30 August 2022: Articles 
Severe Traumatic Brain Injury in a Patient with von Willebrand Disease Type 2A Successfully Treated with Factor VIII/von Willebrand Factor Concentrates: A Case Report
Management of emergency care, Rare disease
Takatoshi Koroki1ABCDEF*, Tomohiro Abe1ABCDEF, Sachiyo Kamimura1DE, Hidenobu Ochiai1DEDOI: 10.12659/AJCR.936690
Am J Case Rep 2022; 23:e936690
Abstract
BACKGROUND: von Willebrand disease (VWD) is characterized by a bleeding tendency due to abnormalities in von Willebrand factor (VWF). Severe traumatic brain injury (TBI) can induce secondary coagulopathy and hemostatic disorders. We herein present a rare case of multiple trauma, including severe TBI, in a patient with VWD who was successfully treated with repeated factor VIII/VWF transfusion in addition to standard critical care.
CASE REPORT: A 22-year-old man with type 2A VWD sustained head and lower limb injuries in a traffic accident and was comatose. Computed tomography indicated multiple trauma, including severe TBI (left-sided traumatic epidural hematoma, left-sided traumatic subdural hematoma, traumatic subarachnoid hemorrhage, skull fracture, and skull base fracture). The patient underwent emergency craniotomy for hematoma removal, external decompression, and intracranial pressure monitoring along with massive transfusion and repeated perioperative transfusion of factor VIII/VWF concentrates according to the level of bleeding. He recovered consciousness and eventually survived without neurological deficits.
CONCLUSIONS: Multiple trauma including TBI in patients with VWD is a critical condition. The active transfusion of factor VIII/VWF is essential for controlling hemorrhage early and in the perioperative period.
Keywords: Blood Transfusion, Brain Concussion, Multiple Trauma, von Willebrand Disease, Type 2, Adult, Brain Injuries, Traumatic, Deamino Arginine Vasopressin, Factor VIII, Hematoma, Hemorrhage, Humans, Male, young adult, von Willebrand Diseases, von Willebrand Factor
Background
von Willebrand disease (VWD) is a common genetic bleeding disorder with a reported prevalence of approximately 1% to 2% [1]. This disease is characterized by von Willebrand factor (VWF) abnormalities, which cause abnormal platelet adhesion/ aggregation to the site of angiopathy and a consequent tendency to bleed [2]. There are 3 types of VWD [2]. The current classification includes types 1 and 3, which are characterized by quantitative deficiencies of VWF, as well as types 2A, 2B, 2M, and 2N, which are qualitative variants [2].
Patients with VWD show bleeding symptoms such as epistaxis, oral mucosal petechiae, and prolonged bleeding from injured sites. Patients with a history of severe bleeding, including spontaneous bleeding, long-lasting bleeding, and gastrointestinal bleeding, require prophylaxis with either l-desamino-8-D-arginine vasopressin (DDAVP; desmopressin) or VWF concentrates [2]. For invasive procedures, it is recommended that VWF levels are increased using DDAVP or VWF concentrate before and/or after any procedure is performed [2]. However, treatments for VWD patients with accidental injuries are challenging for clinicians; the appropriate dose of DDAVP or VWF concentrate that is necessary to maintain adequate levels of VWF depends on multiple factors, such as the patient’s individual features, bleeding severity, concomitant injuries, and necessity of invasive procedures.
Severe traumatic brain injury (TBI) is a critical condition with a mortality rate of approximately 40%; furthermore, poor neurological outcomes occur in about 60% of patients [3]. Craniotomy and the removal of hematoma is crucial for patients with intracranial large hematoma, which results in brain hernia. Additionally, TBI often induces coagulopathy, which can cause perioperative hemorrhagic complications [4]. Tranexamic acid, which is an antifibrinolytic agent, is used for patients with trauma. Tranexamic acid reduces injury-related mortality in mild to moderate TBI patients but has no significant benefits in severe TBI patients [5]. Even in patients without any bleeding disorders, severe TBI is a life-threatening condition, especially when there is an intracranial large hematoma that requires surgical intervention.
Thus, the occurrence of multiple trauma including severe TBI in patients with VWD presents a substantial clinical challenge. However, few cases of severe TBI in patients with VWD have been reported to date. We herein present a case of multiple trauma with severe TBI requiring surgical interventions in a patient with type 2A VWD, who was successfully treated with factor VIII/VWF concentrates.
Case Report
A 22-year-old man (weight, 78.9 kg) with a medical history of type 2A VWD sustained injuries to the head and lower limbs in a traffic accident. He had received VIII/VWF concentrates at several times for dental treatments in childhood before the accident, but anti-VWF antibodies had not been tested. His factor VIII and VWF activities at 19 years of age were 22% (normal range, 60–150%) and 6–7% (normal range: 60–170%), respectively. At the accident scene, his blood pressure, pulse rate, and oxygen saturation were 161/79 mmHg, 62 beats/min, and 95% on oxygen at 10 L/min, respectively. His consciousness level was 3 on the Glasgow coma scale, and his pupil diameter was 4.0/4.0 mm (right/left) without reflex to light.
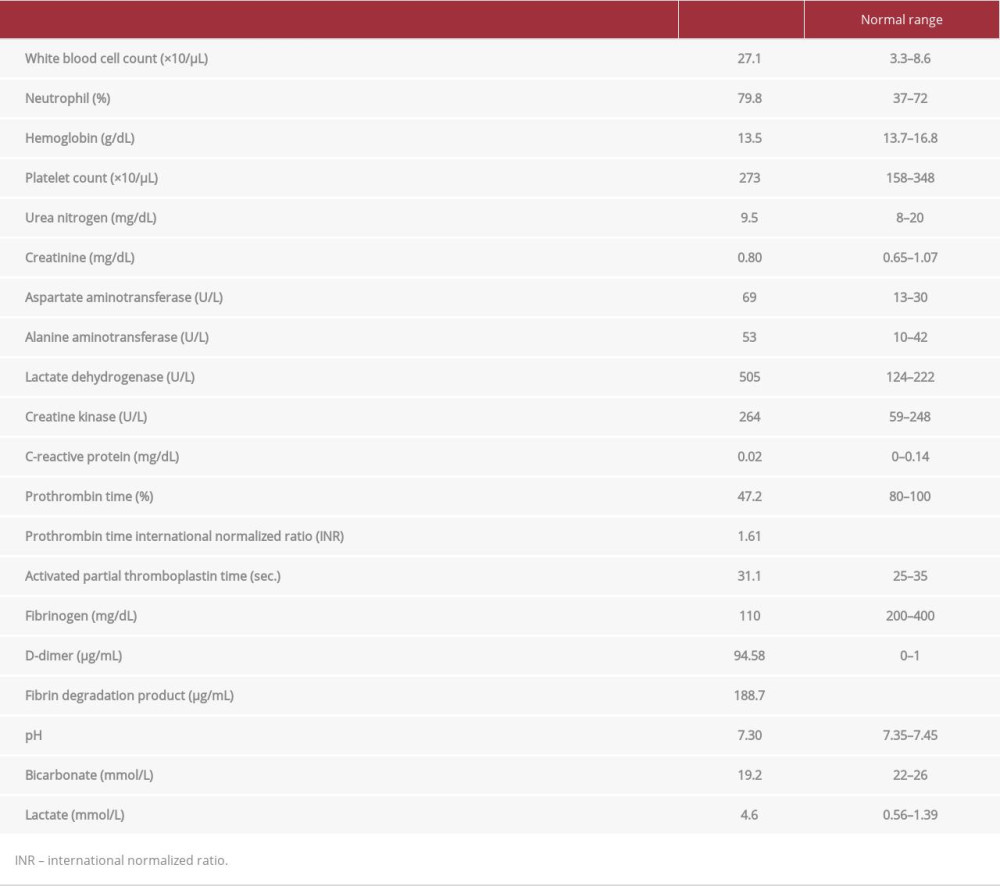
On arrival to the hospital, his blood pressure, pulse rate, respiratory rate, and oxygen saturation were 150/107 mmHg, 108 beats/min, 24 breaths/min, and 98% with oxygen supplementation via reservoir mask, respectively, and his Glasgow coma scale score was 6 (E1V1M4). Physical examination revealed a left head contusion, occipital hematoma, scarring at the forehead, bleeding from the nose and mouth, swelling and deformation on the right side of the lower limb, and no injuries to the trunk. Focused assessment with sonography for trauma showed negative findings. Table 1 shows the laboratory findings on admission.
An X-ray survey revealed right distal tibial and fibular fractures and no abnormal findings on chest and pelvis radiography. Whole-body computed tomography showed a left-sided traumatic epidural hematoma, left-sided traumatic subdural hematoma, traumatic subarachnoid hemorrhage, skull fracture, skull base fracture, pulmonary contusion, and right tibial and fibular fractures (Figure 1). Additionally, the intramuscular pressure on the right side of the lower leg was 70 to 80 mmHg, indicating compartment syndrome. We diagnosed severe TBI, pulmonary contusion, extremity trauma, and compartment syndrome of the lower leg (injury severity score, 38). We administered 1 g of tranexamic acid and transfused 500 units of factor VIII/VWF concentrates along with 3 packs of cryoprecipitate at the Emergency Department.
Emergency craniotomy to remove the hematoma, external decompression, and intracranial pressure monitor insertion were performed in the operating room (Figure 1). Fasciotomy and external fixation were performed for the extremity injury. The bleeding volume was 1823 mL during the operation. Because we could not use thromboelastographic monitoring, we monitored the hemoglobin concentration by blood gas analysis as an indicator for red cell concentrate transfusion, the fibrinogen concentration as an indicator for fresh frozen plasma (FFP), and the patient’s vital signs. Consequently, we transfused 4000 units of factor VIII/VWF concentrates, 14 units of red cell concentrate, and 16 units of FFP during the operation (Figure 2).
After hospital admission, red cell concentrate, FFP, and platelet concentrate were transfused. We also transfused factor VIII/VWF concentrates (4000 units/24 h on days 1-3 and 4000 units/12 h on days 4 and 5). After factor VIII/VWF transfusion on day 3, the patient’s VIII and VWF activities were 140% and 272%, respectively. Because we could not immediately obtain the results of VIII/VWF activity, we transfused factor VIII/VWF in accordance with bleeding from the scars (2000 units of factor VIII/VWF every 12 h on days 6–8, 2000 units every 24 h on day 9, and 3000 units every 24 h on day 12) (Figure 2). Tracheostomy was performed on day 7 because of disturbances in consciousness and prolonged mechanical ventilation. The patient’s respiratory state improved, and he did not need ventilator support on day 13 (Figure 2). His level of consciousness gradually improved, and he was able to communicate. On day 21, we performed internal fixation of the right distal tibial and fibular fractures along with perioperative transfusion of factor VIII/VWF concentrates (4000 units/12 h from days 21 to 25). The patient was able to eat orally on day 24 postoperatively. Magnetic resonance imaging on day 27 showed a brain contusion and diffuse axonal injury. On day 35, we performed cranioplasty using the patient’s own bone flap after cryopreservation and a fixed titanium plate. This was accompanied by the perioperative transfusion of factor VIII/VWF concentrates (4000 units/12 h from days 34 to 37, 4000 plus 2000 units on day 38, and 2000 units on day 39) (Figure 3). The postoperative course was uneventful, and neither thrombotic nor hemorrhagic complications occurred. The patient was eventually discharged from our hospital on day 59.
Discussion
We have presented a case of severe TBI with VWD in which the bleeding was successfully controlled with repeated transfusions of factor VIII/VWF concentrates in addition to standard critical care. The patient survived with a good neurological outcome and was able to integrate into society. To the best of our knowledge, there are few previous reports of multiple trauma with severe TBI in patients with VWD. Notably, VWF is important in both the primary and secondary stages of hemostasis; it is also a known biomarker of endothelial injury, and VWF levels increase in response to many different stimuli [6]. A recent study has proposed that an elevated serum level of VWF suggests endothelial activation in severe head injury [6]. Furthermore, increased VWF following severe TBI may be a marker of unfavorable outcome [7]. However, the interpretation of VWF activity after brain injury in patients with VWD may be unreliable as a marker of head injury.
A recent meta-analysis revealed coagulopathy during the hospital course in 32.7% (range: 10–97.3%) of patients with TBI [8]. Early and rapid correction of traumatic coagulopathy may lead to reduced bleeding, lower transfusion requirements, and improved survival [9]. In the present case, we performed massive transfusion to improve the patient’s coagulopathy and fibrinogen levels. Although FFP and cryoprecipitate can restore fibrinogen and factor VIII/VWF levels to some degree [10], neither is an adequate treatment for patients with VWD.
Some guidelines have proposed that the levels of VWF and factor VIII activities should be maintained at ≥50% (0.5 IU/mL) in patients with severe bleeding or major surgery [2,11,12]. Two main agents that can increase these activities are VIII/VWF concentrates and DDAVP. Of the 2, DDAVP produces a smaller increase in VWF activity and has a later onset and shorter duration of action [3]; therefore, it may not be useful for severe trauma in patients with VWD. However, VIII/VWF concentrates have been demonstrated to provide excellent to good hemostasis in a number of different patient populations and VWD types [13]. In the current case, we successfully treated the patient using factor VIII/VWF concentrates during the acute and perioperative periods. Thus, patients with VWD who sustain severe trauma may benefit from receiving factor VIII/VWF concentrates in addition to the standard transfusion strategy.
The appropriate dose and timing of factor VIII/VWF concentrates that is required to maintain adequate levels of coagulation factors can vary depending on trauma severity, concomitant injuries, and medical and/or surgical treatments. Moreover, the results of factor VIII and VWF activities cannot be promptly obtained. We therefore actively administered factor VIII/VWF concentrates according to the patient’s clinical symptoms, blood examination findings, and imaging findings; this resulted in hemostasis and the patient’s recovery.
In the present case, factor VIII and VWF activities markedly increased after the transfusion of VIII/VWF concentrates. Excessively increased factor VIII and VWF activities are a risk factor for venous thrombosis [14,15]; however, no adverse events occurred in association with the factor VIII/VWF concentrates in the present case. For the optimal transfusion of VIII/VWF concentrates, prompt testing of factor VIII and VWF would therefore be beneficial for the treatment of patients with VWD who sustain trauma; alternatively, thromboelastography can be used for monitoring [16].
We were able to quickly manage and treat the current case because we knew the patient had VWD before his arrival. Trauma patients with known bleeding disorders should be transported to facilities that are able to provide appropriate treatment. If patients with undiagnosed bleeding disorders are injured and transported, they may initially be treated as severe trauma cases. However, when patients reveal a tendency to bleed or show coagulopathy over time, a hematologist should be consulted and the patient should be examined for underlying bleeding disorders so that crucial treatment can be provided [17].
Conclusions
We presented a case of severe trauma with TBI in a patient with VWD 2A. Multiple trauma with severe TBI in patients with VWD is a critical condition both because brain injury-induced coagulopathy can add to the underlying VWD, and because of the need for surgical interventions. We successfully treated our patient with VIII/VWF concentrate transfusion early and in the perioperative period. When critical bleeding occurs in patients with bleeding disorders, the active transfusion of deficient coagulation factors is essential for controlling the bleeding and may be a lifesaving procedure.
Figures
References:
1.. Castaman G, How I treat von Willebrand disease: Thromb. Res, 2020; 196; 618-25
2.. Connell NT, Flood VH, Brignardello-Petersen R, ASH ISTH NHF WFH 2021 guidelines on the management of von Willebrand disease: Blood Adv, 2021; 5(1); 301-25
3.. Rosenfeld JV, Maas AI, Bragge P, Early management of severe traumatic brain injury: Lancet, 2012; 380(9847); 1088-98
4.. Gopalakrishnan MS, Shanbhag NC, Shukla DP, Complications of decompressive craniectomy: Front Neurol, 2018; 9; 977
5.. , Effects of tranexamic acid on death, disability, vascular occlusive events and other morbidities in patients with acute traumatic brain injury (CRASH-3): A randomised, placebo-controlled trial: Lancet, 2019; 394(10210); 1713-23
6.. Yokota H, Naoe Y, Nakabayashi M, Cerebral endothelial injury in severe head injury: The significance of measurements of serum thrombomodulin and the von Willebrand factor: J. Neurotrauma, 2002; 19(9); 1007-15
7.. De Oliveira CO, Reimer AG, Da Rocha AB, Plasma von Willebrand factor levels correlate with clinical outcome of severe traumatic brain injury: J Neurotrauma, 2007; 24(8); 1331-38
8.. Harhangi BS, Kompanje EJ, Leebeek FW, Maas AI, Coagulation disorders after traumatic brain injury: Acta Neurochir (Wien), 2008; 150(2); 165-75 ; discussion 175
9.. Laroche M, Kutcher ME, Huang MC, Coagulopathy after traumatic brain injury: Neurosurgery, 2012; 70(6); 1334-45
10.. O’Shaughnessy DF, Atterbury C, Bolton Maggs P, Guidelines for the use of fresh-frozen plasma, cryoprecipitate and cryosupernatant: Br. J. Haematol, 2004; 126(1); 11-28
11.. : The diagnosis, evaluation, and management of von Willebrand disease, 2007, Bethesda, MD, National Institues of Health, National Heart, Lung and Blood Institute
12.. Satoshi H, Tatsuya A, Akira I, Guidelines for the management of von Willebrand disease 2021: Jpn J Thromb Hemost, 2021; 32; 413-81
13.. Batty P, Chen YH, Bowles L, Safety and efficacy of a von Willebrand factor/factor VIII concentrate (Wilate®): A single-centre experience: Haemophilia, 2014; 20(6); 846-53
14.. Jenkins PV, Rawley O, Smith OP, O’Donnell JS, Elevated factor VIII levels and risk of venous thrombosis: Br J Haematol, 2012; 157(6); 653-63
15.. Edvardsen MS, Hindberg K, Hansen ES, Plasma levels of von Willebrand factor and future risk of incident venous thromboembolism: Blood Adv, 2021; 5(1); 224-32
16.. Regling K, Kakulavarapu S, Thomas R, Utility of thromboelastography for the diagnosis of von Willebrand disease: Pediatr Blood Cancer, 2019; 66(7); e27714
17.. Lee A, Emergency management of patients with bleeding disorders: Practical points for the emergency physician: Transfus Apher Sci, 2019; 58(5); 553-62
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133