24 August 2021: Articles 
Case Report: Surgical Management in Acute Subdural Hematoma Induced by Chronic Myelogenous Leukemia
Unusual clinical course, Challenging differential diagnosis, Management of emergency care, Rare coexistence of disease or pathology
Rana Moshref1ABCDEFG*, Taghreed A. Alsinani1ADE, Asrar Al-Sinani2FGDOI: 10.12659/AJCR.932787
Am J Case Rep 2021; 22:e932787
Abstract
BACKGROUND: Chronic subdural hematoma is defined as collection in the subdural space for more than 3 weeks. The incidence ranges from 1 to 20 per 100 000/year, with increased prevalence in the elderly, in males, and with use of anticoagulants or antiplatelets. Chronic myeloid leukemia has been linked to spontaneous subdural hematoma, with the highest reported cases of 39 patients in Korea, but no management is mentioned in that paper. There are few reported cases of patients who are medically free with no use of anticoagulants, diagnosed as having a subdural hematoma and then later discovered to have chronic myelogenous leukemia.
CASE REPORT: We report the case of a 45-year-old man, with known hypertension and diabetes mellitus type 2, on aspirin weekly, who was referred from another hospital with a history of sudden-onset severe headache not related to trauma. Computed tomography (CT) of the brain done in the other hospital showed a left-side subdural hematoma with midline shift. He underwent left-sided acute subdural hematoma evacuation and decompressive hemicraniectomy. He was started on hydroxyurea for chronic myelogenous leukemia.
CONCLUSIONS: A lesson from this rare case is to look for a differential diagnosis and to keep chronic myelogenous leukemia in mind in patients with leukocytosis. Also, we recommend sending a hematology referral in nontraumatic subdural hematoma. There is no specific treatment or algorithm based on previous research, although from observed data and based on information from the American Heart Association, it is reasonable to perform craniectomy in extra-axial collections of more than 1 cm with midline shift to avoid mortality, as reaching a diagnosis would take too much time.
Keywords: decompressive craniectomy, Hematoma, Subdural, Leukemia, Chronic Disease, Hematoma, Subdural, Acute, Hematoma, Subdural, Chronic, Leukemia, Myelogenous, Chronic, BCR-ABL Positive, United States
Background
Chronic subdural hematoma is defined as a collection in the subdural space for more than 3 weeks [1]. The incidence ranges from 1 to 20 per 100 000/year, with increased prevalence in the elderly, in males, and with use of anticoagulants or antiplatelets [2]. The initial symptoms include headache, dizziness, and motor and sensory deficits [1]. Surgical treatment including evacuation+/– craniectomy is done when thickness is more than 10 mm and there is a midline shift. There is high mortality if treatment is performed for patients over age 70 years and for comatose patients with absent pupillary reflex bilaterally and with pathology in the dominant side [3].
Chronic myeloid leukemia has been linked to spontaneous subdural hematoma, with the highest reported cases of 39 patients in Korea, but no management is mentioned in that paper [4]. Chronic myelogenous leukemia has 3 phases: indolent, accelerated, and terminal blast crisis that is like leukemia. The most common symptoms are weight loss, abdominal pain in the left upper quadrant, with lab results showing leukocytosis [5].
There are few reported cases of patients who are medically free with no use of anticoagulants, diagnosed as having subdural hematoma, and then later discovered to have chronic myelogenous leukemia [6–12]. Evidence shows that thrombocytopenia <50 000 is associated with higher morbidity and mortality and requires aggressive platelets transfusion [13].
We report the case of a patient with leukocytosis who then had double evacuation with craniectomy for subdural hematoma and was later found to have chronic myelogenous leukemia postoperatively determined by lab results.
Case Report
We report the case of a 45-year-old man known to have hypertension and diabetes mellitus type 2, on aspirin weekly (as history has been obtained from children), who was referred from another hospital with a history of sudden-onset severe headache not related to trauma. There was no previous history of convulsions or vomiting. The level of consciousness gradually dropped to Glasgow Coma Score (GCS) of 9/15 when he was rushed to the hospital. A report of computed tomography (CT) brain from another hospital showed a left-side subdural hematoma with midline shift.
There was 7-h time interval between onset of symptoms and presentation to our emergency department. Physical examination in our emergency department showed that the patient was intubated on a mechanical ventilator, on fentanyl 100 mcg/h, midazolam 5 mg/h, pupils 2 mm bilateral and non-reactive, with right-side hemiparesis grade 3/5.
A laboratory work-up upon admission revealed leukocytosis with high-normal coagulation profile (Table 1). Repeated CT brain in our hospital (Figure 1), showed left-sided extra-axial hyperdensity 2 cm in maximum thickness with mass effect and midline shift 1.4 cm, left uncal herniation, and effacement of cortical sulci and left lateral ventricle.
He underwent left-sided acute subdural hematoma evacuation with a big question mark incision on the left side. He was kept in supine position, head tilted to the right side, with left shoulder support. The hair was shaved and the skin was cleaned with antiseptic and draped. A big question mark incision was made, the myocutaneous flap was elevated, and a large bone flap was made with the aim of possible decompressive craniectomy. Then, the dura was opened in a C-shaped fashion with base directed to base, and the hematoma was evacuated. The brain looked healthy and lax, and good hemostasis was achieved with Surgicel, fibrillary Surgicel, and TachoSil. In the end, the dura was closed in watertight fashion, the bone flap was returned as brain was lax, and it was fixed with mini-plates and screws. The scalp was closed in 2 layers, using closed suction, with estimated blood loss 700 mL and duration of the procedure 240 min. He was intubated and sedated, and his pupils were 2 mm and not reactive.
A repeated CT brain after 6 h (Figure 2), showed left- sided craniotomy with pneumocephalus, subgaleal hematoma, and redemonstration of subdural hematoma with midline shift 17 mm and uncal herniation.
He was pushed to the operating room to undergo left subdural hematoma evacuation with craniectomy for recollection of a left-sided acute subdural hematoma. The same question mark incision in the scalp was used, and an old appendectomy incision was used with slight elongation accommodate the bone flap. In the first stage, the patient was kept in supine position with head tilted to the right side with left shoulder support, and the skin was cleaned with antiseptic and draped. The previous question mark incision in the scalp was reopened, a large subgaleal hematoma was evacuated, and the bone flap was removed. A huge extra-axial hematoma was evacuated, the dura was opened, and there was no recollection of subdural hematoma. The brain was found to be visible and pulsating well, and the dura was left open. Long-term hemostasis was needed using Surgicel, fibrillary Surgicel, and TachoSil, and the patient received a perioperative blood transfusion of 6 units platelets and 2 units packed red blood cells (PRBC) as Hb dropped from 11 to 6, as we suspected coagulopathy as a case of the recollecting hematoma. In the second stage, the abdominal skin incision was made, a subcutaneous pocket was created, and the bone flap was kept within. The skin was closed in layers and closed suction was inserted. The estimated blood loss was 150–200 mL and duration of the procedure 120 min. Histopathology of dura mater was not done.
The patient was sedated, intubated, pupils 2 mm, equal and reactive. He was on antiseizure medication: phenytoin 100 mg 3 times daily. The hematology team consulted and the intensive care unit (ICU) were involved.
Laboratory values on day 1 after the second evacuation showed marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes (Table 2). A CT brain showed status postoperative evacuation and left skull craniectomy (Figure 3), significant improvement of left extra-axial hematoma, with improvement of midline shift from 17 mm to 7 mm.
The patient’s GCS started to improve; it was GCS E2V1M3 on day 2 postoperatively and GCS E4V1M6 at the end of 2 weeks postoperatively. In the first week, there was wound oozing, so the suture was taken at the site of the drain. The patient developed 1 attack of fever, so he was started on vancomycin, cefepime, and caspofungin. Cultures showed pansensitive staph and candida in the sputum. The hematology team was highly suspicious of chronic myelogenous leukemia (CML), with blood film showing eosinophilia; therefore, he was started on hydroxyurea 1 g daily oral dose, and a recommendation of PCR ABL gene to be done in a high-level center.
Postoperative clips were removed (both scalp and abdomen) 2 weeks after surgery. There was mild oozing of serosanguinous fluid upon removing the suture, which was controlled by compression and dressing. A repeat CT brain (Figure 4) showed significant interval improvement and newly-seen left temporoparietal extra-axial collection of cerebrospinal fluid (CSF) attenuation, with maximum thickness 3.1 cm.
One month postoperatively, the patient returned to his baseline GCS 15/15 and was extubated, with residual weakness. He sustained residual right-side weakness, spasticity, and left foot drop. The scalp flap was soft, the abdominal wound was malodorous and fluctuating, and the dressing was soaked. As a result, evacuation of infected collection of the previous surgical site in the anterior abdominal wall was achieved with discarding the buried abdominal flap. The patient was kept in supine position and the surgical site was cleaned with antiseptic and draped. The previous incision was opened and a huge amount of slightly malodorous hematoma was removed. The cavity was well cleaned with frequent irrigation with saline and betadine, and good hemostasis was achieved. The skin was closed with single-layer mattress suturing, and closed suction was placed. There was minimal blood loss and the duration of the procedure was 30 min. Cultures were sent, and lab results showed a return to baseline (Table 3).
Discussion
Subdural hematoma usually presents with headaches and decreased level of consciousness. They are treated surgically in severe mass effect and midline shift, and they are usually caused by trauma [1]. Risk factors include use of antiplate-lets, male sex, and elderly [2]. There are 52 reported cases that link chronic myelogenous leukemia with subdural hematoma, with 47 cases of subdural hematoma development in diagnosed leukemic patients [4–13]. These cases reported leukocytosis upon admission [5–7].
However, to the best of our knowledge, there are no reported cases of return of normal WBC count with hydroxyurea. The mortality rate of such cases is 70–100%, with the postulated etiology that the bundle of WBC causes endothelial injury, hypoxia, and hemorrhage to the underlying structure [4,7,8]. There has been 100% mortality reported in age groups 60–70 years [5–7], while 0% mortality is reported in patients 40–55 years [11]. Hematologists recommended in the literature to treat the patients conservatively in the setting of thrombocytopenia, and if surgical management is recommended, they recommend using platelets in emergency settings [4]. We faced some surgical challenges that included uncontrolled oozing, which was managed by electrocautery, antiplatelets, and desmopressin. The decision to perform dural opening was made and decompressive craniectomy was done. A similar case reported emergency double-surgery craniotomy followed by craniectomy managed successfully. The authors found a greenish-red tumor-like mass with brain swelling intraoperatively, with no reported use of hemostatic agents intraoperatively. They kept the patient postoperatively on mannitol and furosemide, with reported improvement of GCS to 13 after the second procedure [8]. Our patient was treated with hydroxyurea after evacuation of the acute subdural hematoma that caused a drop in GCS, in comparison to a case report of chronic subdural hematoma with presentation of headache, but with no thrombocytopenia, that was managed with Gleevec only [11].
Conclusions
A lesson from this rare case is to recommend making a hematology referral in nontraumatic subdural hematoma. It is also important to look for a differential diagnosis and to keep chronic myelogenous leukemia in mind in patients with leukocytosis. There is no specific treatment or algorithm based on previous research, although from observed data [4,7,8] and from the American Heart Association, it is favorable to go to craniectomy in cases with extra-axial collections of more than 1 cm with midline shift [14] to avoid mortality, as reaching a diagnosis would take too much time. Also, we stress the importance of blood products, especially in management of oozing, in the operation room and to involve the ICU staff in the planning.
Figures
Tables
Table 1.. Laboratory values upon admission. Leukocytosis and high-normal coagulation profile.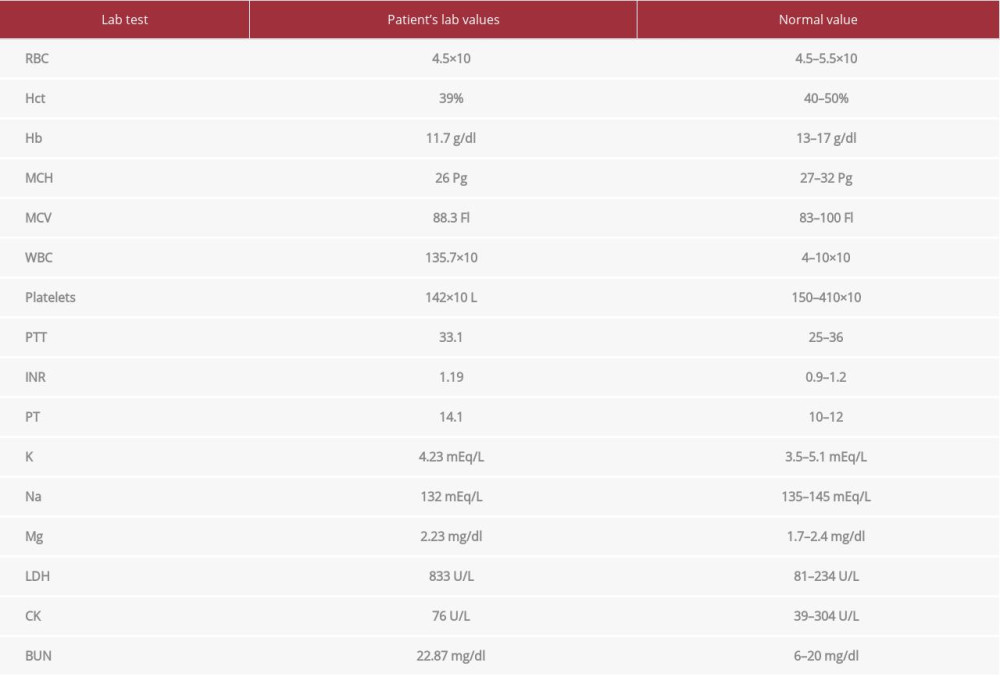 Table 2.. Laboratory values on Day 1 after the second evacuation. Marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes.
Table 2.. Laboratory values on Day 1 after the second evacuation. Marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes.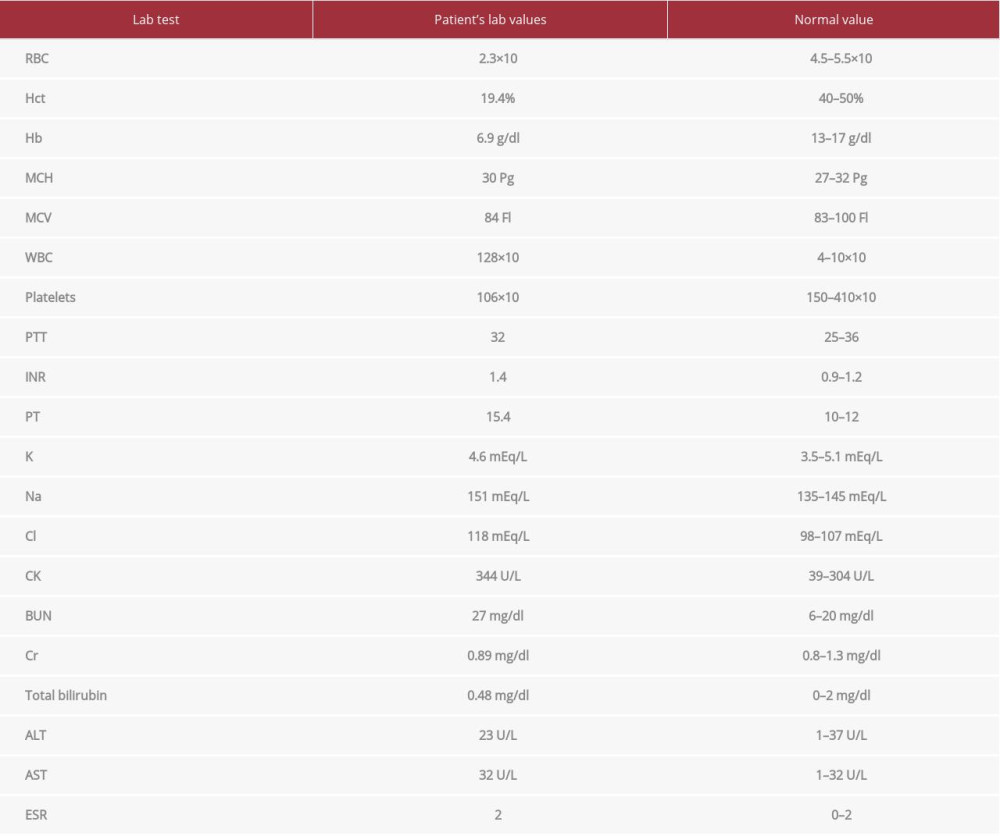 Table 3.. Laboratory values 1 month postoperatively. Return to baseline.
Table 3.. Laboratory values 1 month postoperatively. Return to baseline.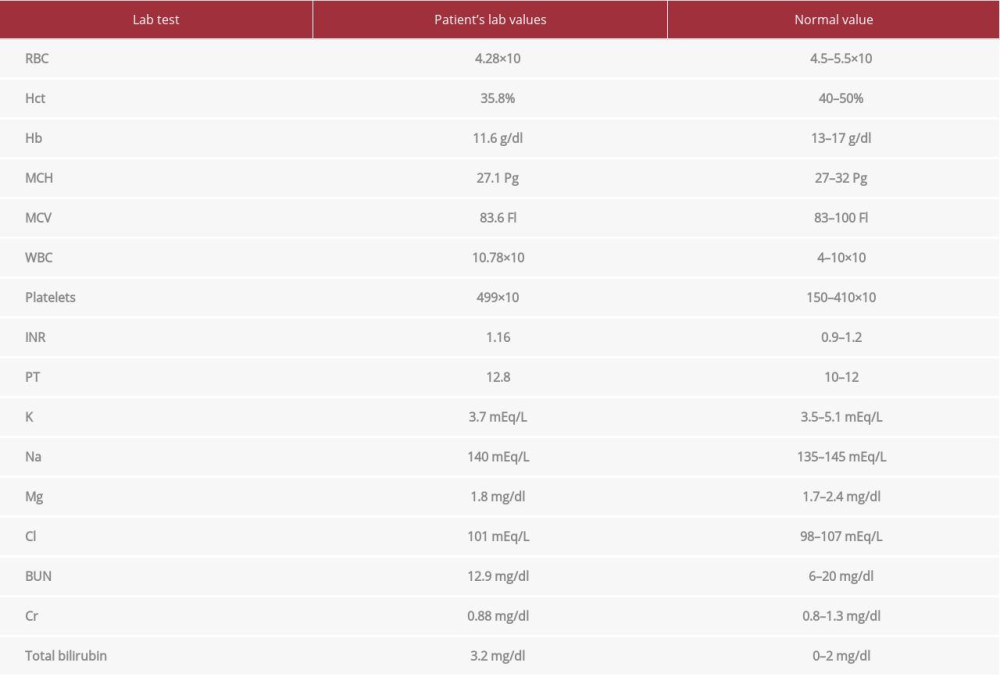
References:
1.. Májovský M, Netuka D, Chronic subdural hematoma – review article: Rozhl Chir, 2018; 97(6); 253-57
2.. Yang W, Huang J, Chronic subdural hematoma: epidemiology and natural history: Neurosurg Clin N Am, 2017; 28(2); 205-10
3.. Bus S, Verbaan D, Kerklaan BJ, Do older patients with acute or subacute subdural hematoma benefit from surgery?: Br J Neurosurg, 2019; 33(1); 51-57
4.. Choi YJ, Rha HK, Park HK, Intracranial hemorrhage in patients with hematologic disorders: J Korean Neurosurg Soc, 2004; 36; 302-5
5.. Sharma SR, Dey B, Blast crisis of chronic myeloid leukemia initially presenting as severe acute intracerebral hemorrhage: J Family Med Prim Care, 2020; 9(2); 1266-69
6.. Ceaușu M, Dragoteanu C, Hostiuc S, Negoi I, Sudden death due to non-traumatic subdural hematoma in a patient with a myeloproliferative disorder: Rom J Leg Med, 2015; 23(4); 251-56
7.. Khaladkar S, Thakkar D, Jantre M, Chronic subdural hematoma-unsu-al cause of headache in a patient with chronic myeloid leukemia treated with high-dose imatinib mesylate: A rare case report with review of liter-ature: Medical Journal of Dr. D.Y. Patil University, 2015; 8(3); 411-13
8.. Wang H, Cao F, Li J, Intracerebral hemorrhage as the initial presentation of chronic myeloid leukemia: A case report and review of the literature: Front Neurol, 2020; 11; 571576
9.. Prasad BC, Chandra VV, Varaprasad G, Dural metastases in chronic myeloid leukemia presenting as subdural hematoma: Turk Neurosurg, 2012; 22(6); 777-78
10.. Raheja A, Satyarthee GD, Mahapatra AK, Chronic subdural hematoma development in accelerated phase of chronic myeloid leukaemia presenting with seizure and rapid progression course with fatal outcome: Romanian Neurosurgery, 2015; 29(2); 196-99
11.. Abdulhamid MM, Li YM, Hall WA, Spontaneous acute subdural hematoma as the initial manifestation of chronic myeloid leukemia: J Neurooncol, 2011; 101(3); 513-16
12.. Jain A, A rare case of chronic myeloid leukemia with acquired von Willebrand disease presenting as subdural hematoma: J Cancer Res Ther, 2015; 11(4); 1022
13.. Chern JJ, Tsung AJ, Humphries W, Clinical outcome of leukemia patients with intracranial hemorrhage: Clinical article. J Neurosurg, 2011; 115(2); 268-72
14.. Naunheim MR, Nahed BV, Walcott BP, Diagnosis of acute lymphoblastic leukemia from intracerebral hemorrhage and blast crisis. A case report and review of the literature: Clin Neurol Neurosurg, 2010; 112(7); 575-77
Figures
Tables
 Table 1.. Laboratory values upon admission. Leukocytosis and high-normal coagulation profile.
Table 1.. Laboratory values upon admission. Leukocytosis and high-normal coagulation profile. Table 2.. Laboratory values on Day 1 after the second evacuation. Marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes.
Table 2.. Laboratory values on Day 1 after the second evacuation. Marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes. Table 3.. Laboratory values 1 month postoperatively. Return to baseline.
Table 3.. Laboratory values 1 month postoperatively. Return to baseline. Table 1.. Laboratory values upon admission. Leukocytosis and high-normal coagulation profile.
Table 1.. Laboratory values upon admission. Leukocytosis and high-normal coagulation profile. Table 2.. Laboratory values on Day 1 after the second evacuation. Marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes.
Table 2.. Laboratory values on Day 1 after the second evacuation. Marked leukocytosis, anemia, thrombocytopenia, and deranged electrolytes. Table 3.. Laboratory values 1 month postoperatively. Return to baseline.
Table 3.. Laboratory values 1 month postoperatively. Return to baseline. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








