetes develops rapid body swelling after initiation of insulin therapy. Rigorous research is needed to explore the pathophysiology, prevalence, and treatment of insulin-induced edema.
Keywords: Bariatric Surgery, Diabetes Mellitus, Obesity, Edema, Diabetes Mellitus, Type 2, Female, Humans, Hypoglycemic Agents, Insulin, Obesity, Morbid
Background
Edema, peripheral or generalized, is a rare complication of insulin therapy, which mostly occurs after initiating intensive insulin treatment in patients with newly diagnosed or poorly controlled diabetes [1,2]. Cases of this rare condition have continued to emerge even in recent years [3–9], despite vast improvements in the chemistry and manufacture of insulin molecules.
We present here a rare case showing the overlap of symptoms and signs in a patient with obesity and diabetes who started insulin therapy. Of note, the cause of weight gain in this patient could be intuitively explained by the failure of gastric banding; however, this should not prevent searching for other possible causes of generalized edema. Since insulin-induced edema is a diagnosis of exclusion, it can take a relatively long time to be established. Nevertheless, it should always be considered when no obvious cause of edema is found in such patients.
Case Report
A 35-year-old woman was admitted to the general medical ward with generalized body swelling occurring over a few days. She had morbid obesity (body mass index of 54.8 kg/m2) and had been diagnosed with hypertension and type 2 diabetes mellitus 8 years before, but her diabetic state was poorly controlled during the last year. Gastric banding was performed 1 year prior to her presentation; however, the procedure failed a few months later because of band slippage. The failure of gastric banding combined with poorly controlled diabetes, hypertension, and morbid obesity had necessitated the consideration of gastric bypass surgery.
The patient had been visiting the medical outpatient clinic regularly for routine investigations and revision of her medications. At her last visit, 2 weeks prior to presentation, her medications were metformin 1g twice daily, gliclazide 120 mg (modified release tablets) once daily before breakfast, sitagliptin 50 mg once daily, valsartan 160 mg once daily, rosuvastatin 10 mg once daily at bedtime, and ranitidine 300 mg once daily. Because her HbA1c was 12.3%, she was started on insulin glargine 50 units at bedtime in an attempt to achieve satisfactory glycemic control. The patient admitted that she was feeling well before starting insulin therapy, after which she immediately started to observe puffiness of her body and legs. She had no known allergy to specific drugs or allergens and was not on oral contraceptives. She was a stay-at-home mother with 1 child, a 7-year-old son, living with her. She did not smoke or drink alcohol. Her mother had obesity, with longstanding but well-controlled diabetes mellitus.
On examination, she weighed 130 kg; however, her weight 2 weeks earlier was 122 kg. She looked slightly dyspneic, lying on the bed with 2 pillows under her head, with a respiratory rate of 20 breaths/min and oxygen saturation of 95% on ambient air. Her pulse rate was 108 beats/min (regular) and blood pressure was 146/85 mmHg. Her jugular venous pressure was not visible when she was lying at 45 degrees. Her first and second heart sounds were clearly audible, with no additional sounds. However, the patient had generalized pitting body edema, which was more evident in her lower limbs. There were neither visible varicose veins nor cutaneous changes, and her edematous legs were neither hot nor tender to palpation. Breath sounds were normal to auscultation, with no audible crackles. The abdomen was soft to palpation, the liver was palpable 2 cm below the costal margin without tenderness, and there was no distension, shifting dullness, or guarding of the abdomen. The neurological examination was within normal limits.
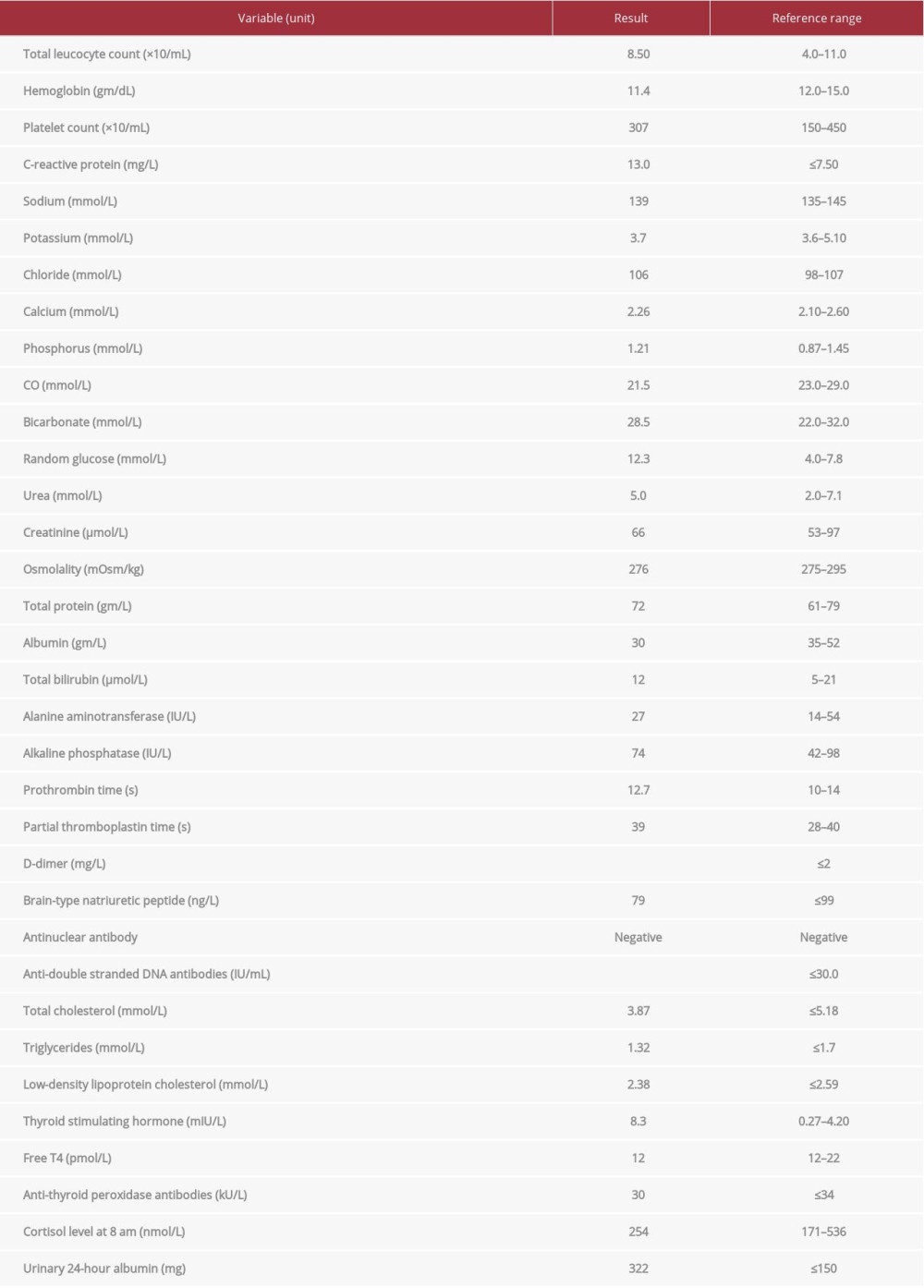
As the patient’s main symptom was generalized body swelling, full laboratory investigations were done, revealing the results shown in Table 1. The chest X-ray showed no evidence of congestive heart failure, cardiomegaly, or pericardial disease. There were no significant electrocardiographic changes. Trans-thoracic echocardiography as well as Duplex ultrasound of the lower limbs showed normal results.
The differential diagnoses of generalized edema considered in this patient included the following: congestive heart failure; constrictive pericarditis; obstructive sleep apnea with pulmonary hypertension; renal failure; atypical nephrotic syndrome; hepatic diseases, including liver cirrhosis and Budd-Chiari syndrome; protein losing enteropathy; pretibial myxedema (hypothyroidism); insulin-induced edema; and idiopathic edema.
The cardiac, renal, and hepatic causes were easily excluded from the differential diagnosis based on the clinical examination findings and results of the investigations that were carried out in the preliminary workup. Idiopathic edema usually occurs in women who are premenstrual. Although it is also called cyclic edema, it has no relationship with the menstrual cycle but has a characteristic diurnal pattern with remarkable body weight variations throughout the day [10]. A difference of body weight of more than 1.4 kg in 1 day is considered the key diagnostic feature of this condition. It usually requires diuretic therapy for a relatively long duration and usually recurs upon withholding the treatment [10]. However, a rapid weight gain of 8 kg in 2 weeks after initiation of insulin therapy in our patient, with a complete resolution after a short-term diuretic therapy, could not be attributed to idiopathic edema.
The new onset of generalized pitting edema in this patient with diabetes could not be explained by the encountered hypothyroidism, as the latter manifests by non-pitting edema, which usually occurs in the myxedematous range. However, insulin-induced edema is a pitting edema, which has occurred in a few patients with uncontrolled diabetes who were exposed to intensive insulin therapy [4] and is a self-limiting disorder, which is usually treated with diuretics for a short period [11,12]. Subclinical hypothyroidism in the present case was obviously an epiphenomenon rather than a conditioning factor; in other words, it was a byproduct or secondary phenomenon occurring simultaneously with insulin-induced edema but not directly related to it. Moreover, the edema subsided with diuretic therapy, specifically without having to use thyroxine.
Protein-losing enteropathy was excluded from the history because the patient had normal stool consistency with absent symptoms of diarrhea, bloating, abdominal pain, or changes in bowel habits.
The patient was initially treated conservatively with a loop diuretic (frusemide 40 mg intravenous injection twice daily), salt restriction, prophylactic low-molecular weight heparin (enoxaparin 40 mg subcutaneous injection once daily), and pantoprazole tablets 40 mg daily. The other medications that the patient had been taking were continued regularly, including insulin.
One week after admission, the edema subsided without the need of further diuretic treatment, and the patient felt well. She was weighed daily, and a reduction of her body weight to 123 kg occurred and remained stable without additional diuretic therapy. Repeated laboratory investigations showed no significant changes compared with index admission, but her albumin level increased from 30 to 37 gm/L. A diagnosis of insulin-induced edema was made, with a probable diagnosis of new-onset subclinical hypothyroidism. However, thyroxine therapy was not started during her hospital stay. A multidisciplinary team, consisting of an endocrinologist, cardiologist, dietitian, bariatric surgeon, and psychologist, was assigned to prepare the patient for bariatric surgery, when the patient’s condition allowed.
Five months later, the patient underwent Roux-en-Y gastric bypass surgery. Thereafter, she stopped receiving insulin therapy, as her body weight started to decrease, and her last weight measurement 2 weeks prior to her last visit was 103 kg. She was maintained on thyroxine tablets 75 microgram once daily after confirmation of hypothyroidism, with a TSH level exceeding 10 mIU/L on repeated tests. Her glycemic control seemed to improve, with a HbA1c of 7.4%. A schematic presentation of the timeline of the various pathologies, interventions and outcomes are shown in Figure 1.
Discussion
In the present case of a female patient with a history of poorly controlled diabetes, insulin-induced edema was the best acceptable explanation of the rapid onset of edema that occurred immediately after the initiation of insulin therapy. Interestingly, most of the reported cases with insulin-induced edema were in female patients with underweight, while this patient had morbid obesity.
Insulin-induced edema (or insulin edema) is a rare condition with a development of rapid generalized edema immediately after receiving insulin therapy. It commonly occurs in individuals who are newly diagnosed with diabetes or in those with uncontrolled type 2 diabetes; it is usually a self-limiting condition but can recur or progress to frank congestive heart failure [11]. It tends to occur in Black women with underweight and can be associated with low albumin levels and/or de-ranged liver function [3,13]. It can also occur in patients with type 1 diabetes when they start insulin therapy, regardless of their glycemic control. It was first described by Leifer in 1928 in a middle-aged man who presented with diabetic ketoacidosis. Leifer suggested that prompt fluid retention is mainly due to the rapid deposition of glycogen in response to initiation of insulin therapy [2].
The pathophysiology of insulin-induced edema has not been fully understood, and it seems that this disorder is under-reported, as observed by the limited number of published reports. It is proposed that the anti-natriuretic effect of insulin plays an important role in the mechanism of fluid retention through its significant influence on the transport of electrolytes in the kidney [14]. It appears that it increases tubular sodium retention in the kidney, and transient hyperaldosteronism coupled with hyperglycemia might enhance this effect, particularly in patients with uncontrolled diabetes [9]. This anti-natriuretic action of insulin as a salt-retaining hormone is now unambiguous. It is associated with obesity and hypertension through its direct effect on extracellular volume expansion, leading eventually to volume-loading hypertension [2,14]. Another theory is that insulin-induced edema may be due to a genetic disorders, particularly mitochondrial disorders, such as mitochondrial tRNA mutation at position 3243 [14]. However, the results of these observations are inconclusive, and further studies are needed to confirm or rule out these associations.
There is no consensus on how to treat insulin-induced edema. Most physicians and endocrinologists adopt a conservative management approach, and most cases respond to this method of treatment. This could be mainly because the majority of cases are diagnosed retrospectively, after exclusion of other disorders with a similar clinical picture. Fluid restriction is important and can be the only treatment measure that is needed in some cases.
Diuretics have been used in the treatment of insulin-induced edema with good results in the majority of patients. Their use could be justified by the fact that insulin is a salt-retaining drug; however, clear evidence is lacking. Some endocrinologists could justify the use of aldosterone antagonists, which is supported by the presence of transient hyperaldosteronism, although loop diuretics usually suffice. However, ephedrine showed promising results in the treatment of insulin-induced edema. Despite being a vasoconstrictor and sympathomimetic agent, it can be superior to diuretic therapy in selected cases [9].
Conclusions
Insulin-induced edema is an under-diagnosed condition. It should be thought of whenever a patient with uncontrolled diabetes develops rapid body swelling after the initiation of insulin therapy. Rigorous research is needed to explore the patho-physiology, prevalence, and treatment of insulin-induced edema.
References:
1.. Hopkins DF, Cotton SJ, Williams G, Effective treatment of insulin-induced edema using ephedrine: Diabetes Care, 1993; 16; 1026-28
2.. Baş VN, Çetinkaya S, Ağladıoğlu SY, Insulin oedema in newly diagnosed type 1 diabetes mellitus: J Clin Res Pediatr Endocrinol, 2010; 2; 46-48
3.. Wong M, Balakrishnan T, Anasarca in newly diagnosed type 1 diabetes: Review of the pathophysiology of insulin edema: Cureus, 2020; 12; e7234
4.. Kan Y, Anno T, Kawasaki F, Takai M, Bilateral lower limb edema induced by rapid improvement of glycemic control with insulin therapy in a subject with poorly controlled type 2 diabetes: Acta Diabetol, 2017; 54; 611-13
5.. Amrapala A, Chowdhury TA, Severe oedema in a patient with diabetes: Clin Med (Lond), 2019; 19; 325-26
6.. Tan GH, My shoes are too small for me to fit in!: Arch Dis Child Educ Pract Ed, 2019; 104; 250-51
7.. Chaker F, Yazid M, Chihaoui M, Insulin-induced edema: An unusual complication in a patient with diabetic ketosis: Tunis Med, 2017; 95; 142-44
8.. Mahévas T, Gobert D, Gatfossé M, Mekinian A, Fain O, [Insulin edema in hepatic glycogenosis]: Rev Med Interne, 2017; 38; 201-3 [in French]
9.. Tufton N, Coppack S, Huda B, An unusual cause of generalized insulin edema and truncal neuropathy: J Med Cases, 2015; 6; 563-65
10.. Dunnigan MG, Henderson JB, Hole D, Pelosi AJ, Unexplained swelling symptoms in women (idiopathic oedema) comprise one component of a common polysymptomatic syndrome: Q J Med, 2004; 97; 755-64
11.. Chelliah A, Burge MR, Insulin edema in the twenty-first century: Review of the existing literature: J Investig Med, 2004; 52; 104-8
12.. Hirshberg B, Muszkat M, Marom T, Shalit M, Natural course of insulin edema: J Endocrinol Invest, 2000; 23; 187-88
13.. Adamo L, Thoelke M, Generalised insulin oedema after intensification of treatment with insulin analogues: BMJ Case Rep, 2013; 2013; bcr2012007037
14.. Brands MW, Manhiani MM, Sodium-retaining effect of insulin in diabetes: Am J Physiol Regul Integr Comp Physiol, 2012; 303; R1101-9
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943118
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942826
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250