19 July 2022: Articles 
Infection Associated with COVID-19: A Case Series and Literature Review
Challenging differential diagnosis, Management of emergency care, Patient complains / malpractice, Rare disease, Adverse events of drug therapy, Educational Purpose (only if useful for a systematic review or synthesis)
Kazuhiro Ishikawa1ABCDEF*, Tomoaki NakamuraDOI: 10.12659/AJCR.936889
Am J Case Rep 2022; 23:e936889
Abstract
BACKGROUND: We aimed to identify the risk factors for Stenotrophomonas maltophilia infection in patients with COVID-19.
CASE REPORT: Case 1. A 52-year-old COVID-19-positive woman with systemic lupus erythematosus was administered remdesivir (RDV) and methylprednisolone (mPSL) 1000 mg/day for 3 days, and subsequently administered baricitinib and ceftriaxone. Following respiratory deterioration, she was transferred to the Intensive Care Unit (ICU) and the antibiotics were switched to meropenem (MEPM). Blood and sputum cultures were positive for S. maltophilia. Administration of trimethoprim-sulfamethoxazole (TMP-SMX) showed clinical improvement. Case 2. An 80-year-old COVID-19-positive man was treated with RDV, dexamethasone, and baricitinib. Owing to severe hypoxia, he was transferred to the ICU and MEPM was administered. Sputum culture was positive for S. maltophilia. TMP-SMX administration temporarily improved his symptoms; however, he died from COVID-19-associated invasive aspergillosis. Case 3. A 48-year-old COVID-19-positive man who was mechanically intubated was transferred to our hospital and treated with RDV, mPSL, and piperacillin/tazobactam. Sputum culture revealed S. maltophilia; treatment with TMP-SMX improved his respiratory status. Case 4. An 80-year-old COVID-19-positive man was treated with RDV and dexamethasone. Owing to severe hypoxemia, he was transferred to the ICU and the antibiotics were switched to MEPM. Sputum culture revealed S. maltophilia. Administration of TMX-SMX improved his respiratory status.
CONCLUSIONS: Isolation of S. maltophilia in respiratory specimens of patients with COVID-19 should prompt clinicians to administer treatment for S. maltophilia-associated pneumonia in ICU-admitted patients who have been intubated, have been administered broad-spectrum antibiotics, or have immunocompromised status.
Keywords: COVID-19 breakthrough infections, Stenotrophomonas maltophilia Bacteremia, Stenotrophomonas maltophilia, Healthcare-associated pneumonia, Aged, 80 and over, Anti-Bacterial Agents, COVID-19, Dexamethasone, Female, Gram-Negative Bacterial Infections, Humans, Male, Middle Aged, Stenotrophomonas maltophilia, Trimethoprim, Sulfamethoxazole Drug Combination
Background
Case Reports
CASE 1:
A 52-year-old woman using prednisolone (PSL) and baricitinib (BARI) with a medical history of systemic lupus erythematosus, rheumatoid arthritis, liver cirrhosis, and interstitial pneumonia was admitted to our Emergency Department with high fever and headache. On admission, her vital signs were as follows: clear consciousness; temperature, 37.2°C; blood pressure, 101/77 mmHg; pulse rate, 100 beats/min; respiratory rate, 22 breaths/min; and oxygen saturation, 94% on room air. The patient’s COVID-19 polymerase chain reaction (PCR) test result was positive. Chest computed tomography (CT) revealed bilateral, non-segmental, ground-glass opacities in the diffuse area, and honeycomb appearance associated with interstitial pneumonia in the bilateral dorsal aspects of the inferior lung lobe (Figure 1A–1C). We initiated administration of remdesivir (RDV) and methylprednisolone (mPSL) 1000 mg/day for 3 days followed by mPSL 40 mg/day, BARI 4 mg/day, and ceftriaxone (CTRX) 1 g intravenously (i.v.) every 24 h. On day 4 of hospital admission, her respiratory status worsened, she was moved to the ICU, and a nasal high flow cannula was inserted. We switched the antibiotics to meropenem (MEPM). On day 8, the patient developed a fever of 37.9°C with 90% oxygen saturation with FIO2 60% and nasal high flow gas 40 L/min, and was intubated. Blood and sputum cultures were positive for S. maltophilia (Figure 2A, 2B). Mucoid-type S. maltophilia was detected by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (Biotyper, Bruker Daltonics, Germany). Adjustment to 0.5 McFarland in the mucoid-type S. maltophilia was difficult; thus, susceptibility testing could not be performed. Intravenous trimethoprim-sulfamethoxazole (TMP-SMX) (equivalent to 15 mg/kg/day TMP) was initiated. Her respiratory status improved; however, the Aspergillus galactomannan antigen level was 1.1. We administered voriconazole to treat COVID-19-associated invasive aspergillosis. The patient’s respiratory condition stabilized, although the duration of intubation was extended; consequently, tracheostomy was performed. Sputum culture revealed elimination of S. maltophilia. At the time of writing this review, the patient’s clinical status had improved (Figure 3).
CASE 2:
An 80-year-old man with a medical history of hypertension and acute myocardial infarction status after coronary artery bypass graft was admitted to our Emergency Department with high fever. On admission, the vital signs were as follows: clear consciousness; temperature, 37.6°C; blood pressure, 101/77 mmHg; pulse rate, 98 beats/min; respiratory rate, 22 breaths/min; and oxygen saturation, 96% (10 L/min on reservoir mask). The patient’s COVID-19 PCR result was positive. Chest CT revealed bilateral ground-glass opacity and reticulation with subpleural distribution (Figure 4A, 4B). We initiated administration of RDV, dexamethasone (DEXA) 6.6 mg/day, BARI 4 mg/day, and CTRX 1 g i.v. every 24 h. Owing to severe hypoxemia, he was transferred to the ICU and mechanically intubated, and a CVC was inserted. On day 4 of hospital admission, he developed a fever of 38°C, and antibiotics were switched to piperacillin/ tazobactam (PIPC/TAZ). A sputum culture on day 8 revealed the presence of Candida glabrata (mold) and S. maltophilia; however, we did not administer any treatment for these as we assumed them to be colonizing organisms. The minimum inhibitory concentrations were measured using the MicroScan WalkAway 96 Plus and NC-NF2J panel (Beckman Coulter Inc). Levofloxacin (LVFX), minocycline (MINO), TMP-SMX, and ceftazi-dime were all effective (Table 1). The patient had persistent prominent fever; however, and a blood culture on day 11 indicated C. glabrata infection. We administered micafungin and replaced the CVC. Follow-up blood cultures were negative for C. glabrata; however, the fever did not abate, and on day 18 the patient showed increased hypoxia. Switching antibiotics from PIPC/TAZ to MEPM did not improve the clinical symptoms.
Therefore, we administered TMP-SMX (equivalent to 15 mg/ kg/day TMP) for treating S. maltophilia-associated pneumonia, and the fever resolved. Owing to acute kidney injury, we switched from TMP-SMX to LVFX and MINO. Aspergillus galactomannan sputum antigen was >5.0, and we administered voriconazole. However, the patient’s respiratory status worsened. He died on day 28 of hospital admission due to COVID-19-associated invasive aspergillosis (Figure 5).
CASE 3:
A 48-year-old man with COVID-19 and a medical history of hypertension, diabetes, and obesity was admitted to another hospital, where he was treated with RDV and DEXA. His respiratory condition deteriorated, and he was mechanically intubated and treated with mPSL 1000 mg/day for 3 days. On day 7 following COVID-19 onset, his PaO2/FiO2 ratio was about 80. He was transferred to our hospital for COVID-19 treatment with extracorporeal membrane oxygenation (ECMO). Chest CT revealed bilateral diffuse consolidation (Figure 6). He was treated with RDV, mPSL 125 mg/day, and PIPC/TAZ. His respiratory status stabilized; therefore, ECMO was discontinued, and he was extubated on day 9. On day 10 of hospital admission, he had increased sputum production and his C-reactive protein level increased to 23.7 mg/dL. Chest radiography revealed consolidation in the right lower lung field (Figure 6). The sputum culture revealed the presence of S. maltophilia; therefore, he was treated with TMP-SMX (equivalent to 15 mg/kg/day TMP). By the time of this report, the patient was clinically stable and preparing for transfer to a rehabilitation hospital (Figure 7).
CASE 4:
An 80-year-old man with a medical history of dementia developed COVID-19 four days before admission. The patient was medically monitored at a health care institute. When his O2 saturation level gradually decreased, he was admitted to our hospital. Chest CT revealed ground-glass opacity with subpleural distribution (Figure 8). We started RDV, DEXA 6.6 mg/day, AZM 500 mg/day, and CTRX 1 g i.v. every 24 h. Owing to severe hypoxemia, he was transferred to the ICU and mechanically intubated on day 6 after hospital admission. On day 9, his sputum culture revealed S. maltophilia, Klebsiella aerogenes, and Enterococcus faecalis. Although susceptible to LVFX, MINO, and TMP-SMX, the bacteria were resistant to ceftazidime (Table 2). The patient was treated with LVFX 500 mg/day. On day 18 of hospital admission, he was extubated. However, his sputum production increased, and he was re-intubated on day 34. He was treated with TMP-SMX (equivalent to 15 mg/kg/day TMP) and MEPM. His respiratory status improved, although a tracheostomy had to be performed because of CO2 retention. He was finally transferred to a rehabilitation hospital on day 56 of hospital admission (Figure 9).
Discussion
Two authors independently reviewed the relevant titles and abstracts in the database records, retrieved full texts for eligibility assessment, and extracted the information from these cases. We performed a search using the keywords “
We found 68 articles and 8 case reports on COVID-19-associated
We extracted the data of 8 patients with COVID-19 and positive
The median age of the 12 patients, including the 4 patients in this review, with COVID-19-associated
Among the 13 observational studies (Table 5), 4 were from China, 2 from Italy, 2 from Spain, and 5 from the United States. Nine of the 13 studies were conducted in the ICU (2 studies had mixed ICU and non-ICU patients). Eleven studies included respiratory infections, such as VAP and hospital-acquired pneumonia (HAP). Mortality rates ranged from 15.4% to 52.4%, although we did not find any mortality rate for
Here, we report 4 cases of COVID-19-associated
Among patients without COVID-19, the risk of
There is limited evidence on effective antimicrobials against
This study had some limitations. As VAP and HAP definitions differed among the studies, it was not possible to discern whether
Conclusions
Figures
Tables
Table 1.. MINO, TMP-SMX, LVFX, and CAZ were susceptible to Stenotrophomonas maltophilia.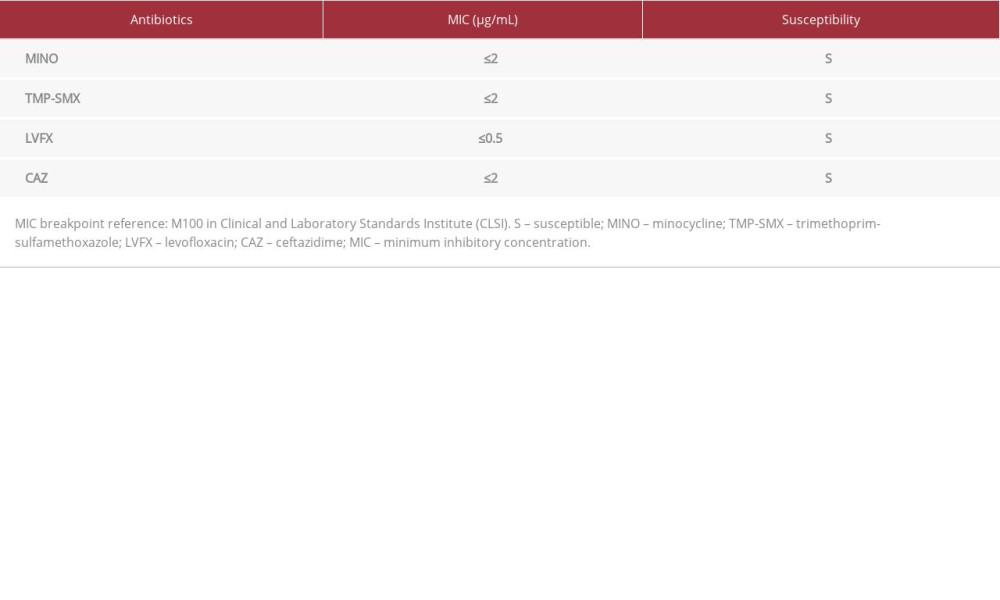 Table 2.. MINO, TMP-SMX, and LVFX were susceptible to Stenotrophomonas maltophilia.
Table 2.. MINO, TMP-SMX, and LVFX were susceptible to Stenotrophomonas maltophilia.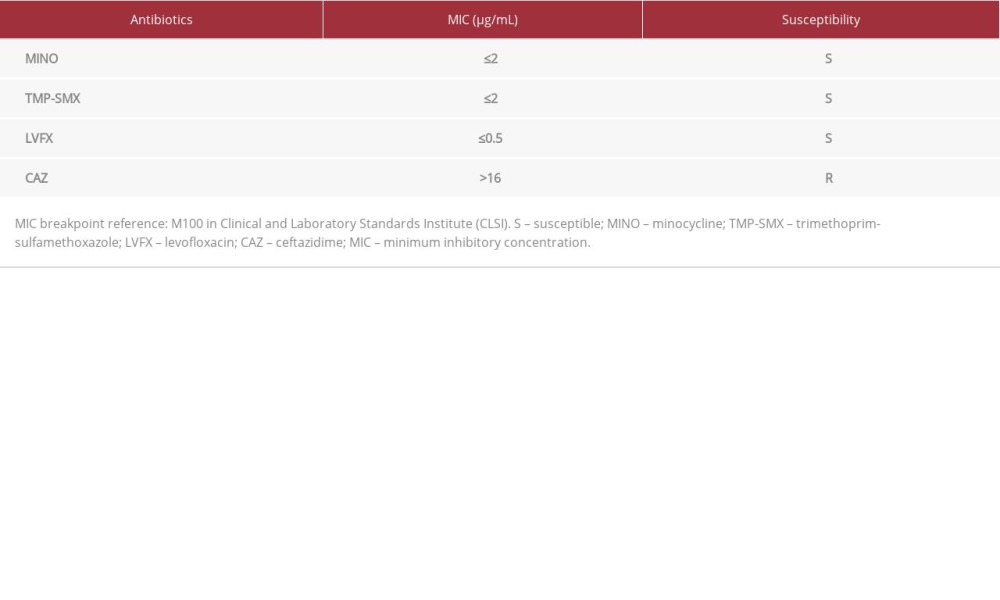 Table 3.. Search terms used to search 3 databases (PubMed, Embase, and Ichushi) for literature review of COVID-19-associated Stenotrophomonas maltophilia.
Table 3.. Search terms used to search 3 databases (PubMed, Embase, and Ichushi) for literature review of COVID-19-associated Stenotrophomonas maltophilia. Table 4.. Literature review of COVID-19-associated Stenotrophomonas maltophilia + cases at our hospital.
Table 4.. Literature review of COVID-19-associated Stenotrophomonas maltophilia + cases at our hospital.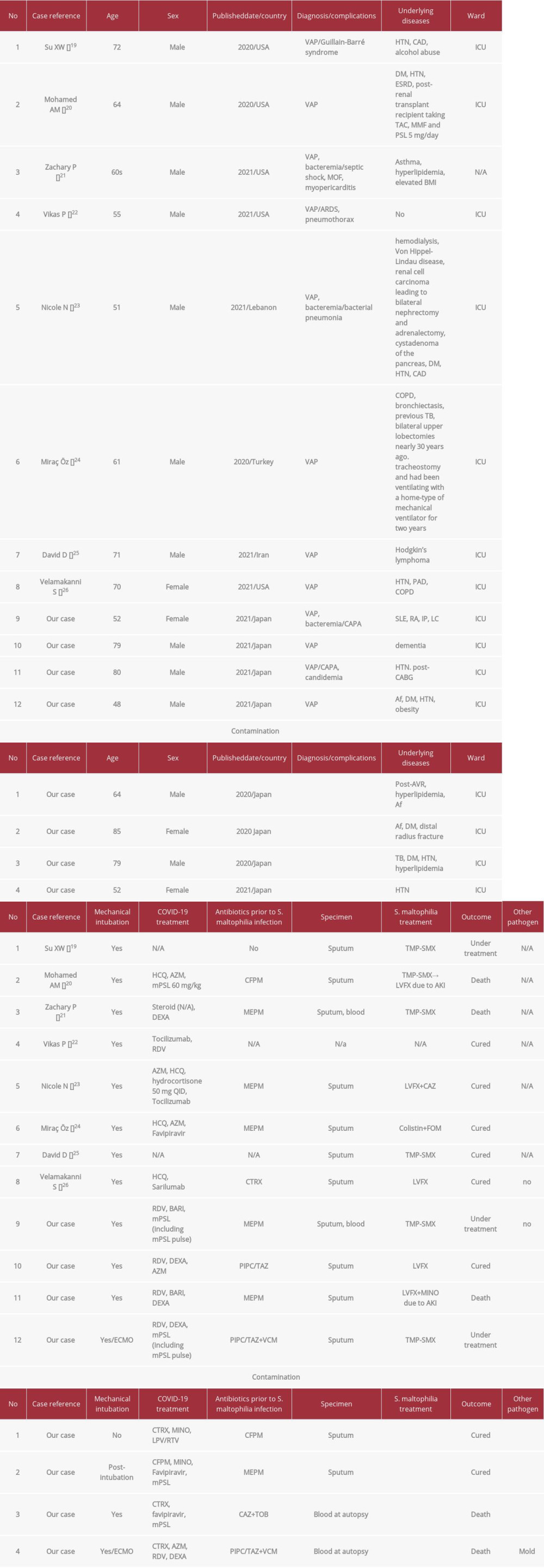 Table 5.. List of retrospective cohort studies on Stenotrophomonas maltophilia secondary infection to COVID-19.
Table 5.. List of retrospective cohort studies on Stenotrophomonas maltophilia secondary infection to COVID-19.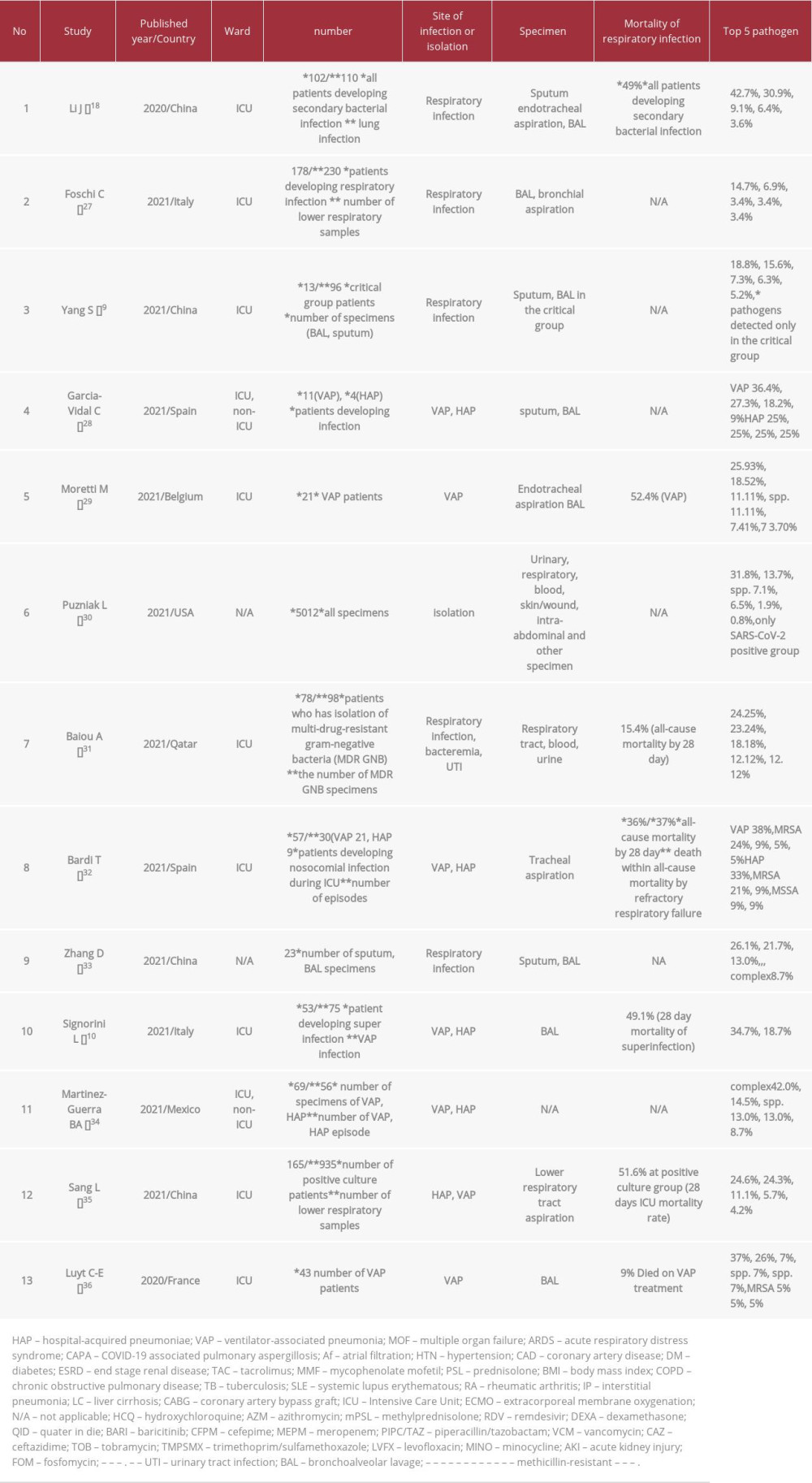
References:
1.. Guyot A, Turton JF, Garner D: J Hosp Infect, 2013; 85; 303-7
2.. Brooke JS: Clin Microbiol Rev, 2012; 25; 2-41
3.. Hotta G, Matsumura Y, Kato K: PLoS One, 2014; 9; e112208
4.. Imoto W, Yamada K, Yamairi K: Intern Med, 2020; 59; 193-98
5.. Imoto W, Yamada K, Kuwabara G: J Hosp Infect, 2021; 111; 169-75
6.. Anderson SW, Stapp JR, Burns JL, Qin X: J Clin Microbiol, 2007; 45; 529-35
7.. Irifune K, Ishida T, Shimoguchi K: J Clin Microbiol, 1994; 32; 2856-57
8.. Guerci P, Bellut H, Mokhtari M: Crit Care, 2019; 23; 371
9.. Yang S, Hua M, Liu X, Bacterial and fungal co-infections among COVID-19 patients in Intensive Care Unit: Microbes Infect, 2021; 23; 104806
10.. Signorini L, Moioli G, Calza S, Epidemiological and clinical characterization of superinfections in critically ill coronavirus disease 2019 patients: Crit Care Explor, 2021; 3; e0430
11.. Shi H, Han X, Jiang N, Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: A descriptive study: Lancet Infect Dis, 2020; 20; 425-34
12.. Kassel TW, Ryan LK, Li A: Multidiscip Respir Med, 2013; 8; 14
13.. Kang YR, Cha YK, Kim JS: Acta Radiol, 2020; 61; 903-9
14.. Gil-Gil T, Martinez JL, Blanco P: Expert Rev Anti Infect Ther, 2020; 18; 335-47
15.. Sader HS, Farrell DJ, Flamm RK, Jones RN, Antimicrobial susceptibility of Gram-negative organisms isolated from patients hospitalised with pneumonia in US and European hospitals: Results from the SENTRY Antimicrobial Surveillance Program, 2009-2012: Int J Antimicrob Agents, 2014; 43; 328-34
16.. Wang YL, Scipione MR, Dubrovskaya Y, Papadopoulos J: Antimicrob Agents Chemother, 2014; 58; 176-82
17.. Wang C-H, Yu C-M, Hsu S-T, Wu R-X: J Hosp Infect, 2020; 104; 46-52
18.. Li J, Wang J, Yang Y, Etiology and antimicrobial resistance of secondary bacterial infections in patients hospitalized with COVID-19 in Wuhan, China: A retrospective analysis: Antimicrob Resist Infect Control, 2020; 9; 153
19.. Su XW, Palka SV, Rao RR, SARS-CoV-2-associated Guillain-Barre syndrome with dysautonomia: Muscle Nerve, 2020; 62; E48-49
20.. Mohamed MA, Kaur J, Wani F, Kichloo A, Bhanot R: Am J Case Rep, 2020; 21; e926464
21.. Pek Z, Cabanilla MG, Ahmed S: BMJ Case Rep, 2021; 14; e242670
22.. Pathak V, Waite J, Chalise SN, Use of endobronchial valve to treat COVID-19 adult respiratory distress syndrome-related alveolopleural fistula: Lung India, 2021; 38; S69-71
23.. Nourié N, Chamaa MA, Mouawad S, Effective treatment with Tocilizumab in a COVID-19 patient on maintenance hemodialysis: A case report: CEN Case Rep, 2021; 10; 364-69
24.. Öz M, Kaya AG, Yakut ÖV, Successful treatment of COVID-19 infection in a patient with tracheostomy: Tuberk Toraks, 2020; 68; 444-48
25.. Darvishnia D, Sadeghi M, Sharifpour A: Infect Disord Drug Targets, 2021; 21; e160921191124
26.. , Abstracts from the 2021 Annual Meeting of the Society of General Internal Medicine: J Gen Intern Med, 2021; 36; 1-469
27.. Foschi C, Zignoli A, Gaibani P, Respiratory bacterial co-infections in Intensive Care Unit-hospitalized COVID-19 patients: Conventional culture vs BioFire FilmArray pneumonia Plus panel: J Microbiol Methods, 2021; 186; 106259
28.. Garcia-Vidal C, Sanjuan G, Moreno-García E, Incidence of co-infections and superinfections in hospitalized patients with COVID-19: A retrospective cohort study: Clin Microbiol Infect, 2021; 27; 83-88
29.. Moretti M, Van Laethem J, Minini A, Ventilator-associated bacterial pneumonia in coronavirus 2019 disease, a retrospective monocentric cohort study: J Infect Chemother, 2021; 27; 826-33
30.. Puzniak L, Finelli L, Yu KC, A multicenter analysis of the clinical microbiology and antimicrobial usage in hospitalized patients in the US with or without COVID-19: BMC Infect Dis, 2021; 21; 227
31.. Baiou A, Elbuzidi AA, Bakdach D, Clinical characteristics and risk factors for the isolation of multi-drug-resistant Gram-negative bacteria from critically ill patients with COVID-19: J Hosp Infect, 2021; 110; 165-71
32.. Bardi T, Pintado V, Gomez-Rojo M, Escudero-Sanchez R, Nosocomial infections associated to COVID-19 in the Intensive Care Unit: Clinical characteristics and outcome: Eur J Clin Microbiol Infect Dis, 2021; 40; 495-502
33.. Zhang H, Zhang Y, Wu J, Risks and features of secondary infections in severe and critical ill COVID-19 patients: Emerg Microbes Infect, 2020; 9; 1958-64
34.. Martinez-Guerra BA, Gonzalez-Lara MF, de-Leon-Cividanes NA, Antimicrobial resistance patterns and antibiotic use during hospital conversion in the COVID-19 pandemic: Antibiotics (Basel), 2021; 10; 182
35.. Sang L, Xi Y, Lin Z, Secondary infection in severe and critical COVID-19 patients in China: a multicenter retrospective study: Ann Palliat Med, 2021; 10; 8557-70
36.. Luyt C-E, Sahnoun T, Gautier M, Ventilator-associated pneumonia in patients with SARS-CoV-2-associated acute respiratory distress syndrome requiring ECMO: A retrospective cohort study: Ann Intensive Care, 2020; 10; 158
Figures
Tables
 Table 1.. MINO, TMP-SMX, LVFX, and CAZ were susceptible to Stenotrophomonas maltophilia.
Table 1.. MINO, TMP-SMX, LVFX, and CAZ were susceptible to Stenotrophomonas maltophilia. Table 2.. MINO, TMP-SMX, and LVFX were susceptible to Stenotrophomonas maltophilia.
Table 2.. MINO, TMP-SMX, and LVFX were susceptible to Stenotrophomonas maltophilia. Table 3.. Search terms used to search 3 databases (PubMed, Embase, and Ichushi) for literature review of COVID-19-associated Stenotrophomonas maltophilia.
Table 3.. Search terms used to search 3 databases (PubMed, Embase, and Ichushi) for literature review of COVID-19-associated Stenotrophomonas maltophilia. Table 4.. Literature review of COVID-19-associated Stenotrophomonas maltophilia + cases at our hospital.
Table 4.. Literature review of COVID-19-associated Stenotrophomonas maltophilia + cases at our hospital. Table 5.. List of retrospective cohort studies on Stenotrophomonas maltophilia secondary infection to COVID-19.
Table 5.. List of retrospective cohort studies on Stenotrophomonas maltophilia secondary infection to COVID-19. Table 1.. MINO, TMP-SMX, LVFX, and CAZ were susceptible to Stenotrophomonas maltophilia.
Table 1.. MINO, TMP-SMX, LVFX, and CAZ were susceptible to Stenotrophomonas maltophilia. Table 2.. MINO, TMP-SMX, and LVFX were susceptible to Stenotrophomonas maltophilia.
Table 2.. MINO, TMP-SMX, and LVFX were susceptible to Stenotrophomonas maltophilia. Table 3.. Search terms used to search 3 databases (PubMed, Embase, and Ichushi) for literature review of COVID-19-associated Stenotrophomonas maltophilia.
Table 3.. Search terms used to search 3 databases (PubMed, Embase, and Ichushi) for literature review of COVID-19-associated Stenotrophomonas maltophilia. Table 4.. Literature review of COVID-19-associated Stenotrophomonas maltophilia + cases at our hospital.
Table 4.. Literature review of COVID-19-associated Stenotrophomonas maltophilia + cases at our hospital. Table 5.. List of retrospective cohort studies on Stenotrophomonas maltophilia secondary infection to COVID-19.
Table 5.. List of retrospective cohort studies on Stenotrophomonas maltophilia secondary infection to COVID-19. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








