29 June 2022: Articles 
Acute Hepatitis with Positive Autoantibodies: A Case of Natalizumab-Induced Early-Onset Liver Injury
Unusual clinical course, Challenging differential diagnosis, Unusual or unexpected effect of treatment, Diagnostic / therapeutic accidents, Unexpected drug reaction, Educational Purpose (only if useful for a systematic review or synthesis)
Marlone Cunha-SilvaDOI: 10.12659/AJCR.936318
Am J Case Rep 2022; 23:e936318
Abstract
BACKGROUND: Natalizumab is an anti-integrin monoclonal antibody used as an alternative treatment regimen for patients with autoimmune disorders, especially multiple sclerosis and Crohn’s disease. Natalizumab-induced liver injury has been rarely reported and may follow the first dose (with increases in liver enzymes usually after 6 or more days), or after multiple doses. In general, it is non-severe acute hepatitis (with a hepatocellular pattern) and autoantibodies can be positive, mainly anti-nuclear and anti-smooth muscle antibodies.
CASE REPORT: We are reporting the case of a 60-year-old woman diagnosed with multiple sclerosis previously treated with interferon-beta, dimethyl fumarate, and fingolimod, who presented jaundice 1 day after the first infusion of natalizumab. She had an early-onset acute hepatitis with aminotransferases levels higher than 1000 IU/L and total bilirubin almost 41 mg/dL. Anti-nuclear and anti-smooth muscle antibodies were positive and the histopathological analysis of the liver showed intrahepatic cholestasis associated with moderate necroinflammatory activity (subacute cholestatic hepatitis) and mild diffuse perisinusoidal fibrosis, which could be compatible with the hypothesis of drug-induced liver injury. The scenario of an autoimmune-like hepatitis led the medical team to start oral prednisone and she progressively improved in clinical and laboratory features. Serum levels of liver enzymes and bilirubin were normal within 3 months and there was no further increase after discontinuation of corticosteroid therapy.
CONCLUSIONS: Physicians should be aware of the risk of early-onset acute hepatitis in patients starting natalizumab, especially women with multiple sclerosis. Treatment with corticosteroid for a few months may be beneficial.
Keywords: Chemical and Drug Induced Liver Injury, Hepatitis, Multiple Sclerosis, Natalizumab, Acute Disease, Autoantibodies, bilirubin, Female, Humans, Liver, Middle Aged
Background
Natalizumab is a monoclonal antibody with selective inhibition of the α-4 subunit of integrins. It is used as an alternative therapeutic regimen for autoimmune disorders, especially multiple sclerosis (MS) and Crohn’s disease [1,2]. Several adverse effects have already been described, such as rash, abdominal discomfort, gastroenteritis, nausea, urinary tract infection, depression, fatigue, and lower and upper respiratory tracts infection. The most feared complication is progressive multifocal leukoencephalopathy, a serious neurological condition possibly associated with JC virus reactivation in the neural system [1–3].
Liver toxicity is not a common event and has an idiosyncratic autoimmune-like pattern, mainly described in subjects with MS. Most of them show signs of liver injury within 6 days following the first dose, or after multiple doses of natalizumab [1,4–6]. We report the case of a female patient who presented natal-izumab-induced early-onset acute hepatitis with positive anti-nuclear and anti-smooth muscle antibodies.
Case Report
A 60-year-old woman was diagnosed with MS at age 52 and had undergone multiple therapies: 1) interferon-beta (interrupted due to skin lesions), 2) dimethyl fumarate (suspended due to drowsiness and weakness), and 3) fingolimod (withdrawn due to treatment failure). The medical staff then decided to perform pulse therapy with methylprednisolone and start natal-izumab (300 mg intravenously).
On the day after the first dose of natalizumab, she reported jaundice with progressive worsening. A week later, she was admitted to the hospital with jaundice, fecal acholia, nausea, and itching. There was no fever, myalgia, diarrhea, or abdominal pain. She denied risky sexual exposure, recent trips, alcohol in-take, use of illicit drugs, other medications, and herbal products.
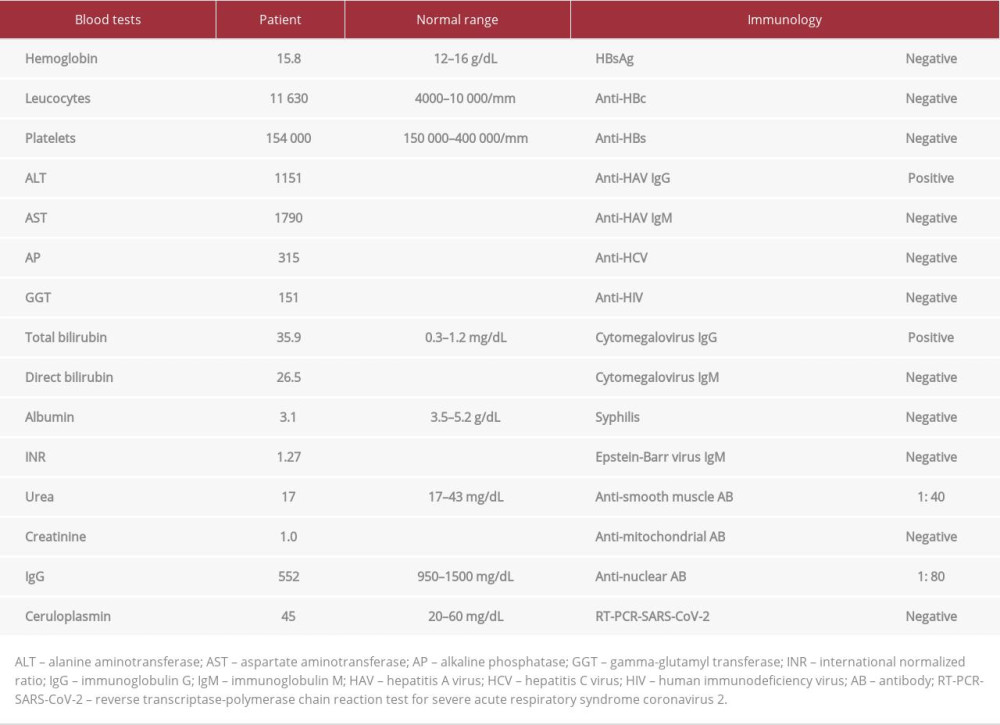
On physical examination, she was overweight (body-mass index=29.3 kg/m2) and there was no flapping or features of chronic liver disease. Laboratory test results are described in Table 1. She had non-severe acute hepatitis with a pattern of hepatocellular injury and positive anti-smooth muscle and anti-nuclear antibodies (cytoplasmic pattern). Abdominal ultra-sound showed a liver with normal morphology, cholelithiasis with no signs of cholecystitis, and no dilatation of the biliary tract. During hospitalization, it was necessary to prescribe cholestyramine and hydroxyzine for pruritus.
The autoimmune hepatitis diagnostic score was 7 points (unlikely diagnosis). Therefore, the main diagnostic hypothesis was natalizumab-induced liver injury. Ten days after hospital admission, serum liver enzymes had a partial improvement: alanine aminotransferase (from 1151 U/L to 540 U/L) and aspar-tate aminotransferase (from 1790 U/L to 794 U/L); but there was an increase in total bilirubin levels (from 35.9 mg/dL to 40.9 mg/dL) (Figure 1). Coagulation tests and platelets count remained normal, so the medical staff performed ultrasound-guided percutaneous liver biopsy and started empirical treatment with oral prednisone 40 mg/day. She was then discharged from the hospital for outpatient follow-up.
There was a progressive improvement in symptoms and laboratory test results (Figure 1) and the dosage of corticosteroid has been progressively reduced in the outpatient follow-up. Histopathological analysis of the liver showed intrahepatic cholestasis associated with moderate necroinflammatory activity (subacute cholestatic hepatitis) and mild diffuse perisinusoidal fibrosis, which could be compatible with the hypothesis of drug-induced liver injury (DILI) (Figure 2). Liver enzymes and bilirubin levels were normal 3 months after starting oral prednisone and there was no subsequent increase after corticosteroid withdrawal.
Discussion
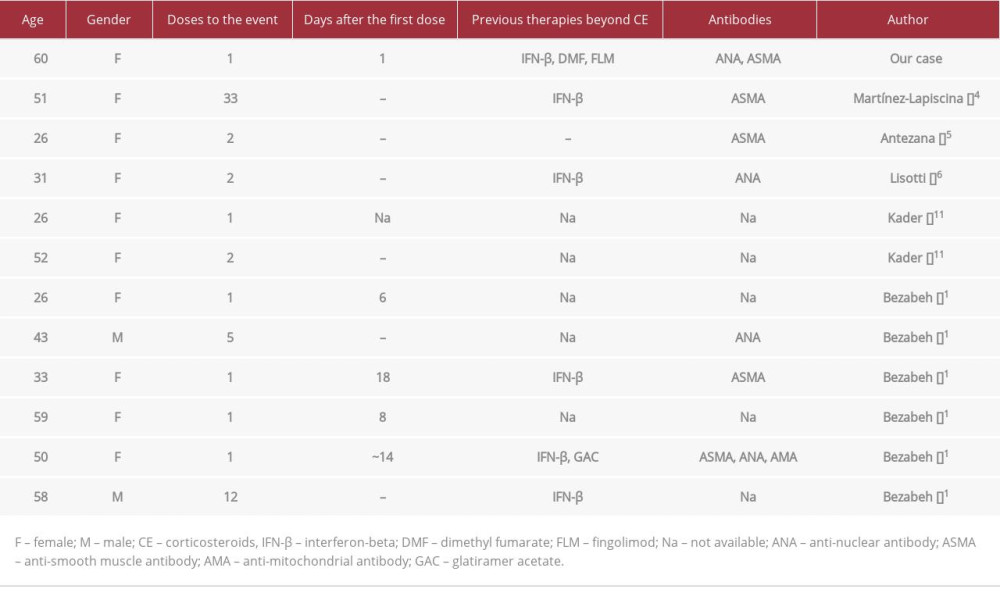
Natalizumab-induced liver toxicity is rare and may occur since the first or second dose or after multiple infusions. It is usually non-severe acute hepatitis and resolves spontaneously [1]. Time to normalization of liver parameters is variable since the discontinuation of natalizumab to up to 6 months later. Re-exposure can lead to earlier and more serious hepatitis [1,4]. In a series of 6 cases, 4 of them started having liver toxicity after the first dose of natalizumab, especially 6 or more days later. Three patients presented positive autoantibodies, mainly anti-nuclear and anti-smooth muscle antibodies [1], similar to our case.
Bezabeh et al mentioned 3 mechanisms for natalizumab-induced liver injury: enhanced activity of autoreactive immune cells; toxicity or hypersensitivity effects caused by the biological agent, or any substance contained in the formulation; and re-infection and/or activation of an atypical pathogen that causes liver damage due to immunosuppression [1]. In these cases, hypersensitivity reactions are usually liver-selective, as systemic symptoms such as fever, rash, urticaria, or eosinophilia have not been reported [1,5,6]. A case of hepatitis B virus reactivation leading to liver failure has already been reported in a patient with MS who started natalizumab [7].
The detection of positive autoantibodies in patients with DILI is not uncommon and has been described with several drugs, including nitrofurantoin, hydralazine, alpha-methyldopa, and amoxicillin/clavulanate [8,9]. It is also possible and even frequent in patients with natalizumab-induced liver injury, and it may become negative after drug discontinuation [1,4–6]. It is important to highlight that some individuals with MS may have positive liver autoantibodies that are investigated only after a suspected event of liver toxicity [10].
Compiling the reported cases, most patients with natalizumab-induced liver injury were women diagnosed with MS and previously treated with interferon-beta, as natalizumab is used as an alternative therapeutic regimen [5,11]. Our patient was older than the mean age described (60 years vs 41 years), and her total bilirubin levels were higher (41 vs 25 mg/dL). Symptoms of liver damage started the day after the first dose (earlier than all reported cases).
Histological changes, as well as clinical, laboratory, immunological features, and the evolution are compatible with DILI. The temporal relationship between the initiation of natalizumab and the problem of jaundice (in the absence of other hepatotoxic drugs) is sufficient to establish the diagnosis of natalizumab-induced liver injury. The summary of previous reports is shown in Table 2.
Corticosteroids are generally not chosen as a treatment for DILI; however, physicians may prescribe prednisone/methylprednisolone or even immunosuppressants for a few months to shorten the recovery period [1,12], especially in an autoimmune environment, as suggested by a recent systematic review [13].
Conclusions
Patients starting natalizumab therapy need to be informed about the symptoms of liver toxicity and failure. Since most cases have been reported after the first or second dose, physicians should request appropriate laboratory exams and be aware of the risk of early-onset acute hepatitis. A good recovery is expected after discontinuation of natalizumab and using corticosteroids.
Figures
References:
1.. Bezabeh S, Flowers CM, Kortepeter C, Clinically significant liver injury in patients treated with natalizumab: Aliment Pharmacol Ther, 2010; 31; 1028-35
2.. Niino M, Bodner C, Simard ML, Natalizumab effects on immune cell responses in multiple sclerosis: Ann Neurol, 2006; 59(5); 748-54
3.. Stüve O, Marra CM, Jerome KR, Immune surveillance in multiple sclerosis patients treated with natalizumab: Ann Neurol, 2006; 59(5); 743-47
4.. Martínez-Lapiscina EH, Lacruz F, Bolado-Concejo F, Natalizumab-induced autoimmune hepatitis in a patient with multiple sclerosis: Mult Scler, 2013; 19(9); 1234-35
5.. Antezana A, Sigal S, Herbert J, Natalizumab-induced hepatic injury: A case report and review of literature: Mult Scler Relat Disord, 2015; 4(6); 495-98
6.. Lisotti A, Azzaroli F, Brillanti S, Severe acute autoimmune hepatitis after natalizumab treatment: Dig Liver Dis, 2012; 44(4); 356-57
7.. Hillen ME, Cook SD, Samanta A, Fatal acute liver failure with hepatitis B virus infection during natalizumab treatment in multiple sclerosis: Neurol Neuroimmunol Neuroinflamm, 2015; 2(2); e72
8.. Lammert C, Zhu C, Lian Y, Exploratory study of autoantibody profiling in drug-induced liver injury with an autoimmune phenotype: Hepatol Commun, 2020; 4(11); 1651-63
9.. Gerussi A, Natalini A, Antonangeli F, Immune-mediated drug-induced liver injury: Immunogenetics and experimental models: Int J Mol Sci, 2021; 22(9); 4557
10.. Tsouris Z, Liaskos C, Dardiotis E, A comprehensive analysis of antigen-specific autoimmune liver disease related autoantibodies in patients with multiple sclerosis: Auto Immun Highlights, 2020; 11(1); 7
11.. Kader ME, Mayes T, Katzman P, Acute liver dysfunction after addition of natalizumab to interferon in two patients with MS: Am J Gastroenterol, 2007; 102(2); 309 abstr. 524
12.. Stine JG, Northup PG, Autoimmune-like drug-induced liver injury: A review and update for the clinician: Expert Opin Drug Metab Toxicol, 2016; 12(11); 1291-301
13.. Björnsson ES, Vucic V, Stirnimann G, Robles-Díaz M, Role of corticosteroids in drug-induced liver injury. A systematic review: Front Pharmacol, 2022; 13; 820724
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133